Abstract
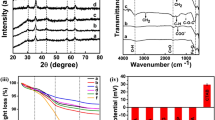
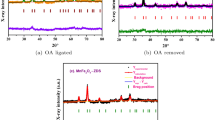
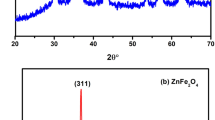
Here Brown relaxation time was altered to enhance magnetic hyperthermia value. Narrow size distributed, hydrophobic Fe3O4 nanoparticles were synthesized by solvothermal reflux method. It is reported that the existing both single step and two-step ligand exchange protocols could not replace hydrophobic surfactant ligand completely. Hence, as-synthesized samples were transferred into hydrophilic by completely removing the hydrophobic surfactant on the nanoparticles by high polar ionic liquids and ligated short-chain bidentate zwitterionic dopamine sulfonate on nanoparticles surface. The short molecules will fast up the Brown relaxation time compared to long molecule surfactant such as polyethylene glycol (PEG). The resultant sample shows magnetic hyperthermia value of 558 Js− 1g− 1 of Fe content at 316 kHz and 35.33 kA/m, at 2 mg/mL. The obtained hyperthermia is very high compared to long-chain PEG ligated Fe3O4 nanoparticles. It is attributed to relaxation time difference of long-chain PEG ligated Fe3O4 and short molecule zwitterion surfactant stabilized Fe3O4. Present hyperthermia results are the first report on zwitterionic dopamine sulfonate anchored Fe3O4 superparamagnetic nanoparticles.







Similar content being viewed by others
Data Availability
Not applicable.
References
Akbarzadeh, A., Samiei, M., Davaran, S.: Magnetic nanoparticles: preparation, physical properties, and applications in biomedicine. Nanoscale Res. Lett. 7(1), 144 (2012). https://doi.org/10.1186/1556-276x-7-144
Balivada, S., Rachakatla, R.S., Wang, H., Samarakoon, T.N., Dani, R.K., Pyle, M., Kroh, F.O., Walker, B., Leaym, X., Koper, O.B., Tamura, M., Chikan, V., Bossmann, S.H., Troyer, D.L.: A/C magnetic hyperthermia of melanoma mediated by iron(0)/iron oxide core/shell magnetic nanoparticles: a mouse study. BMC Cancer 10(1), 119 (2010). https://doi.org/10.1186/1471-2407-10-119
Chen, R., Christiansen, M.G., Sourakov, A., Mohr, A., Matsumoto, Y., Okada, S., Jasanoff, A., Anikeeva, P.: High-performance ferrite nanoparticles through nonaqueous redox phase tuning. Nano. Lett. 16(2), 1345–1351 (2016). https://doi.org/10.1021/acs.nanolett.5b04761
Chung, S.H., Hoffmann, A., Bader, S.D., Liu, C., Kay, B., Makowski, L., Chen, L.: Biological sensors based on Brownian relaxation of magnetic nanoparticles. Appl. Phys. Lett. 85(14), 2971–2973 (2004). https://doi.org/10.1063/1.1801687
Cornell, R.M., Schwertmann, U.: The Iron Oxides: Structure, Properties, Reactions, Occurrences and Uses. Wiley-Blackwell, Hoboken (2003). https://doi.org/10.1002/3527602097
Dehsari, H.S., Harris, R.A., Ribeiro, A.H., Tremel, W., Asadi, K.: Optimizing the binding energy of the surfactant to iron oxide yields truly monodisperse nanoparticles. Langmuir 34(22), 6582–6590 (2018). https://doi.org/10.1021/acs.langmuir.8b01337
Deissler, R.J., Martens, M.A., Wu, Y., Brown, R.: Brownian and Néel relaxation times in magnetic particle dynamics. In: 2013 International Workshop on Magnetic Particle Imaging. IEEE. https://doi.org/10.1109/iwmpi.2013.6528375 (2013)
Dieckhoff, J., Eberbeck, D., Schilling, M., Ludwig, F.: Magnetic-field dependence of Brownian and Néel relaxation times. J. Appl. Phys. 119(4), 043,903 (2016). https://doi.org/10.1063/1.4940724
Dill, K.A., Ghosh, K., Schmit, J.D.: Physical limits of cells and proteomes. Proc. Natl. Acad. Sci. 108(44), 17,876–17,882 (2011). https://doi.org/10.1073/pnas.1114477108
Dutta, B., Shetake, N.G., Gawali, S.L., Barick, B., Barick, K., Babu, P., Pandey, B., Priyadarsini, K., Hassan, P.: PEG mediated shape-selective synthesis of cubic Fe3O4 nanoparticles for cancer therapeutics. J. Alloys Compd. 737, 347–355 (2018). https://doi.org/10.1016/j.jallcom.2017.12.028
