Abstract
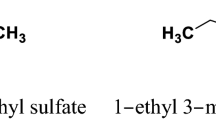
The apparent molar volumes and viscosities of N,N′-bis(salicylaldehyde)-1,3-diaminopropane Schiff base (Salpr) have been determined in ionic liquid {1-pentyl-3-methylimidazolium bromide ([PnMIm]Br)} + N,N-dimethylformamide (DMF) solutions at 298.15 K from density and viscosity measurements using a vibrating tube densimeter and Übbelohde type viscometer, respectively. These data have been used to calculate standard partial molar volumes, \(V_{\phi} ^{0}\), transfer partial molar volumes, Δtr V 0, and viscosity B-coefficients of the solutions. The transfer partial molar volumes are negative, and decrease with increasing the concentration of ionic liquid for all of the investigated solutions. It found that this ionic liquid interacts strongly with the Schiff base (Salpr) and has desolvation effect on the Schiff base molecules.



Similar content being viewed by others
References
Habibi, M.H., Montazerozohori, M., Lalegani, A., Harrington, R.W., Clegg, W.: Synthesis, structural and spectroscopic properties of a new Schiff base ligand N,N′-bis(trifluoromethylbenzylidene)ethylenediamine. J. Fluorine Chem. 127, 769–773 (2006)
Kovbasyuk, L.A., Fritzky, I.O., Kokozay, V.N., Iskenderov, T.S.: Synthesis and structure of diaqua-bis(ethylenediamine)copper(II) salts with anions of carbamic acids. Tetrahedron 16, 1723–1729 (1997)
Ho, C.-W., Cheng, W.-C., Cheng, M.-C., Peng, S.-M., Cheng, K.-F., Che, C.-M.: Preparation and reactivities of chiral manganese(III) and copper(II) complexes of binaphthyl Schiff bases. J. Chem. Soc. Dalton Trans. 405–414 (1996)
Larrow, J.F., Jacobsen, E.N., Gao, Y., Hong, Y., Nie, X., Zepp, C.M.: Practical method for the large-scale preparation of N,N′-[bis(3,5-di-tertbutylsalicylidene)-1,2-cyclohexanediaminato(2-)]manganese(III) chloride, a highly enantioselective epoxidation catalyst. J. Org. Chem. 59, 1939–1942 (1994)
Jacobsen, E.N.: In: Ojima, I. (Ed.) Catalytic Asymmetric Syntheses. VCH, New York (1993). Chap. 4.2
Irie, R., Noda, K., Ito, Y., Matsumoto, N., Katsuki, T.: Process for preparing chiral epoxides using chiral manganese triazanonane complexes as oxidation catalysts. Tetrahedron Lett. 31, 7345–7348 (1990)
Srinivasan, K., Kochi, J.K.: Synthesis and molecular structure of oxochromium(V) cations. Coordination with donor ligands. Inorg. Chem. 24, 4671–4679 (1985)
Plays, B.J., Bukowska, J., Jackowska, K.: SERS of 1,8-diaminonaphthalene on gold, silver and copper electrodes. Polymerisation and complexes formed with the electrode material. J. Electroanal. Chem. 428, 19–24 (1997)
Aly, A.A., El-Shaieb, K.M.: Reaction of 1,8-diaminonaphthalene with some selected π-acceptors; prospective optically active non-linear cyanovinylated naphthalenes as well as synthesis of novel perimidin and pleiadene derivatives. Tetrahedron 60, 3797–3802 (2004)
Yakuphanoglua, F., Sekerci, M.: Determination of the optical constants of Co(II) complex of Schiff base obtained from 1,8-diaminonaphthalene thin film by infrared spectra. J. Mol. Struct. 751, 200–2003 (2005)
Kanthimathi, M., Dhathathreyan, A.: Synthesis and photoreaction of Schiff bases derived from p-nitro cinnamaldehyde and diamines in Langmuir and Langmuir–Blodgett films. Chem. Phys. Lett. 367, 193–198 (2003)
Kumar, S., Dhar, D.N., Saxena, P.N.: Applications of metal complexes of Schiff bases—a review. J. Sci. Ind. Res. 68, 181–187 (2009)
Abdel-Gader, A.M., Masoud, M.S., Khalil, E.A., Shehata, E.E.: Electrochemical study on the effect of Schiff base and its cobalt complex on the acid corrosion of steel. Corrosion Sci. 51, 3021–3024 (2009)
Chen, L.-L., Qiu, Z.-M., Huang, J.-L., Xu, L.-F.: Study on the extraction of heavy metal ions with Schiff base derived from PMBP-2-aminobenzothiazole/ionic liquid binary–water phase system. Yejin Fenxi/Metallurgical Analysis 30, 33–37 (2010)
Ramakrishna, D., Bhat, B.R., Karvembu, R.: Catalytic oxidation of alcohols by nickel(II) Schiff base complexes containing triphenylphosphine in ionic liquid: an attempt towards green oxidation process, catalysis. Catal. Commun. 11, 498–501 (2010)
Belibagli, K.B., Ayranci, E.: Viscosity and apparent molar volumes of some amino acids in water and in 6 M guanidine hydrochloride at 25 °C. J. Solution Chem. 19, 867–882 (1990)
Yang, J.Z., Tong, J., Li, J.B.: Study of the volumetric properties of the aqueous ionic liquid 1-pentyl-3-methyl imidazolium tetrafluoroborate. J. Solution Chem. 36, 573–582 (2007)
Pei, Y., Wang, J., Liu, L., Wu, K., Zhao, Y.: Liquid–liquid equilibria of aqueous biphasic systems containing selected imidazolium ionic liquids and salts. J. Chem. Eng. Data 52, 2026–2031 (2007)
Holbrey, J.H., Rojers, R.D.: Clean synthesis of 1,3-dialkylimidazolium ionic liquid. Abstr. Pap. Am. Chem. Soc. 224, U612 (2002)
Holbrey, J.D., Seddon, K.R.: The phase behaviour of 1-alkyl-3-methylimidazolium tetrafluoroborates; ionic liquids and ionic liquid crystals. J. Chem. Soc. Dalton Trans. 2133–2140 (1999)
Zamyatnin, A.A.: Amino acid, peptide, and protein volume in solution. Annu. Rev. Biophys. Bioeng. 13, 145–165 (1984)
Zhao, C.A., Ma, P.B., Li, J.: Partial molar volumes and viscosity B-coefficients of arginine in aqueous glucose, sucrose and L-ascorbic acid solutions at T=298.15 K. J. Chem. Thermodyn. 37, 37–42 (2005)
Pal, A., Chauhan, N.: Volumetric, viscometric, and acoustic behaviour of diglycine in aqueous saccharide solutions at different temperatures. J. Mol. Liq. 149, 29 (2009)
Shekaari, H., Bezaatpour, A., Soltanpour, A.: Partial molar volumes of N,N′-1,2-ethyl-bis(salicyladimine) Schiff base (salen) in organic solvents at T=283.15 to 318.15 K. J. Chem. Eng. Data 55, 5927–5931 (2010)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shekaari, H., Bezaatpour, A. & Elhami, R. Volumetric and Viscometric Studies of N,N′-Bis(salicylaldehyde)-1,3-diaminopropane Schiff Base (Salpr) in Ionic Liquid + DMF solutions. J Solution Chem 41, 516–524 (2012). https://doi.org/10.1007/s10953-012-9811-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-012-9811-x