Abstract
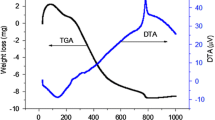
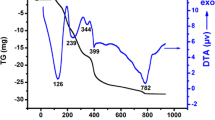
In this paper, an attempt was made to synthesize LaNiO3, CeNiO3, and BaNiO3, and Ce3+ and Ba2+ co-substituted LaNiO3. These samples were further subjected to various material characterization techniques in order to evaluate their physio-chemical properties. Scanning electron microscope (SEM) images showed large chunks of aggregated nanoparticles with minute voids. The EDX-derived atomic composition deviated from the nominal composition suggesting the occurrence of multiple phases. In addition, transmission electron microscope (TEM) images revealed that the samples exhibit uneven spherical shape with a high degree of aggregation. The Fourier transformed-infrared (FT-IR) spectra of the synthesized samples show vibrations of the BO6 octahedral indicating the presence of Ni–O bonds. In addition, metal-carboxyl vibrations were identified from the peaks at 1400 and 860 cm−1. Optical diffuse reflectance spectra (DRS) showed certain peaks originating from the O2− (2p)\(\to\) Ni2+ (3d) charge transfer. The X-ray powder diffraction (XRPD) analysis revealed the existence of multiple phases for the samples CeNiO3, BaNiO3, and La(Ce, Ba)NiO3. Moreover, La(Ce, Ba)NiO3 contained four phases showing that the co-substitution of Ba2+ and Ce3+ into LaNiO3 may require more sophisticated methodologies. The sample BaNiO3 showed maximum weight loss, due to the existence of carbonate phase. The dielectric properties decreased with increasing frequency, while the ac electrical conductivity enhanced with increasing frequencies obeying the Maxwell–Wagner two-layer model in accordance with Koop’s phenomenological theory.









Similar content being viewed by others
References
Chen, J., He, Z., Li, G., An, T., Shi, H., Li, Y.: Visible-light-enhanced photothermocatalytic activity of ABO3-type perovskites for the decontamination of gaseous styrene. Appl. Catal. B Environ. 209, 146–154 (2017). https://doi.org/10.1016/j.apcatb.2017.02.066
Exner, J., Nazarenus, T., Kita, J., Moos, R.: Dense Y-doped ion conducting perovskite films of BaZrO3, BaSnO3, and BaCeO3 for SOFC applications produced by powder aerosol deposition at room temperature. Int. J. Hydrogen Energy. 45, 10000–10016 (2020). https://doi.org/10.1016/j.ijhydene.2020.01.164
Ji, Q., Bi, L., Zhang, J., Cao, H., Zhao, X.S.: The role of oxygen vacancies of ABO3 perovskite oxides in the oxygen reduction reaction. Energy Environ. Sci. 13, 1408–1428 (2020). https://doi.org/10.1039/d0ee00092b
Bulemo, P.M., Kim, I.-D.: Recent advances in ABO3 perovskites: their gas-sensing performance as resistive-type gas sensors. J. Korean Ceram. Soc. 57, 24–39 (2020). https://doi.org/10.1007/s43207-019-00003-1
Sun, C., Alonso, J.A., Bian, J.: Recent advances in perovskite-type oxides for energy conversion and storage applications. Adv. Energy Mater. 11, (2021). https://doi.org/10.1002/aenm.202000459
Malkhandi, S., Trinh, P., Manohar, A.K., Manivannan, A., Balasubramanian, M., Prakash, G.K.S., Narayanan, S.R.: Design insights for tuning the electrocatalytic activity of perovskite oxides for the oxygen evolution reaction. J. Phys. Chem. C. 119, 8004–8013 (2015). https://doi.org/10.1021/jp512722x
