Abstract
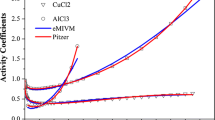
The mean spherical approximation (MSA) model, coupled with two hard sphere models, was used to predict the activity coefficients of mixtures of electrolyte solutions at different temperatures and concentrations. The models, namely the Ghotbi-Vera-MSA (GV-MSA) and Mansoori et al.-MSA (BMCSL-MSA), were directly used without introducing any new adjustable parameters for mixing of electrolyte solutions. In the correlation step, the anion diameters were considered to be constant, whereas the cation diameters were considered to be concentration dependent. The adjustable parameters were determined by fitting the models to the experimental mean ionic activity coefficients for single aqueous electrolytes at fixed temperature. The results showed that the studied models predict accurately the activity coefficients for single electrolyte aqueous solutions at different temperatures. In the systems of binary aqueous electrolyte solutions with a common anion, the GV-MSA model has slightly better accuracy in predicting the activity coefficients. Also, it was observed that the GV-MSA model can more accurately predict the activity coefficients for ternary electrolyte solutions with a common anion, especially at higher concentrations.


Similar content being viewed by others
Abbreviations
- R :
-
universal gas constant (J⋅K−1⋅mol−1)
- m :
-
molality (mol⋅(kg of solvent)−1)
- c :
-
molarity (mol⋅dm−3)
- D :
-
dielectric constant
- T :
-
absolute temperature (K)
- t :
-
relative temperature (°C)
- d :
-
diameter of ions (m)
- k :
-
Boltzmann constant (J⋅K−1)
- I :
-
ionic strength of solution (mol⋅kg−1)
- z :
-
ionic charge
- e :
-
unit electronic charge (J⋅m)0.5
- AARD%:
-
percent of average absolute relative deviation
- Y i :
-
volume, independent group
- γ ± :
-
mean ionic activity coefficient
- Γ:
-
inverse shielding length (m−1)
- ρ :
-
number density (m−3)
- υ :
-
stoichiometric coefficient
- elec:
-
electrostatic
- hs:
-
hard sphere
References
Prausnitz, J.M., Lichtenthaler, R.N., de Azevedo, E.G.: Molecular Thermodynamics of Fluid Phase Equilibria, 3rd ed. Prentice Hall, New York (1999)
Narayanan, K.V., Ananth, M.S.: A modified SCLC model for the thermodynamic properties of single and mixed electrolytes. Fluid Phase Equilib. 114, 89–121 (1996)
Moggia, E., Bianco, B.: Mean activity coefficient of electrolyte solutions. J. Phys. Chem. B 111, 3183–3191 (2007)
Chen, C.C.: Toward development of activity coefficient models for process and product design of complex chemical systems. Fluid Phase Equilib. 24, 103–112 (2006)
Blum, L.: Mean spherical model for asymmetric electrolytes. I: Method of solution. Mol. Phys. 30, 1529–1535 (1975)
Liu, W.B., Li, Y.G., Lu, J.F.: Correlation of mean ionic activity coefficients of electrolyte solutions by the simplified non primitive mean spherical approximation. Fluid Phase Equilib. 162, 131–141 (1999)
Seyfkar, N., Ghotbi, C., Taghikhani, V., Azimi, G.: Application of the non-primitive MSA-based models in predicting the activity and the osmotic coefficients of aqueous electrolyte solutions. Fluid Phase Equilib. 221, 189–196 (2004)
Salimi, H.R., Taghikhani, V., Ghotbi, C.: Application of the GV-MSA model to the electrolyte solutions containing mixed salts and mixed solvents. Fluid Phase Equilib. 231, 67–76 (2005)
Yu, Y.X., Gao, G.H., Li, Y.G.: Surface tension for aqueous electrolyte solutions by the modified mean spherical approximation. Fluid Phase Equilib. 173, 23–38 (2000)
Lu, J.F., Yu, Y.X., Li, Y.G.: Modification and application of the mean spherical approximation method. Fluid Phase Equilib. 85, 81–100 (1993)
Ghotbi, C., Azimi, G., Taghikhani, V., Vera, J.H.: On the correlation of the activity coefficients in aqueous electrolyte solutions using the K-MSA model. Ind. Eng. Chem. Res. 42, 1279–1284 (2003)
Sadeghi, M., Taghikhani, V., Ghotbi, C.: Application of the MSA-based models in correlating the surface tension for single and mixed electrolyte solutions. J. Chem. Thermodyn. 41, 1264–1271 (2009)
Ghotbi, C., Vera, J.H.: A general expression for the ordered-packed volume fraction of hard spheres of different diameters. Ind. Eng. Chem. Res. 41, 1122–1128 (2002)
Robinson, R.A., Stokes, R.H.: Electrolyte Solutions. Butterworth, London (1959)
Tippetts, E.A., Newton, R.F.: The thermodynamics of aqueous barium chloride solutions from electromotive force measurements. J. Am. Chem. Soc. 56, 1675–1680 (1934)
Ananthaswamy, J., Atkinson, G.: Thermodynamics of concentrated electrolyte mixtures. 5. A review of the thermodynamic properties of aqueous calcium chloride in the temperature range 273.15–373.15 K. J. Chem. Eng. Data 30, 120–128 (1985)