Ehsani, M.H., Esmaeili, S., Aghazadeh, M., Kameli, P., Karimzadeh, I.: Magnetic evaluation of the nanoparticles coated with polyvinylpyrrolidone and polyvinyl chloride nanoparticles synthesized by electro-deposition method for hyperthermia application. J. Supercond. Nov. Magn. 32(7), 2021–2030 (2018). https://doi.org/10.1007/s10948-018-4908-0
Estephan, Z.G., Jaber, J.A., Schlenoff, J.B.: Zwitterion-stabilized silica nanoparticles: toward nonstick nano. Langmuir 26(22), 16,884–16,889 (2010). https://doi.org/10.1021/la103095d
Farzin, A., Etesami, S.A., Quint, J., Memic, A., Tamayol, A.: Magnetic nanoparticles in cancer therapy and diagnosis. Adv. Healthc. Mater. 9(9), 1901,058 (2020). https://doi.org/10.1002/adhm.201901058
Franco, V., Blázquez, J., Ipus, J., Law, J., Moreno-Ramírez, L., Conde, A.: Magnetocaloric effect: from materials research to refrigeration devices. Prog. Mater. Sci. 93, 112–232 (2018). https://doi.org/10.1016/j.pmatsci.2017.10.005
García, K.P., Zarschler, K., Barbaro, L., Barreto, J.A., O’Malley, W., Spiccia, L., Stephan, H., Graham, B.: Zwitterionic-coated “stealth” nanoparticles for biomedical applications: recent advances in countering biomolecular corona formation and uptake by the mononuclear phagocyte system. Small 10(13), 2516–2529 (2014). https://doi.org/10.1002/smll.201303540
Gawali, S.L., Barick, B., Barick, K., Hassan, P.: Effect of sugar alcohol on colloidal stabilization of magnetic nanoparticles for hyperthermia and drug delivery applications. J. Alloys Compd. 725, 800–806 (2017). https://doi.org/10.1016/j.jallcom.2017.07.206
Gilchrist, R., Medal, R., Shorey, W.D., Hanselman, R.C., Parrott, J.C., Taylor, C.B.: Selective inductive heating of lymph nodes. Ann. Surg. 146(4), 596–599 (1957)
Gillooly, J.F.: Effects of size and temperature on metabolic rate. Science 293(5538), 2248–2251 (2001). https://doi.org/10.1126/science.1061967
Ishii, M., Nakahira, M., Yamanaka, T.: Infrared absorption spectra and cation distributions in (Mn,Fe)3O4. Solid State Commun. 11(1), 209–212 (1972). https://doig.org/10.1016/0038-1098(72)91162-3
Jordan, A., Wust, P., Fähling, H., John, W., Hinz, A., Felix, R.: Inductive heating of ferrimagnetic particles and magnetic fluids: physical evaluation of their potential for hyperthermia. Int. J. Hyperthermia 25(7), 499–511 (2009). https://doi.org/10.3109/02656730903287790
Keiser, J.T.: The electrochemical reduction of rust films on weathering steel surfaces. J. Electrochem. Soc. 129(12), 2686–2689 (1982). https://doi.org/10.1149/1.2123648
Kolen’ko, Y.V., Bañobre-López, M., Rodríguez-Abreu, C., Carbó-Argibay, E., Sailsman, A., Piñeiro-Redondo, Y., Cerqueira, M.F., Petrovykh, D.Y., Kovnir, K., Lebedev, O.I., Rivas, J.: Large-scale synthesis of colloidal Fe3O4 nanoparticles exhibiting high heating efficiency in magnetic hyperthermia. J. Phys. Chem. C 118(16), 8691–8701 (2014). https://doi.org/10.1021/jp500816u
Kronmüller, H., Parkin, S. (eds.): Novel Materials: Ferro-, Ferri-, and Antiferrimagnetic Nanoparticles. Wiley, New York (2007). https://doi.org/10.1002/9780470022184
Lee, J.S., Cha, J.M., Yoon, H.Y., Lee, J.K., Kim, Y.K.: Magnetic multi-granule nanoclusters: a model system that exhibits universal size effect of magnetic coercivity. Sci. Rep. 5(1), 12,135 (2015). https://doi.org/10.1038/srep12135
Li, Q., Kartikowati, C.W., Horie, S., Ogi, T., Iwaki, T., Okuyama, K.: Correlation between particle size/domain structure and magnetic properties of highly crystalline Fe3O4 nanoparticles. Sci. Rep. 7(1), 9894 (2017). https://doi.org/10.1038/s41598-017-09897-5
Lide, D.R.: CRC handbook of chemistry and physics, 86th edition 2005-2006, 86 edn. CRC. https://www.crcpress.com/CRC-Handbook-of-Chemistry-and-Physics-86th-Edition/Lide/p/book/9780849304859 (2005)
Manohar, A., Krishnamoorthi, C.: Site selective Cu2+ substitution in single crystal Fe3O4 biocompatible nanospheres by solvothermal reflux method. J. Cryst. Growth 473, 66–74 (2017). https://doi.org/10.1016/j.jcrysgro.2017.05.013
