Abstract
Molecular dynamic simulations and 2D-NMR spectroscopy were performed to study the conformations and hydrogen-bonding interactions of ACE-inhibitory valine–isoleucine–proline (VIP) tripeptide in aqueous solution. Intra-molecular distance, solvent-accessible surface, radius of gyration and root-mean-square deviations were used to describe the properties of the VIP in aqueous solution. MD results showed that the VIP molecule is highly flexible in water and the conformations can change from extended state to folded states. The VIP molecule exists in the extended state most of the time, which is in good agreement with the 2D-NMR spectra.
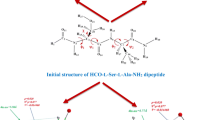
Graphical Abstract
Molecular dynamic simulations revealed that the ACE-inhibitory tri-peptide VIP is highly flexible in aqueous solution. The conformations can change from extended states to folded states. At most of the time, the VIP molecule exists in the extended state. Findings in simulations have been confirmed by 2D-NOESY spectrum.










Similar content being viewed by others
References
Poulter, N.R., Prabhakaran, D., Caulfield, M.: Hypertension. J. Lancet. 386, 801–812 (2015)
Scordo, K.A., Pickett, K.A.: Managing hypertension. J. Nursing 45, 28–34 (2015)
Russell, R.P., Whelton, P.K.: Hypertension in chronic renal failure. J. Am. Nephrol. 3, 185–192 (1983)
Kearney, P.M., Whelton, M., Reynolds, K., Muntner, P., Whelton, P.K., He, J.: Global burden of hypertension: analysis of worldwide data. J. Lancet 365, 217–223 (2005)
Ribeiro-Oliveira Jr., A., Nogueira, A., Pereira, R.M., Boas, W.W.V., Santos, R.A.S.D., Silva, A.C.S.: The reni-angiotensin system and diatetes: an update. J. Vasc. Health Risk Manag. 4, 787–803 (2008)
Yesil, S., Yesil, M., Bayata, S., Postaci, N.: ACE inhibitors and cough. J. Angiol. 45, 805–808 (1994)
Meisel, H.: food-derived bioactive proteins and peptides as components of nutraceutical. J. Curr. Pharm. Des. 13, 873–874 (2007)
Korhonen, H., Pihlanto, A.: Bioactive peptides: production and functionality. J. Int. Dairy 16, 945–960 (2006)
Wu, J.P., Aluko, R.E., Nakai, S.: Structural requirement of angiotensin I-converting enzyme inhibitory peptides: quantitative structure-activity relationship study of di- and tripeptides. J. Agric. Food Chem. 54, 732–738 (2006)
Wu, S.F., Qi, W., Su, R.X., Li, T.H., Lu, D., He, Z.M.: COMFA and COMSIA analysis of ACE-inhibitory, antimicrobial and bitter-tasting peptides. Eur. J. Med. Chem. 84, 100–106 (2014)
Gu, Y.C., Majumder, K., Wu, J.P.: QSAR-aided in silico approach in evaluation of food proteins as precursors of ACE-inhibitory peptides. J. Food Res. Int. 44, 2465–2474 (2011)
Huang, G.D., Zhang, R., Luo, Y.P., Zeng, W., Wu, W.J.: Studies on the QSAR of ACE inhibitory tripeptides with proline as C-terminal and determination inhibitory activities. Chin. J. Struct. Chem. 33, 1741–1748 (2014)
Li, G.H., Liu, H., Shi, Y.H., Le, G.W.: Antihypertensive effect of alcalase generated mung bean protein hydrolysates in spontaneously hypertensive rats. J. Pharm. Biomed. Anal. 37, 219–224 (2005)
Zhang, R., Huang, J.M., Meng, X., Wu, W.J.: Molecular dynamics simulations and NMR experimental study of oxidized glutathione in aqueous solution. J. Solution Chem. 41, 879–887 (2012)
Zhang, R., Huang, G.D., Zeng, W., Wu, W.J.: Conformations of oxidized glutathione in aqueous urea solution by all-atom molecular dynamic simulations and 2D-NOESY spectrum. J. Solution Chem. 42, 2229–2239 (2013)
Yang, J., Aslimovska, L., Glaubitz, C.: Molecular dynamics of proteorhodopsin in lipid bilayers by solid-state NMR. J. Am. Chem. Soc. 133, 4874–4881 (2011)
Rosenzweig, R., Kay, L.E.: Bringing dynamic molecular machines into focus by methyl-trosy NMR. J. Ann. Rev. Biochem. 83, 291–315 (2014)
Nygaard, R., Zou, Y., Dror, R.O., Mildorf, T.J., Arlow, D.H., Manglik, A., Pan, A.C., Liu, C.W., Fung, J.J., Bokoch, M.P.: The dynamic process of β2-adrenergic receptor activation. J. Cell. 152, 532–542 (2013)