Machado, P., Scigaj, M., Gazquez, J., Rueda, E., Sánchez-Díaz, A., Fina, I., Gibert-Roca, M., Puig, T., Obradors, X., Campoy-Quiles, M., Coll, M.: Band gap tuning of solution-processed ferroelectric perovskite BiFe1- xCoxO3 thin films. Chem. Mater. 31, 947–954 (2019). https://doi.org/10.1021/acs.chemmater.8b04380
Yi, Y., Liu, H., Chu, B., Qin, Z., Dong, L., He, H., Tang, C., Fan, M., Bin, L.: Catalytic removal NO by CO over LaNi0.5M0.5O3 (M = Co, Mn, Cu) perovskite oxide catalysts: tune surface chemical composition to improve N2 selectivity. Chem. Eng. J. 369, 511–521 (2019). https://doi.org/10.1016/j.cej.2019.03.066
Tsounis, C., Wang, Y., Arandiyan, H., Wong, R.J., Toe, C.Y., Amal, R., Scott, J.: Tuning the selectivity of LaNiO3 perovskites for CO2 hydrogenation through potassium substitution. Catalysts. 10, (2020). https://doi.org/10.3390/catal10040409
Arandiyan, H., S. Mofarah, S., Sorrell, C.C., Doustkhah, E., Sajjadi, B., Hao, D., Wang, Y., Sun, H., Ni, B.-J., Rezaei, M., Shao, Z., Maschmeyer, T.: Defect engineering of oxide perovskites for catalysis and energy storage: synthesis of chemistry and materials science. Chem. Soc. Rev. 50, 10116–10211 (2021). https://doi.org/10.1039/d0cs00639d
Zhu, H., Zhang, P., Dai, S.: Recent advances of lanthanum-based perovskite oxides for catalysis. ACS Catal. 5, 6370–6385 (2015). https://doi.org/10.1021/acscatal.5b01667
Mudu, F., Olsbye, U., Arstad, B., Diplas, S., Li, Y., Fjellvåg, H.: Aluminium substituted lanthanum based perovskite type oxides, non-stoichiometry and performance in methane partial oxidation by framework oxygen. Appl. Catal. A Gen. 523, 171–181 (2016). https://doi.org/10.1016/j.apcata.2016.05.013
Hwang, J., Rao, R.R., Giordano, L., Katayama, Y., Yu, Y., Shao-Horn, Y.: Perovskites in catalysis and electrocatalysis. Science (80). 358, 751–756 (2017). https://doi.org/10.1126/science.aam7092
Rizwan, M., Gul, S., Iqbal, T., Mushtaq, U., Farooq, M.H., Farman, M., Bibi, R., Ijaz, M.: A review on perovskite lanthanum aluminate (LaAlO3), its properties and applications. Mater. Res. Express. 6, (2019). https://doi.org/10.1088/2053-1591/ab4629
Dias, J.A., Andrade M.A.S., J., Santos, H.L.S., Morelli, M.R., Mascaro, L.H.: Lanthanum-based perovskites for catalytic oxygen evolution reaction. Chem. Electro. Chem 7, 3173–3192 (2020). https://doi.org/10.1002/celc.202000451
Jiang, Q., Cao, Y., Liu, X., Zhang, H., Hong, H., Jin, H.: Chemical looping combustion over a lanthanum nickel perovskite-type oxygen carrier with facilitated O2-transport. Energy Fuels 34, 8732–8739 (2020). https://doi.org/10.1021/acs.energyfuels.0c01038
Sharma, V., Mahapatra, M.K., Krishnan, S., Thatcher, Z., Huey, B.D., Singh, P., Ramprasad, R.: Effects of moisture on (La, A)MnO3 (A = Ca, Sr, and Ba) solid oxide fuel cell cathodes: a first-principles and experimental study. J. Mater. Chem. A. 4, 5605–5615 (2016). https://doi.org/10.1039/c6ta00603e
Bian, L., Duan, C., Wang, L., O’Hayre, R., Cheng, J., Chou, K.-C.: Ce-doped La0.7Sr0.3Fe0.9Ni0.1O3-δ as symmetrical electrodes for high performance direct hydrocarbon solid oxide fuel cells. J. Mater. Chem. A. 5, 15253–15259 (2017). https://doi.org/10.1039/c7ta03001k
Kolisetty, A., Fu, Z., Koc, R.: Development of La(CrCoFeNi)O3 system perovskites as interconnect and cathode materials for solid oxide fuel cells. Ceram. Int. 43, 7647–7652 (2017). https://doi.org/10.1016/j.ceramint.2017.03.061
