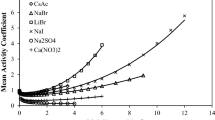
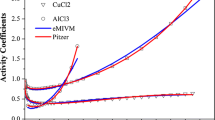
Abstract
The Debye–Hückel and non-Debye–Hückel contributions to the Gibbs energy interaction parameters are investigated for electrolyte (E) + non-electrolyte (N) + water (W) systems. A method is proposed for calculating the interaction parameters, C DH n ,C N n , and C T n , which represent the Debye–Hückel, non-Debye–Hückel, and total contributions, respectively. Four ternary E + N + W systems are chosen and the interaction parameters are computed with different forms of the Debye–Hückel equation. Results show that: (1) the Gibbs energy interaction parameters between E and N can be divided into two parts: the Debye–Hückel contribution and the non-Debye–Hückel contribution, C T n =C DH n +C N n ; (2) the signs and magnitudes of the Debye–Hückel contribution to the interaction parameters, C DH n , depend mainly on the change in the dielectric constant of the solvent due to the addition of the non-electrolyte into the solvent; and (3) when the addition of the non-electrolyte only affects slightly the dielectric constant of the solvent, C DH1 (indicating the Debye–Hückel contribution to the interaction parameter for E + N) has a very small value and consequently can be neglected. In general, C DH1 is large, even larger than C N1 .
Similar content being viewed by others
References
Scatchard, G.: Equilibria in non-electrolyte solutions in relation to the vapor pressures and densities of the components. Chem. Rev. 8, 321–333 (1931)
Scatchard, G.: Equilibrium in non-electrolyte mixtures. Chem. Rev. 44, 7–35 (1949)
Scatchard, G., Raymond, C.J.: Vapor-liquid equilibrium. II. Chloroform-ethanol mixtures at 35, 45 and 55 °C. J. Am. Chem. Soc. 60, 1278–1287 (1938)
McMillan, W.G., Mayer, J.E.: The statistical mechanics of multicomponent systems. J. Chem. Phys. 13, 276–305 (1945)
Scatchard, G.: Excess free energy and related properties of solutions containing electrolytes. J. Am. Chem. Soc. 90, 3124–3127 (1968)
Briggs, C.C., Lilley, T.H., Rucherford, J., Woodhead, S.: Activity of calcium chloride in aqueous solutions of some amino acids at 25 °C. J. Solution Chem. 3, 649–658 (1974)
Khoshkbarchi, M.K., Vera, J.H.: Measurement of activity coefficients of amino acids in aqueous electrolyte solutions: experimental data for the systems H2O + NaCl + glycine and H2O + NaCl + DL-alanine at 25 °C. Ind. Eng. Chem. Res. 35, 829–836 (1996)
Khoshkbarchi, M.K., Soto-Campos, A.M., Vera, J.H.: Interactions of DL-serine and L-serine with NaCl and KC1 in aqueous solutions. J. Solution Chem. 26, 941–948 (1997)
Perron, G., Joly, D., Desnoyers, J.E., Avedikian, L., Morel, J.P.: Thermodynamics of the salting effect, free energies, enthalpies, entropies, heat capacities, and volumes of the ternary systems electrolyte-alcohol–water at 25 °C. Can. J. Chem. 56, 552–559 (1978)
Desnoyers, J.E., Perron, G., Avédidikian, L., Morel, J.P.: Enthalpies of the urea-tert-butanol–water at 25 °C. J. Solution Chem. 5, 631–644 (1976)
Visser, C.D., Perron, G., Desnoyers, J.E.: Volumes and heat capacities of ternary aqueous systems at 25 °C mixtures of urea tert-butyl alcohol, dimethyl formamide, and water. J. Am. Chem. Soc. 99, 5894–5900 (1977)
Zhuo, K.L., Wang, J.J., Liu, W.B., Lu, J.S.: Thermodynamic parameters of interaction of HCl with alcohols in water III. HCl–ethanol–water system at 278.15–318.15 K. Chin. J. Chem. 14, 193–199 (1996)
Zhuo, K.L., Wang, J.J., Wang, H.Q.: Activity coefficients for NaCl-monosaccharide (D-glucose, D-galactose, D-xylose, D-arabinose)—water systems at 298.15 K. Carbohydr. Res. 325, 46–55 (2000)
Robinson, R.A., Stokes, R.H.: Electrolyte Solutions, 2nd edn. Butterworth, London (1959)
Zhuo, K.L., Wang, J.J.: Activity coefficients on different concentration scales and reference states. Am. Inst. Chem. Eng. J. 52, 2988–2990 (2006)
Zhuo, K.L.: Thermodynamic pair interaction parameters on various concentration scales. J. Phys. Chem. B 109, 7460–7462 (2005)
John, A.: Dean Lange’s Handbook of Chemistry, 11th edn., vol. 10, pp. 85–146 (1973)
Zhuo, K.L., Liu, Q., Wang, Y.P., Wang, J.J.: Volumetric and viscosity properties of monosaccharides in aqueous amino acid solutions at 298.15 K. J. Chem. Eng. Data 51, 919–927 (2006)
Haki, A.W., Duke, M.M., Klassen, S.A., Mckey, R.M., Preuss, K.E.: Apparent molar heat capacities and volumes of some aqueous solutions of diphatic amino acid at 298.15 K, 315.15 K, 328.15 K. Can. J. Chem. 72, 362–368 (1994)
Hasted, J.B., Ritson, D.M., Collie, C.H.: Dielectric properties of aqueous ionic solutions. Part I and Part II. J. Chem. Phys. 16, 1–21 (1948)
Satio, A., Miyawaki, O., Nakamura, K.: Dielectric relaxation of aqueous solution with low-molecular-weight non-electrolytes and its relationship with solution structure. Biosci. Biotech. Biochem. 61, 1831–1835 (1997)
Feakins, D., Knox, M., Hickey, B.E.: Studies in ions solution in non-aqueous solvent and their aqueous mixtures. J. Chem. Soc. Faraday Trans. I 80, 961–968 (1984)
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Zhuo, K., Ren, Q., Zhao, Y. et al. Debye–Hückel Contributions to the Gibbs Energy Interaction Parameters for Ternary Electrolyte + Non-Electrolyte + Water Systems. J Solution Chem 37, 283–289 (2008). https://doi.org/10.1007/s10953-007-9232-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9232-4