Abstract
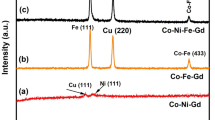
CoFeCu thin films were electrodeposited from baths with natural pH (instead of pH∼2.8 used in conventional baths) and containing different sodium citrate dosages. ChemEQL V.3.0 software was employed to study speciation diagrams of citrate-added CoFeCu bath with natural pH. At low sodium citrate dosage, Co++, Fe++, and Cu++ species were dominant in CoFeCu bath with natural pH (around 5.2). However, as dosage of sodium citrate in the bath increased, the concentration of complexed species (especially Co(C6H5O7)−, Fe(C6H5O7)−, and Cu(OHC6H5O7)2−) significantly raised. Cyclic voltammetry (CV) studies showed that the formation of complexed species in the bath shifted reduction potential of metals towards more negative potentials. Moreover, in order to deposit cobalt and iron simultaneously with copper, it was necessary to increase the reverse potential (E λ ) value gradually with sodium citrate dosage, otherwise, only copper would have deposited from citrate-added CoFeCu bath. Scanning electron micrographs illustrated that using natural pH (about 5.2) remarkably decreased the number of microvoids in the deposited films compared with the film deposited from conventional baths with pH level of 2.8. EDS, XRD, and VSM were also used for characterization of the deposited films. All deposited films exhibited nanocrystalline structures, and increasing sodium citrate into the baths led to reduction in grain sizes (D) and coercivity (H c) of the CoFeCu thin films. Plotting log(H c) versus log(D 6) demonstrated that films coercivity followed the “D 6 law”. There were only two phase structures (FCC (Co) or BCC (Fe)) observed in the X-ray diffraction patterns of the films. In addition, films with double-phase structures (FCC+BCC) showed finer grain sizes and therefore exhibited lower coercivity in comparison with single-phase (FCC or BCC) films. CoFeCu thin films deposited at higher dosages of sodium citrate (>20 g/L) were poor in diamagnetic copper and consequently showed higher saturation magnetizations.








Similar content being viewed by others
References
Chen, Y., Wang, Q.P., Cai, C., Yuan, Y.N., Cao, F.H., Zhang, Z., Zhang, J.Q.: Electrodeposition and characterization of nanocrystalline CoNiFe films. Thin Solid Films 520, 3553–3557 (2012)
Osaka, T.: Electrodeposition of highly functional thin films for magnetic recording devices of the next century. Electrochim. Acta 45, 3311–3321 (2000)
Vlad, L., Sandu, A.V., Georgescu, V.: The effects of the thermal treatment on the structural and magnetic properties of Zn–Co alloys prepared by electrochemical deposition. J. Supercond. Nov. Magn. 25, 469–474 (2012)
Herzer, G.: Grain size dependence of coercivity and permeability in nanocrystalline ferromagnets. IEEE Trans. Magn. 26, 1397–1402 (1990)
Esmaili, S., Bahrololoom, M.E., Péter, L.: Magnetoresistance of electrodeposited NiFeCu alloys. Thin Solid Films 520, 2190–2194 (2012)
Kalu, E.E., Bell, R., Dupree, M.: Improvement of the corrosion behavior of electrodeposited CoFeCu thin films. Mater. Chem. Phys. 124, 689–693 (2010)
Lee, J.O., Kim, H.K., Kim, G.H., Jeung, W.Y.: Tailoring the magnetic properties of CoFeNi alloys with variations in copper contents. J. Appl. Phys. 99, 08B704 (2006)
Cooper, E.I., Bonhote, C., Heidmann, J., Hsu, Y., Kern, P., Lam, J.W., Ramasubramanian, M., Robertson, N., Romankiw, L.T., Xu, H.: Recent developments in high-moment electroplated materials for recording heads. IBM J. Res. Dev. 49, 103–126 (2005)
Mehrizi, S., Heydarzadeh Sohi, M., Shafahian, E., Khangholi, A.A.: Studies of electrical resistivity and magnetic properties of nanocrystalline CoFeCu thin films electrodeposited from citrate-added baths. J. Mater. Sci., Mater. Electron. 23(6), 1174–1181 (2012)
Mehrizi, S., Heydarzadeh Sohi, M., Seyyed Ebrahimi, S.A.: Study of microstructure and magnetic properties of electrodeposited nanocrystalline CoFeNiCu thin films. Surf. Coat. Technol. 205, 4757–4763 (2011)
Karaagac, O., Kockar, H., Alper, M.: Electrodeposited cobalt films: alteration caused by the electrolyte pH. J. Supercond. Nov. Magn. 24, 801–804 (2011)
Celalettin Baykul, M., Sarac, U., Alper, M.: Surface morphology, structural and magnetic properties of electrodeposited NiFeCu/Cu films. J. Supercond. Nov. Magn. (2001). doi:10.1007/s10948-011-1225-2
Zhang, Y., Ivey, D.G.: Characterization of Co–Fe and Co–Fe–Ni soft magnetic films electrodeposited from citrate-stabilized sulfate baths. Mater. Sci. Eng. B, Solid-State Mater. Adv. Technol. 140, 15–22 (2007)
Zhang, Y., Ivey, D.G.: Electroplating of nanocrystalline CoFeNi soft magnetic thin films from a stable citrate-based bath. Chem. Mater. 16, 1189–1194 (2004)
Mueller, B.: ChemEQL, a program to calculate chemical speciation, Version 3.0. Limnological Research Center EAWAG/ETH, CH-6047 Kastanienbaum, Switzerland (1996)
Martell, A.E., Smith, R.M.: Critical Stability Constants, vol. 6. Plenum Press, London/New York (1989)
Martell, A.E., Smith, R.M.: Critical Stability Constants, vol. 6. Plenum Press, London/New York (1974)
Kanani, N.: Electroplating—Basic Principle, Processes and Practice. Elsevier, Berlin (2004)
Bakar, H.: ASM Handbook, Vol. 3: Alloy Phase Diagrams. ASM International, Materials Park (1992)
Acknowledgements
The authors would like to thank University of Tehran and Iranian nanotechnology initiative council for financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mehrizi, S., Heydarzadeh Sohi, M. Soft Magnetic Properties and Electrochemical Behavior of Nanocrystalline CoFeCu Thin Films Electrodeposited from Citrate-Added Baths. J Supercond Nov Magn 27, 1701–1708 (2014). https://doi.org/10.1007/s10948-014-2488-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10948-014-2488-1