Rehman, S.U., Shaur, A., Song, R.-H., Lim, T.-H., Hong, J.-E., Park, S.-J., Lee, S.-B.: Nano-fabrication of a high-performance LaNiO3 cathode for solid oxide fuel cells using an electrochemical route. J. Power Sources. 429, 97–104 (2019). https://doi.org/10.1016/j.jpowsour.2019.05.007
Petrie, J.R., Cooper, V.R., Freeland, J.W., Meyer, T.L., Zhang, Z., Lutterman, D.A., Lee, H.N.: Enhanced bifunctional oxygen catalysis in strained LaNiO3 perovskites. J. Am. Chem. Soc. 138, 2488–2491 (2016). https://doi.org/10.1021/jacs.5b11713
Zhao, Y., Hang, Y., Zhang, Y., Wang, Z., Yao, Y., He, X., Zhang, C., Zhang, D.: Strontium-doped perovskite oxide La1-xSrxMnO3 (x = 0, 0.2, 0.6) as a highly efficient electrocatalyst for nonaqueous Li-O2 batteries. Electrochim. Acta. 232, 296–302 (2017). https://doi.org/10.1016/j.electacta.2017.02.155
Dai, Y., Yu, J., Cheng, C., Tan, P., Ni, M.: Mini-review of perovskite oxides as oxygen electrocatalysts for rechargeable zinc–air batteries. Chem. Eng. J. 397, (2020). https://doi.org/10.1016/j.cej.2020.125516
Hu, Q., Yue, B., Shao, H., Yang, F., Wang, J., Wang, Y., Liu, J.: Facile syntheses of perovskite type LaMO3 (M=Fe, Co, Ni) nanofibers for high performance supercapacitor electrodes and lithium-ion battery anodes. J. Alloys Compd. 852, (2021). https://doi.org/10.1016/j.jallcom.2020.157002
Yan, K.-L., Fan, R.-H., Chen, M., Sun, K., Yin, L.-W., Li, H., Pan, S.-B., Yu, M.-X.: Perovskite (La, Sr)MnO3 with tunable electrical properties by the Sr-doping effect. J. Alloys Compd. 628, 429–432 (2015). https://doi.org/10.1016/j.jallcom.2014.12.137
Peng, T., Liu, X., Dai, K., Xiao, J., Song, H.: Effect of acidity on the glycine-nitrate combustion synthesis of nanocrystalline alumina powder. Mater. Res. Bull. 41, 1638–1645 (2006). https://doi.org/10.1016/j.materresbull.2006.02.026
Zhu, Y., Zhou, W., Yu, J., Chen, Y., Liu, M., Shao, Z.: Enhancing electrocatalytic activity of perovskite oxides by tuning cation deficiency for oxygen reduction and evolution reactions. Chem. Mater. 28, 1691–1697 (2016). https://doi.org/10.1021/acs.chemmater.5b04457
Wang, L., Stoerzinger, K.A., Chang, L., Zhao, J., Li, Y., Tang, C.S., Yin, X., Bowden, M.E., Yang, Z., Guo, H., You, L., Guo, R., Wang, J., Ibrahim, K., Chen, J., Rusydi, A., Wang, J., Chambers, S.A., Du, Y.: Tuning bifunctional oxygen electrocatalysts by changing the A-site rare-earth element in perovskite nickelates. Adv. Funct. Mater. 28, (2018). https://doi.org/10.1002/adfm.201803712
Hossain, A., Roy, S., Sakthipandi, K.: The external and internal influences on the tuning of the properties of perovskites: an overview. Ceram. Int. 45, 4152–4166 (2019). https://doi.org/10.1016/j.ceramint.2018.11.102
Herklotz, A., Wong, A.T., Meyer, T., Biegalski, M.D., Lee, H.N., Ward, T.Z.: Controlling octahedral rotations in a perovskite via strain doping. Sci. Rep. 6, (2016). https://doi.org/10.1038/srep26491
Coşkun, M., Polat, Ö., Coşkun, F.M., Durmuş, Z., Çağlar, M., Türüt, A.: Frequency and temperature dependent electrical and dielectric properties of LaCrO3 and Ir doped LaCrO3 perovskite compounds. J. Alloys Compd. 740, 1012–1023 (2018). https://doi.org/10.1016/j.jallcom.2018.01.022
Ma, P.P., Zhu, B., Lei, N., Liu, Y.K., Yu, B., Lu, Q.L., Dai, J.M., Li, S.H., Jiang, G.H.: Effect of Sr substitution on structure and electrochemical properties of perovskite-type LaMn0.9Ni0.1O3 nanofibers. Mater. Lett. 252, 23–26 (2019). https://doi.org/10.1016/j.matlet.2019.05.090