Rosenman, D.J., Connors, C.R., Chen, W., Wang, C., García, A.E.: A β monomers transiently sample oligomer and fibril-like configurations: ensemble characterization using a combined MD/NMR approach. J. Mol. Biol. 425, 3338–3359 (2013)
Hazelbaker, E.D., Budhathoki, S., Katihar, A., Shah, J.K., Maginn, E.J., Vasenkov, S.: Combined application of high-filed diffusion NMR and molecular dynamics simulations to study dynamics in a mixture of carbon dioxide and an imidazolium-based ionic liquid. J. Phys. Chem. B 116, 9141–9151 (2012)
Huang, G.D., Zhang, R., Zeng, W., Chen, L., Wu, W.J.: Conformations and interactions of ACE inhibitor tripeptide in aqueous and DMSO solution by all-atom MD simulations and 2D-NMR spectra. J. Mol. Liq. 206, 75–81 (2015)
Kobayashi, K., Liang, Y.F., Matsuoka, T.: Molecular dynamics study of aqueous NaCl solutions: flash crystallization caused by solution phase change. J. Solution Chem. 43, 1799–1809 (2014)
Jorgensen, W.L., Swenson, C.J.: Optimized intermolecular potential functions for amides and peptides structure and properties of liquid amides. J. Am. Chem. Soc. 107, 569–578 (1985)
Jorgensen, W.L., Maxwell, D.S., Tirado-Rives, J.: Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J. Am. Chem. Soc. 118, 11225–11236 (1996)
Mark, P., Nilsson, L.: Structure and dynamics of the TIP3P, SPC, and SPC/E water models at 298 K. J. Phys. Chem. A 105, 9954–9960 (2001)
Berweger, C.D., Gunsteren, W.F., Muller-Plathe, F.: Force field parametrization by weak coupling teengineering SPC water. J Chem. Phys. Lett. 232, 429–436 (1995)
Ponder, J.W.: TINKER: Software Tools for Molecular Design 7.1. Washington University School of Medicine, Saint Louis (2015)
Lei, Y., Li, H., Pan, H., Han, S.: Structures and hydrogen bonding analysis of N,N-dimethylformamide and N,N-dimethylformamide–water mixtures by molecular dynamics simulations. J. Phys. Chem. A 107, 1574–1583 (2003)
Connolly, M.L.: Analytical molecular surface calculation. J. Appl. Crystallogr. 16, 548–558 (1983)
Clairac, R.P.L., Geierstanger, B.H., Mrksich, M., Dervan, P.B., Wemmer, D.E.: NMR characterization of hairpin polyamide complexes with the minor groove of DNA. J. Am. Chem. Soc. 119, 7909–7916 (1997)
Zheng, G., Stait-Gardner, T., Anil Kumar, P.G., Torres, A.M., Price, W.S.: PGSTE-WATERGATE: an STE-based PGSE NMR sequence with excellent solvent suppression. J. Magn. Reson. 191, 159–163 (2008)
Lei, Y., Li, H., Zhang, R., Han, S.: Molecular dynamics simulations of biotin in aqueous solution. J. Phys. Chem. B. 108, 10131–10137 (2004)
Schedlbauer, A., Coudevylle, N., Auer, R., Kloiber, K., Tollinger, M., Konrat, R.: Autocorrelation analysis of NOESY data provides residue compactness for folded and unfolded proteins. J. Am. Chem. Soc. 131, 6038–6039 (2009)
Lo, J., Yen, H., Tsai, C., Chen, B., Hou, S.: Interaction between hydrophobically modified 2-hydroxyethyl cellulose and sodium dodecyl sulfate studied by viscometry and two-dimensional NOE NMR spectroscopy. J. Phys. Chem. B 118, 6922–6930 (2014)
Khodov, I.A., Nikiforov, M.Y., Alper, G.A., Blokhin, D.S., Efimov, S.V., Klochkov, V.V., Georgi, N.: Spatial structure of felodipine dissolved in DMSO by 1D NOE and 2D NOESY NMR spectroscopy. J. Mol. Struct. 1035, 358–362 (2013)
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Nos: 21202021, 20903026), the Talents Introduction Foundation for Universities of Guangdong Province (2011), Natural Science Foundation of Shang-Hai (12ZR1440700), and the Science and Technology Planning Project of Guangzhou (No. 2013J4100071).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qi, C., Chen, L., Wei, H. et al. Conformational Features of ACE-Inhibitory Valine-Isoleucine-Proline Tripeptide in Aqueous Medium According to Molecular Dynamic Simulations and 2D-NMR Spectroscopy. J Solution Chem 45, 1213–1226 (2016). https://doi.org/10.1007/s10953-016-0505-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-016-0505-7