Abstract
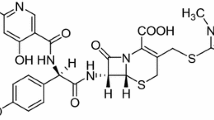
The solubility parameter of gatifloxacin was calculated theoretically by Fedors’ method and also determined experimentally using the standard solubility method. The molar volume of gatifloxacin was determined experimentally by the flotation method. Three solvents, ethyl acetate, propylene glycol and water were used to prepare nine binary mixtures having different solubility parameter values varying from 8.9 to 23.4 H. The δ 2 solubility parameter of gatifloxacin was found to be 12.4 H. The cumulative transport of gatifloxacin from the binary solvent mixtures (ethyl acetate–propylene glycol and propylene glycol–water) exhibited an inverse parabolic relationship to the solubility parameter of the solvent mixtures. Transport studies revealed that the extent of gatifloxacin rejection from solvent mixtures was dependent on its solubility parameter and system composition. In the selected organisms (bacteria), a minimum zone of inhibition was observed in a solvent mixture having the solubility parameter nearest to the solubility parameter of gatifloxacin. Two models were employed for the evaluation of the antibacterial activity of gatifloxacin in binary mixtures.







Similar content being viewed by others
References
James, K.C.: Solubility and Related Properties. Dekker, New York (1986)
Mullins, L.: Some physical mechanism in narcosis. Chem. Rev. 54, 289–323 (1954)
Khalil, S.A., Abdullah, O.A., Moustafa, M.A.: The use of the solubility parameter as an index of drug activity. Can. J. Pharm. Sci. 11, 121–126 (1976)
Khalil, S.A., Abdallah, O.A., Moustafa, M.A.: Absorption of some barbiturates by gambusia fish and its correlation to solubility parameter. Can. J. Pharm. Sci. 11, 26–30 (1976)
Khalil, S.A., Martin, A.N.: Drug transport through model membranes and its correlation with solubility parameters. J. Pharm. Sci. 56, 1223–1233 (1967)
Adjei, A., Newburger, J., Stavchansky, S., Martin, A.: Membrane solubility parameter and in situ release of theophylline. J. Pharm. Sci. 73, 742–745 (1984)
Beckett, A.M., Stenlake, J.B.: Practical Pharmaceutical Chemistry, vol. 2, 3rd edn. CBS Publisher, New Delhi (1986)
Fedors, R.F.: A method of estimating both the solubility parameters and molar volumes of liquids. Polym. Eng. Sci. 14, 147–154 (1974)
Subrahmanyam, C.V.S.: Essentials of Physical Pharmacy. Vallabh Prakashan, New Delhi (2003)
Ananthanarayan, R., Paniker, C.K.J.: Textbook of Microbiology, 5th edn. Longman, Chennai (1997)
Adjei, A., Newburger, J., Martin, A.: Extended Hildebrand approach: solubility of caffeine in dioxane–water mixtures. J. Pharm. Sci. 69, 659–661 (1980)
Chertkoff, M.J., Martin, A.N.: The solubility of benzoic acid in mixed solvents. J. Am. Pharm. Assoc. (Sci. Ed.) 49, 444–447 (1960)
Gradon, J.L.: Encyclopedia of Polymer Science and Technology. Wiley, New York (1965)
Martin, A., Newburger, J., Adjei, A.: Extended Hildebrand solubility approach: II. Solubility of theophylline in polar binary solvents. J. Pharm. Sci. 69, 487–491 (1980)
Gilman, A.G.: The Pharmacological Basis of Therapeutics, 9th edn. McGraw-Hill, New York (1996)
Saeed, E., Abbas, S., Alireza, F.: Quinolones: Recent structural and clinical developments. Iran. J. Pharm. Res. 3, 123–136 (2005)
Martin, A., Swarbrick, J., Cammarata, A.: Physical Pharmacy, 3rd edn. Lea and Febiger, Philadelphia (1983)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kopparam, M., Subrahmanyam, C.V.S., Juturu, T. et al. Solubility Parameter of Gatifloxacin and Its Correlation with Antibacterial Activity. J Solution Chem 41, 381–391 (2012). https://doi.org/10.1007/s10953-012-9803-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-012-9803-x