Zhang, X., Pei, C., Chang, X., Chen, S., Liu, R., Zhao, Z.-J., Mu, R., Gong, J.: FeO6 octahedral distortion activates lattice oxygen in perovskite ferrite for methane partial oxidation coupled with CO2 splitting. J. Am. Chem. Soc. 142, 11540–11549 (2020). https://doi.org/10.1021/jacs.0c04643
Megarajan, S.K., Rayalu, S., Nishibori, M., Teraoka, Y., Labhsetwar, N.: Effects of surface and bulk silver on PrMnO3+δ perovskite for CO and soot oxidation: experimental evidence for the chemical state of silver. ACS Catal. 5, 301–309 (2015). https://doi.org/10.1021/cs500880w
Zhang, Z., Chen, D., Dong, F., Xu, X., Hao, Y., Shao, Z.: Understanding the doping effect toward the design of CO2-tolerant perovskite membranes with enhanced oxygen permeability. J. Memb. Sci. 519, 11–21 (2016). https://doi.org/10.1016/j.memsci.2016.07.043
Chen, G., Zhou, W., Guan, D., Sunarso, J., Zhu, Y., Hu, X., Zhang, W., Shao, Z.: Two orders of magnitude enhancement in oxygen evolution reactivity on amorphous Ba0.5Sr0.5Co0.8Fe0.2O3−d nanofilms with tunable oxidation state. Sci. Adv. 3, (2017). https://doi.org/10.1126/sciadv.1603206
Xiong, J., Zhong, H., Li, J., Zhang, X., Shi, J., Cai, W., Qu, K., Zhu, C., Yang, Z., Beckman, S.P., Cheng, H.: Engineering highly active oxygen sites in perovskite oxides for stable and efficient oxygen evolution. Appl. Catal. B Environ. 256, (2019). https://doi.org/10.1016/j.apcatb.2019.117817
Yang, J., Hu, S., Fang, Y., Hoang, S., Li, L., Yang, W., Liang, Z., Wu, J., Hu, J., Xiao, W., Pan, C., Luo, Z., Ding, J., Zhang, L., Guo, Y.: Oxygen vacancy promoted o2 activation over perovskite oxide for low-temperature co oxidation. ACS Catal. 9, 9751–9763 (2019). https://doi.org/10.1021/acscatal.9b02408
Cui, Z., Lyu, N., Ding, Y., Bai, K.: Noncovalently functionalization of Janus MoSSe monolayer with organic molecules. Phys. E Low-Dimensional Syst. Nanostructures. 127, (2021). https://doi.org/10.1016/j.physe.2020.114503
Cui, Z., Luo, Y., Yu, J., Xu, Y.: Tuning the electronic properties of MoSi2N4 by molecular doping: a first principles investigation. Phys. E Low-Dimensional Syst. Nanostructures. 134, (2021). https://doi.org/10.1016/j.physe.2021.114873
Cui, Z., Bai, K., Wang, X., Li, E., Zheng, J.: Electronic, magnetism, and optical properties of transition metals adsorbed g-GaN. Phys. E Low-Dimensional Syst. Nanostructures. 118, (2020). https://doi.org/10.1016/j.physe.2019.113871
Cui, Z., Wang, M., Lyu, N., Zhang, S., Ding, Y., Bai, K.: Electronic, magnetism and optical properties of transition metals adsorbed puckered arsenene. Superlattices Microstruct. 152, (2021). https://doi.org/10.1016/j.spmi.2021.106852
Sankannavar, R., Sandeep, K.C., Kamath, S., Suresh, A.K., Sarkar, A.: Impact of strontium-substitution on oxygen evolution reaction of lanthanum nickelates in alkaline solution. J. Electrochem. Soc. 165, J3236–J3245 (2018). https://doi.org/10.1149/2.0301815jes
Sankannavar, R., Sarkar, A.: The electrocatalysis of oxygen evolution reaction on La1−xCaxFeO3−δ perovskites in alkaline solution. Int. J. Hydrogen Energy. 43, 4682–4690 (2018). https://doi.org/10.1016/j.ijhydene.2017.08.092
Kim, H.-Y., Shin, J., Jang, I.-C., Ju, Y.-W.: Hydrothermal synthesis of three-dimensional perovskite NiMnO3 oxide and application in supercapacitor electrode. Energies. 13, (2019). https://doi.org/10.3390/en13010036