de Mello, L.B., Varanda, L.C., Sigoli, F.A., Mazali, I.O.: Co-precipitation synthesis of (Zn-Mn)-co-doped magnetite nanoparticles and their application in magnetic hyperthermia. J. Alloys Compd. 779, 698–705 (2019). https://doi.org/10.1016/j.jallcom.2018.11.280
Ritchie, M.E.: Reaction and diffusion thermodynamics explain optimal temperatures of biochemical reactions. Sci. Rep. 8(1), 11,105 (2018). https://doi.org/10.1038/s41598-018-28833-9
Rosensweig, R.: Heating magnetic fluid with alternating magnetic field. J. Magn. Magn. Mater. 252, 370–374 (2002). https://doi.org/10.1016/s0304-8853(02)00706-0
Sarma, L., Borah, J.P., Srinivasan, A., Sarma, S.: Synthesis and characterization of tea polyphenol–coated magnetite nanoparticles for hyperthermia application. J. Supercond. Nov. Magn. 33(6), 1637–1644 (2019). https://doi.org/10.1007/s10948-019-05189-3
Shaghasemi, B.S., Dehghani, E.S., Benetti, E.M., Reimhult, E.: Host–guest driven ligand replacement on monodisperse inorganic nanoparticles. Nanoscale 9(26), 8925–8929 (2017). https://doi.org/10.1039/c7nr02199b
Shao, Q., Jiang, S.: Molecular understanding and design of zwitterionic materials. Adv. Mater. 27(1), 15–26 (2014). https://doi.org/10.1002/adma.201404059
Sharma, K.S., Ningthoujam, R.S., Dubey, A.K., Chattopadhyay, A., Phapale, S., Juluri, R.R., Mukherjee, S., Tewari, R., Shetake, N.G., Pandey, B.N., Vatsa, R.K.: Synthesis and characterization of monodispersed water dispersible Fe3O4 nanoparticles and in vitro studies on human breast carcinoma cell line under hyperthermia condition. Sci. Rep. 8(1), 14,766 (2018). https://doi.org/10.1038/s41598-018-32934-w
Smolensky, E.D., Park, H.Y.E., Berquó, T. S., Pierre, V.C.: Surface functionalization of magnetic iron oxide nanoparticles for MRI applications - effect of anchoring group and ligand exchange protocol. Contrast Media Mol. Imaging 6, 189–199 (2010). https://doi.org/10.1002/cmmi.417
Soares, P.I.P., Lochte, F., Echeverria, C., Pereira, L.C.J., Coutinho, J.T., Ferreira, I.M.M., Novo, C.M.M., Borges, J.P.M.R.: Thermal and magnetic properties of iron oxide colloids: influence of surfactants. Nanotechnology 26(42), 425,704 (2015). https://doi.org/10.1088/0957-4484/26/42/425704
Stauffer, P.R., Cetas, T.C., Jones, R.C.: Magnetic induction heating of ferromagnetic implants for inducing localized hyperthermia in deep-seated tumors. IEEE Trans. Biomed. Eng. BME-31(2), 235–251 (1984). https://doi.org/10.1109/tbme.1984.325334
Teicher, B.A. (ed.): Cancer Drug Resistance. Humana Press, New Jersey (2006). https://doi.org/10.1007/978-1-59745-035-5
Torres, T.E., Lima, E., Calatayud, M.P., Sanz, B., Ibarra, A., Fernández-Pacheco, R., Mayoral, A., Marquina, C., Ibarra, M.R., Goya, G.F.: The relevance of Brownian relaxation as power absorption mechanism in magnetic hyperthermia. Sci. Rep. 9, 3992 (2019). https://doi.org/10.1038/s41598-019-40341-y
Yuen, A.K.L., Hutton, G.A., Masters, A.F., Maschmeyer, T.: The interplay of catechol ligands with nanoparticulate iron oxides. Dalton Trans. 41(9), 2545 (2012). https://doi.org/10.1039/c2dt11864e
Ziabari, S.A.M., Babamoradi, M., Hajizadeh, Z., Maleki, A.: The effect of magnetic field on the magnetic and hyperthermia properties of Bentonite/Fe3O4 nanocomposite. Physica. B 588, 412,167 (2020). https://doi.org/10.1016/j.physb.2020.412167
Acknowledgments
We thank Dr. D. Prabhu, ARCI Chennai, for M-H loop and FESEM micrograph data measurement and Mr. T. Viswanathan, Department of Chemistry, VIT Vellore, for assisting in zwitterionic dopamine sulfonate synthesis.
Funding
The authors thank Vellore Institute of Technology (VIT) for providing “VIT SEED GRANT” for carrying out this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Code Availability
Not applicable.
Rights and permissions
About this article
Cite this article
Vijayakanth, V., Krishnamoorthi, C. Effect of Zwitterionic Surfactant Ligand Monolayer on Magnetic Hyperthermia Properties of Monosize Fe3O4 Nanoparticles. J Supercond Nov Magn 34, 623–632 (2021). https://doi.org/10.1007/s10948-020-05716-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-020-05716-7