Abstract
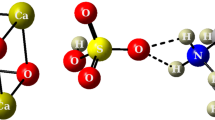
The reaction mechanism of the formation of alkali metal ethynides C2H2 + MOH → C2HM + H2O (M = Li, Na, K) is studied for the gas phase (MP2/6-311++G**//RHF/6-31+G*) and also with regard to the solvent effect of dimethyl sulfoxide (DMSO) included within the continuum model. Among all acetylene complexes with alkali metal hydroxides considered (C2H2·MOH (M = Li, Na, K)), only the complex with KOH is thermodynamically stable in DMSO solution. The formation of this structure results in activation of the acetylene molecule towards electrophilic attack. The formation of alkali metal ethynide in solution is also thermodynamically favorable only in the system with potassium hydroxide of a whole series of metals considered. Further, the ethynide ion can interact in KCCK·HOH systems.
Similar content being viewed by others
References
B. A. Trofimov, Zh. Organ. Khim., 31, No. 9, 1368–1387 (1995).
B. A. Trofimov, Curr. Organ. Chem., 6, No. 13, 1121–1162 (2002).
B. A. Trofimov, Sovremennye Problemy Organicheskoi Khimii, Vyp. 14, 131–175, SPb (2004).
B. A. Trofimov, V. V. Nosyreva, and A.G. Mal’kina, Zh. Organ. Khim., 41, No. 9, 1282–1286 (2005).
R. J. Tedeschi, J. Org. Chem., 30, 3045–3049 (1965).
N. M. Vitkovskaya, V. B. Kobychev, N. Yu. Matvienko, et al., Izv. Akad. Nauk, Ser. Khim., No. 8, 1793–1797 (1990).
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, et al., J. Comput. Chem., 14, 1347–1363 (1993).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian-98, Revision A. 6, Gaussian, Inc., Pittsburgh PA (1998).
C. Gonzalez and H. B. Schlegel, J. Phys. Chem., 94, 5523–5527 (1990).
V. B. Kobychev, J. Struct. Chem., 45, No. 1, 20–28 (2004).
V. B. Kobychev, Dokt. Diss. [in Russian], Irkutsk (2004).
L. Onsager, J. Am. Chem. Soc., 58, 1486–1493 (1936).
J. G. Kirkwood, J. Chem. Phys., 2, 351–361 (1934).
O. Tapia and O. Goscinski, Mol. Phys., 29, 1653–1661 (1975).
A. D. Becke, J. Chem. Phys., 98, 5648–5652 (1993).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev. B, 37, 785–789 (1988).
J. Tomasi, B. Mennucci, and E. Cancès, J. Mol. Struct. (Theochem.), 464, 211–226 (1999).
G. I. Almerindo, D. W. Tondo, J. R. Pliego, and Jr., J. Phys. Chem. A, 108, 166–171 (2004).
V. Barone, M. Cossi, and J. Tomasi, J. Chem. Phys., 107, 3210–3221 (1997).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text Copyright © 2009 by E. Yu. Larionova, N. M. Vitkovskaya, V. B. Kobychev, N. V. Kaempf, A. D. Skitnevskaya, and B. A. Trofimov
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 50, No. 1, pp. 33–39, January–February, 2009.
Rights and permissions
About this article
Cite this article
Larionova, E.Y., Vitkovskaya, N.M., Kobychev, V.B. et al. AB initio quantum chemical study of the reaction mechanism of ethynide ion formation in the C2H2/MOH/DMSO system (M = Li, Na, K). J Struct Chem 50, 27–33 (2009). https://doi.org/10.1007/s10947-009-0004-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10947-009-0004-4