Nguyen, A.T., Pham, V.N.T., Nguyen, T.T.L., Mittova, V.O., Vo, Q.M., Berezhnaya, M. V, Mittova, I.Y., Do, T.H., Chau, H.D.: Crystal structure and magnetic properties of perovskite YFe1-xMnxO3 nanopowders synthesized BY CO-PRECIPITATION method. Solid State Sci. 96, (2019). https://doi.org/10.1016/j.solidstatesciences.2019.06.011
Durai, L., Badhulika, S.: A facile, solid-state reaction assisted synthesis of a berry-like NaNbO3 perovskite structure for binder-free, highly selective sensing of dopamine in blood samples. New J. Chem. 43, 11994–12003 (2019). https://doi.org/10.1039/c9nj02282a
Bibi, I., Maqbool, H., Iqbal, S., Majid, F., Kamal, S., Alwadai, N., Iqbal, M.: La1-xGdxCr1-yNiyO3 perovskite nanoparticles synthesis by micro-emulsion route: dielectric, magnetic and photocatalytic properties evaluation. Ceram. Int. 47, 5822–5831 (2021). https://doi.org/10.1016/j.ceramint.2020.11.033
Yu, L., Xu, N., Zhu, T., Xu, Z., Sun, M., Geng, D.: La0.4Sr0.6Co0.7Fe0.2Nb0.1O3-δ perovskite prepared by the sol-gel method with superior performance as a bifunctional oxygen electrocatalyst. Int. J. Hydrogen Energy. 45, 30583–30591 (2020). https://doi.org/10.1016/j.ijhydene.2020.08.105
Jouannaux, J., Haeussler, A., Drobek, M., Ayral, A., Abanades, S., Julbe, A.: Lanthanum manganite perovskite ceramic powders for CO2 splitting: influence of Pechini synthesis parameters on sinterability and reactivity. Ceram. Int. 45, 15636–15648 (2019). https://doi.org/10.1016/j.ceramint.2019.05.075
Du, X., Ai, H., Chen, M., Liu, D., Chen, S., Wang, X., Lo, K.H., Pan, H.: PLD-fabricated perovskite oxide nanofilm as efficient electrocatalyst with highly enhanced water oxidation performance. Appl. Catal. B Environ. 272, (2020). https://doi.org/10.1016/j.apcatb.2020.119046
Petrović, S., Rožić, L., Grbić, B., Radić, N., Cherkezova-Zheleva, Z., Stojadinović, S.: Structural, optical and photocatalytic properties of LaTi0.4Mg0.4Fe0.2O3 perovskite prepared by high-energy ball milling. J. Solid State Chem. 297, (2021). https://doi.org/10.1016/j.jssc.2021.122085
Wang, Y., Liu, M., Chen, W., Mao, L., Shangguan, W.: Ag loaded on layered perovskite H2SrTa2O7 to enhance the selectivity of photocatalytic CO2 reduction with H2O. J. Alloys Compd. 786, 149–154 (2019). https://doi.org/10.1016/j.jallcom.2019.01.325
Wang, W., Lin, B., Zhang, H., Sun, Y., Zhang, X., Yang, H.: Synthesis, morphology and electrochemical performances of perovskite-type oxide LaxSr1-xFeO3 nanofibers prepared by electrospinning. J. Phys. Chem. Solids. 124, 144–150 (2019). https://doi.org/10.1016/j.jpcs.2018.09.011
Hkiri, K., Mohamed, H.E.A., Khanyile, B.S., Mtshali, C., Nkosi, M., Ben Salem, M., Maaza, M., Zouaoui, M.: Deposition of CaZrO3 thin films by EB-PVD: effects of substrate on the composition, the structure, the morphology and the optical properties. Surfaces and Interfaces. 25, (2021). https://doi.org/10.1016/j.surfin.2021.101259
Zhou, Y., Guan, X., Zhou, H., Ramadoss, K., Adam, S., Liu, H., Lee, S., Shi, J., Tsuchiya, M., Fong, D.D., Ramanathan, S.: Strongly correlated perovskite fuel cells. Nature 534, 231–234 (2016). https://doi.org/10.1038/nature17653
Guo, H., Huang, J., Zhou, H., Zuo, F., Jiang, Y., Zhang, K.H.L., Fu, X., Bu, Y., Cheng, W., Sun, Y.: Unusual role of point defects in perovskite nickelate electrocatalysts. ACS Appl. Mater. Interfaces. (2021). https://doi.org/10.1021/acsami.1c04903
