Abstract
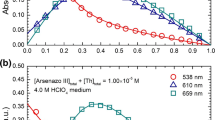
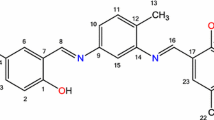
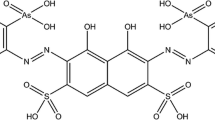
Protometric studies were performed in aqueous solutions at 25^C and 0.1 ,mol.dm−3 ionic strength (NaClO4) to determine the complexing abilities of eight (o-hydroxy-phenyl) mono- and di-methylenephosphonic acids (differently substituted by chromophoric or auxochromic groups) towards thorium(IV). The number, the nature of the species present in solution, their overall stability constants over a broad acidity range and their individual electronic spectra, as resolved by computation, have been determined by potentiometry and UV-visible spectrophotometry.
The formation of 1:1 species, partially protonated MLHx and totally deprotonated [ML], as well as hydroxo species -- mononuclear ML(OH)x and dinuclear M2L(OH) x is reported with thorium(IV). The results show that the complexing power, which is not very different in the lanthanide series, is much higher for thorium(IV). The ratio Th4+/Eu3+ reaches eight log10 units with some of the ligands.
Similar content being viewed by others
References
Kabachnik, M.I., Laslovskii, R.P., Medved, T.Y., Medynster, V.V., Kolpakova, I.D., Dyatlova, N.M.: Complex-forming properties of hydroxyethylidenediphosphonic acid in aqueous solutions. Proc. Acad. Sci. (USSR) 177, 1060–1063 (1967)
Novikov, A., Korpusov, S., Zhou, R.N., Geckeler, K.E.: Trennung und Anreicherung von Aktiniden mit Hilfe eines Phosphonsäure-Polymeren. Chemischa Technik 45, 464–466 (1993)
Sabharwal, K.N., Rao, P.R.V., Svinivassan, M.: Extraction of actinides by bifunctional phosphonic acid resin. Solv. Extr & Ion Exch. 12, 1085–???? (1994)
Nash, K.L.: Actinide phophonate complexes in aqueous solutions. J. Alloy Compounds 213, 300–304 (1994)
Tao, Z., Gao, H.: Use of the ion exchange method for the determination of stability constants of thorium with humic and fulvic acids. Radiochim Acta 65, 121–123 (1994)
Karayannis, N.M., Mikulski, C.M., Strocko, M.J., Pytlewski, L.L., Labes, M.M.: Uranium(IV) and thorium(IV) tetrakis-(dialkoxyphosphato) and tetrakis-(alkoxy-alkylphosphonato) polynuclear complexes. Inorg. Chim. Acta 4, 455–459 (1970)
Carey, G.H., Martell, A.E.: Formation, hydrolysis, and olation of uranium(IV) chelates. J. Am. Chem. Soc. 90, 32–38 (1968)
Kinard, W.F., Grant, P.M., Baisden, P.A.: Calorimetric determination of the enthalpies of complexation of thorium(IV) with amine- N-polycarboxylic acids. Polyhedron 8, 2385–2388 (1989)
Bohigian, T.A., Martell, A.E.: Metal chelates of triethylenetetraaminehexaacetic acid. Inorg. Chem. 4, 1264–1270 (1965)
Benghanem, F., Chafaa, S., Bouet, G.M., Khan, M.A.: Potentiometric studies of 4-mono(dihydroxyphosphonyl) methyl phenol and 2,6-bis(dihydroxyphosphonyl) methyl 4-methyl phenol complexes with cobalt(II), nickel(II), copper(II), zinc(II) and cadmium(II). phosphorous. Sulfur, Silicon 170, 159–169 (2001)
Holy, A., Günter, J., Dvorakova, H., Masojidkova, M., Andrei, G., Snoeck, R., Balzarini, J., De Clercq, E.: Structure-antiviral activity relationship in the series of pyrimidine and purine N-[2-(2-phosphonomethoxy)ethyl] nucleotide analogues. 1. Derivatives substituted at the carbon atoms of the base. J. Med. Chem. 42, 2064–2086 (1999)
Perez-Jurado, L.A., Wang, Y.K., Peoples, R., Coloma, A., Cruces, J., Francke, U.: A duplicated gene in the breakpoint regions of the 7q11.23 Williams-beuren syndrome deletion encodes the initiator binding protein TFII-I and BAP-135, a phosphorylation target of BTK. Hum. Mol. Genet. 7, 325–334 (1998)
Rae, C., Karmiloff-Smith, A., Lee, M.A., Dixon, R.M., Grant, J., Blamire, A.M., Thompson, C.H., Styles, P., Radda, G.K.: Brain biochemistry in Williams syndrome (evidence of role of the cerebellum in cognition?). Neurology 51, 33–40 (1998)
Böhmer, V., Vogt, W., Chafaa, S., Meullemeestre, J., Schwinig, M.J., Vierling, F.: ( o-Hydroxyphenyl)methylphosphonic acids: synthesis and potentiometric determinations of their pKa values. Helv. Chim. Acta 76, 139–149 (1993)
Merck, E.: Méthodes d'Analyses Complexométriques par les Titriplex, 3rd edn., p. 57. Darmstadt, Allemagne (1964)
Bentouhami, E., Bouet, G.M., Khan, M.A.: Acidity and complexation of ( o-hydroxyphenyl) monomethylenephosphonic acids towards lanthanide(III) ions, Phosphorus. Sulfur. Silicon 178, 903–921 (2003)
Bentouhami, E., Bouet, G.M., Khan, M.A.: A comparison of the acidity and the complexing ability of ( o-hydroxyphenyl) bis- and ( o-hydroxyphenyl) mono-methylenephosphonic acids towards lanthanide(III) ions. Talanta 57, 545–554 (2002)
Bentouhami, E., Bouet, G.M., Meullemeestre, J., Vierling, F., Khan, M.A.: Physicochemical study of the hydrolysis of rare-earth elements(III) and thorium (IV). C. R Chimie. 7, 537–545 (2004)
Vetrogon, V.I., Lukyanenko, N.G., Scwing-Weill, M.J., Arnaud-Neu, F.: A PC compatible computer program for the calculation of equilibrium constants by the simultaneous processing of different sets of experimental results. Talanta 41, 2105–2112 (1994)
Sillen, L.G., Warnqvist, B.: High speed computers as a supplement to graphical methods. 10. Application of LETAGROP to spectrophotometric data, for testing models and adjusting equilibrium constants. Ark. Kemi. 31, 377–390 (1968)
Ingri, N., Kakolowicz, W., Sillen, L.G., Warnqvist, B.: High speed computers as a supplement to graphical methods-V: haltafall, a general program for calculating the composition of equilibrium mixtures. Talanta 14, 1261–1286 (1967)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bentouhami, E., Bouet, G.M., Schwing, MJ. et al. Potentiometric and UV-Visible Spectrophotometric Studies of the Stability of Thorium(IV) Complexes with (o-Hydroxyphenyl) Mono- and Di-Methylenephosphonic Acids. J Solution Chem 35, 889–916 (2006). https://doi.org/10.1007/s10953-006-9033-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-006-9033-1