Faita, G., Mussini, T., Oggioni, R.: Thermodynamic functions of aqueous hydrobromic acid at various concentrations and temperatures. J. Chem. Eng. Data 11, 162–165 (1966)
Holmes, H.F., Mesmer, R.E.: An isopiestic study of aqueous solutions of the alkali metal bromides at elevated temperatures. J. Chem. Thermodyn. 30, 723–741 (1998)
Åkerlöf, G., Kegeles, G.: Thermodynamics of concentrated aqueous solutions of sodium hydroxide. J. Am. Chem. Soc. 62, 620–640 (1940)
Holmes, H.F., Mesmer, R.E.: Isopiestic molalities for aqueous solutions of the alkali metal hydroxides at elevated temperatures. J. Chem. Thermodyn. 30, 311–326 (1998)
Rogers, P.S.Z., Pitzer, K.S.: High-temperature thermodynamic properties of aqueous sodium sulfate solutions. J. Phys. Chem. 85, 2886–2895 (1981)
Mortazavi-Manesh, S., Taghikhani, V., Ghotbi, C.: Modification of the GV-MSA model in obtaining the activity and osmotic coefficients of aqueous electrolyte solutions. Fluid Phase Equilib. 240, 167–172 (2006)
Robinson, R.A., Harned, H.S.: Some aspects of the thermodynamics of strong electrolytes from electromotive force and vapor pressure measurements. Chem. Rev. 28, 419–476 (1941)
Harned, H.S., Cook, M.A.: The thermodynamics of aqueous potassium chloride solutions from electromotive force measurements. J. Am. Chem. Soc. 59, 1290–1292 (1937)
Harned, H.S., Crawford, C.C.: The thermodynamics of aqueous sodium bromide solutions from electromotive force measurements. J. Am. Chem. Soc. 59, 1903–1905 (1937)
Lanier, R.D.: Activity coefficients of sodium chloride in aqueous three-component solutions by cation-sensitive glass electrodes. J. Phys. Chem. 69, 3992–3998 (1965)
Deyhimi, F., Salamat-Ahangari, R., Karimzadeh, Z.: Thermodynamic investigation of KCl in the ternary KCl/LiCl/H2O mixed electrolyte system based on potentiometric method. Calphad 31, 522–528 (2007)
Harned, H.S.: The activity coefficient of hydrochloric acid in concentrated solutions of strong electrolytes. J. Am. Chem. Soc. 48, 326–342 (1926)
Lindenbaum, S., Rush, R.M., Robinson, R.A.: Osmotic and activity coefficients for mixtures of lithium chloride with barium chloride and cesium chloride with barium chloride in water at 298.15 K. J. Chem. Thermodyn. 4, 381–389 (1972)
Ji, X., Lu, X., Li, S., Zhang, L., Wang, Y., Shi, J.: Activity coefficients of HCl in the HCl+NH4Cl+H2O systems at 298.15 and 313.15 K. J. Chem. Eng. Data 45, 29–33 (2000)
Harned, H.S., Copson, H.R.: The dissociation of water in lithium chloride solutions. J. Am. Chem. Soc. 55, 2206–2215 (1933)
Harned, H.S., Gary, R.: The activity coefficient of hydrochloric acid in concentrated aqueous higher valence type chloride solutions at 25 °C. I. The system hydrochloric acid-barium chloride. J. Am. Chem. Soc. 76, 5924–5927 (1954)
Harned, H.S., Gary, R.: The activity coefficient of hydrochloric acid in concentrated aqueous higher valence type chloride solutions at 25 °C. II. The system hydrochloric acid-strontium chloride. J. Am. Chem. Soc. 77, 1994–1995 (1955)
Manohar, S., Sarada, S., Ananthaswamy, J.: Thermodynamics of electrolyte solutions: an e.m.f. study of the activity coefficients of KCl in {KCl(mA)+CaCl2(mB)}(aq) at 298.15, 308.15, and 318.15 K. J. Chem. Thermodyn. 21, 969–976 (1989)
Roy, R.N., Gibbons, J.J., Ovens, L.K., Bliss, G.A., Hartley, J.J.: Activity coefficients for the system HCl+CaCl2+H2O at various temperatures: Applications of Pitzer’s equations. J. Chem. Soc. Faraday Trans. 78, 1405–1422 (1981)
Khoo, K.H., Lim, T.K., Chan, C.Y.: Activity coefficients for the system HCl+CoCl2+H2O at 298.15 K–effects of higher order electrostatic terms. J. Chem. Soc. Faraday Trans. 74, 2037–2042 (1978)
Khoo, K.H., Chan, C.: Activity coefficient of hydrochloric acid in the system hydrochloric acid–ammonium chloride–potassium chloride–water at constant total molality 0.5 mol⋅kg−1 at 298.15 K. J. Chem. Eng. Data 24, 28–30 (1979)
Jiang, C.: Activity coefficients of hydrochloric acid in concentrated electrolyte solutions. 2. HCl+BaCl2+KCl+H2O, HCl+LiCl+KCl+H2O, and HCl+NaCl+KCl+H2O at 298.15 K. J. Chem. Eng. Data 41, 117–120 (1996)
Reilly, P.J., Wood, R.H., Robinson, R.A.: Prediction of osmotic and activity coefficients in mixed-electrolyte solutions. J. Phys. Chem. 75, 1305–1315 (1971)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadeghi, M., Ghotbi, C. & Abdekhodaie, M.J. Activity Coefficient Prediction for Binary and Ternary Aqueous Electrolyte Solutions at Different Temperatures and Concentrations. J Solution Chem 41, 75–88 (2012). https://doi.org/10.1007/s10953-011-9789-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-011-9789-9