Cao, C., Shang, C., Li, X., Wang, Y., Liu, C., Wang, X., Zhou, S., Zeng, J.: Dimensionality control of electrocatalytic activity in perovskite nickelates. Nano Lett. 20, 2837–2842 (2020). https://doi.org/10.1021/acs.nanolett.0c00553
Ramadoss, K., Zuo, F., Sun, Y., Zhang, Z., Lin, J., Bhaskar, U., Shin, S., Alam, M.A., Guha, S., Weinstein, D., Ramanathan, S.: Proton-doped strongly correlated perovskite nickelate memory devices. IEEE Electron Device Lett. 39, 1500–1503 (2018). https://doi.org/10.1109/LED.2018.2865776
Wang, L., Dash, S., Chang, L., You, L., Feng, Y., He, X., Jin, K.-J., Zhou, Y., Ong, H.G., Ren, P., Wang, S., Chen, L., Wang, J.: Oxygen vacancy induced room-temperature metal-insulator transition in nickelate films and its potential application in photovoltaics. ACS Appl. Mater. Interfaces. 8, 9769–9776 (2016). https://doi.org/10.1021/acsami.6b00650
Chang, L., Wang, L., You, L., Yang, Z., Abdelsamie, A., Zhang, Q., Zhou, Y., Gu, L., Chambers, S.A., Wang, J.: Tuning photovoltaic performance of perovskite nickelates heterostructures by changing the A-site rare-earth element. ACS Appl. Mater. Interfaces. 11, 16191–16197 (2019). https://doi.org/10.1021/acsami.9b01851
Chang, L., Wang, L., You, L., Zhou, Y., Fang, L., Wang, S., Wang, J.: Band gap tuning of nickelates for photovoltaic applications. J. Phys. D. Appl. Phys. 49, (2016). https://doi.org/10.1088/0022-3727/49/44/44LT02
Peña, M.A., Fierro, J.L.G.: Chemical structures and performance of perovskite oxides. Chem. Rev. 101, 1981–2017 (2001). https://doi.org/10.1021/cr980129f
Rietveld, H.M.: A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 2, 65–71 (1969). https://doi.org/10.1107/S0021889869006558
Bruker AXS.: TOPAS V6: “General profile and structure analysis software for powder diffraction data”-User’s Manual, Bruker AXS, Karlruche, German (2017)
Cheary, R.W., Coelho, A.: A fundamental parameters approach to X-ray line-profile fitting. J. Appl. Crystallogr. 25, 109–121 (1992). https://doi.org/10.1107/S0021889891010804
Cheary, R.W., Coelho, A.A.: Axial divergence in a conventional X-ray powder diffractometer. I. Theoretical foundations. J. Appl. Crystallogr. 31, 851–861 (1998). https://doi.org/10.1107/S0021889898006876
Cheary, R.W., Coelho, A.A.: Axial divergence in a conventional X-ray powder diffractometer. II. Realization and evaluation in a fundamental-parameter profile fitting procedure. J. Appl. Crystallogr. 31, 862–868 (1998). https://doi.org/10.1107/S0021889898006888
Balzar, D.: Voigt-function model in diffraction line-broadening analysis. Int. union Crystallogr. Monogr. Crystallogr. 10, 94–126 (1999)
Zhang, Y., Jin, Z., Chen, L., Wang, J.: SrFexNi1-xO3-δ perovskites coated on Ti anodes and their electrocatalytic properties for cleaning nitrogenous wastewater. Materials (Basel). 12, (2019). https://doi.org/10.3390/ma12030511
Zhu, J., Xiao, D., Li, J., Yang, X., Wu, Y.: Effect of Ce on NO direct decomposition in the absence/presence of O2 over La1-xCexSrNiO4 (0 ≤ x ≤ 0.3). J. Mol. Catal. A Chem.234, 99–105 (2005). https://doi.org/10.1016/j.molcata.2005.02.015
Wu, B., Xiong, Y.: A novel low-temperature NO removal approach with •OH from catalytic decomposition of H2O2 over La1- xCaxFeO3 oxides. J. Chem. Technol. Biotechnol. 93, 43–53 (2018). https://doi.org/10.1002/jctb.5317
