Abstract
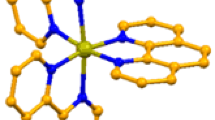
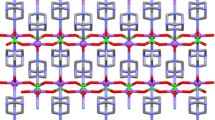
Double complex [RuNO(NH3)4OH][PtCl4] (I) and [RuNO(NH3)4OH][PdCl4] (II) salts have been prepared and explored with TGA, IR spectroscopy, powder and single crystals X-ray diffraction. Crystal phases of I and II are isostructural (space group Cmc21) and have the following crystal chemical characteristics: a = 8.106 Å, b = 18.190(3) Å, c = 8.097 Å, V = 1194.0 Å3, Z = 4, ρcalc = 3.077 g/cm3 (I), and a = 8.116 Å, b = 18.135 Å, c = 8.062 Å, V = 1186.5 Å3, Z = 4, ρcalc = 2.600 g/cm3 (II). The product of thermal decomposition of I in inert and hydrogen atmospheres is a substitution solid solution Pt0.5Ru0.5 with the parameter of the FCC unit cell a = 3.856(3) Å. Thermolysis of II affords two-phase mixtures of limited solid solutions of the metals featuring Ru-based HCP and Pd-based FCC cells.
Similar content being viewed by others
References
L. Carrette, K. A. Friedrich, and U. Stimming, Fuel Cells., 1, No. 1, 5–39 (2001).
A. Pozio, R. F. Silva, M. D. Franchesco, et al., Electrochim. Acta, 48, 255 (2002).
V. A. Emel’yanov, S. P. Khranenko, and A. V. Belyaev, Zh. Neorg. Khim., 46, No. 3, 404–410 (2001).
M. J. Clarke, Coord. Chem. Rev., 232, 69–93 (2002).
N. M. Sinitzyn, V. N. Kokunova, and A.A. Svetlov, Zh. Neorg. Khim., 27, No. 9, 2335–2342 (1982).
N. M. Sinitzyn, V. N. Kokunova, and A. A. Svetlov, ibid., 33, No. 9, 2340–2346 (1988).
N. M. Sinitzyn, V. N. Kokunova, and G. G. Novitzkii, ibid., 30, No. 11, 2870–2875 (1985).
V. N. Kokunova and V. V. Kravchenko, Koordinats. Khim., 23, No. 9, 686–689 (1997).
Yu. V. Shubin and S. V. Korenev, Zh. Neorg. Khim., 47, No. 11, 1812–1816 (2002).
I. I. Chernyaev (ed.), Preparation of Coordination Compounds of Platinum Group Metals [n Russian], Nauka, Moscow (1964).
E. E. Mercer, W. A. McAlister, and J. R. Durig, Inorg. Chem., 5, No. 11, 1881–1886 (1966).
The International Centre for Diffraction Data Powder Diffraction File 2001 (ICDD/JCPDS PDF 2001).
G. M. Sheldrick, SHELX-97, release 97-1, Univ. Göttingen, Germany (1997).
W. Kraus and G. Nolze, J. Appl. Crystallogr., 9, 301–303 (1996).
F. Bottomley, J. Chem. Soc. Dalton, 15, 1600–1605 (1974).
S. Ohba, S. Sato, and Y. Saito, Acta Crystallogr. B, 39, 49–53 (1983).
S. A. Gromilov, V. I. Alekseev, I. A. Baidina, et al, Zh. Neorg. Khim., 39, No. 12, 2017–2021 (1994).
M. A. Il’in, V. A. Emel’yanov, I. A. Baidina, and I. V. Korol’kov, Theses of the IV National Crystal Chemical Conference [in Russian], Chernogolovka (2006), pp. 159/160.
A. A. Svetlov, N. M. Sinitzyn, and V. V. Kravchenko, Zh. Neorg. Khim., 32, No. 4, 953–960 (1989).
N. M. Sinitzyn and A. A. Svetlov, Koordinats. Khim., 3, No. 4, 593–598 (1977).
N. M. Sinitzyn and A. A. Svetlov, ibid., 2, No. 10, 1381–1395 (1976).
N. M. Sinitzyn, A. A. Svetlov, and N. V. Brykova, ibid., 2, No. 5, 662–670 (1976).
N. P. Lyakishev (ed.), Phase Diagrams of Double Metal Systems: Reference Book [in Russian], V. 3, book 1, Masshinostroenie, Moscow (2001).
D. Chu and S. Gilman, J. Electrochem. Soc., 143, No. 5, 1685–1690 (1996).
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 48, No.1, pp.114–121, January–February, 2007.
Rights and permissions
About this article
Cite this article
Plyusnina, O.A., Emel’yanov, V.A., Baidina, I.A. et al. Synthesis, structure, and properties of [RuNO(NH3)4OH][PtCl4] and [RuNO(NH3)4OH][PdCl4]. J Struct Chem 48, 114–121 (2007). https://doi.org/10.1007/s10947-007-0017-9
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10947-007-0017-9