Kubelka, P., Munk, F.: Ein Beitrag zur Optik der Farbanstriche. Z. Tech. Phys. 12, 593–601 (1931)
López, R., Gómez, R.: Band-gap energy estimation from diffuse reflectance measurements on sol-gel and commercial TiO2: a comparative study. J. Sol-Gel Sci. Technol. 61, 1–7 (2012). https://doi.org/10.1007/s10971-011-2582-9
Tauc, J., Grigorovici, R., Vancu, A.: Optical properties and electronic structure of amorphous germanium. Phys. status solidi. 15, 627–637 (1966). https://doi.org/10.1002/pssb.19660150224
Davis, E.A., Mott, N.F.: Conduction in non-crystalline systems V. Conductivity, optical absorption and photoconductivity in amorphous semiconductors. Philos. Mag. 22, 903–922 (1970). https://doi.org/10.1080/14786437008221061
Mott, N.F., Davis, E.A.: Electronic processes in non-crystalline materials. Oxford university press (2012)
Pankove, J.I.: Optical processes in semiconductors. Courier Corporation (1975)
Makuła, P., Pacia, M., Macyk, W.: How to correctly determine the band gap energy of modified semiconductor photocatalysts based on UV-Vis spectra. J. Phys. Chem. Lett. 9, 6814–6817 (2018). https://doi.org/10.1021/acs.jpclett.8b02892
Saleem, M., Singh, D., Mishra, A., Varshney, D.: Structural, transport and collosal dielectric properties of A-site substituted La2NiO4. Mater. Res. Express. 6, (2019). https://doi.org/10.1088/2053-1591/aaecf7
Toby, B.H.: R factors in Rietveld analysis: how good is good enough? Powder Diffr. 21, 67–70 (2006). https://doi.org/10.1154/1.2179804
Mccusker, L.B., Von Dreele, R.B., Cox, D.E., Louër, D., Scardi, P.: Rietveld refinement guidelines. J. Appl. Crystallogr. 32, 36–50 (1999). https://doi.org/10.1107/S0021889898009856
Li, L., Jiang, B., Tang, D., Zhang, Q., Zheng, Z.: Hydrogen generation by acetic acid steam reforming over Ni-based catalysts derived from La1−xCexNiO3 perovskite. Int. J. Hydrogen Energy. 43, 6795–6803 (2018). https://doi.org/10.1016/j.ijhydene.2018.02.128
Maxwell, J.C.: A treatise on electricity and magnetism. Clarendon press (1873)
Wagner, K.W.: Zur Theorie der unvollkommenen Dielektrika. Ann. Phys. 345, 817–855 (1913). https://doi.org/10.1002/andp.19133450502
Jonscher, A.K.: The “universal” dielectric response. Nature 267, 673–679 (1977). https://doi.org/10.1038/267673a0
Acknowledgements
The authors acknowledge the Centre for Advanced Materials Technology of RIT Bangalore for the necessary characterization facilities used in this study. A portion of this research was also performed using facilities at CeNSE, funded by Department of Information Technology, Govt. of India, located at Indian Institute of Science, Bangalore.
Author information
Authors and Affiliations
Contributions
Shreyas J. Kashyap: conceptualization, validation, formal analysis, investigation, data curation, writing-original draft, writing-reviewing and editing, visualization; Ravi Sankannavar: conceptualization, methodology, validation, resources, visualization; G. M. Madhu: resources, formal analysis.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kashyap, S.J., Sankannavar, R. & Madhu, G.M. Synthesis and Characterization of La(Ce, Ba)NiO3 Perovskite-Type Oxides. J Supercond Nov Magn 35, 2107–2118 (2022). https://doi.org/10.1007/s10948-022-06219-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-022-06219-3