Abstract
A number of experiments on the synthesis of SAPO-11 crystals in different media (water; equimolar H2O/EtOH; low H2O/EtOH), using two methods of precursor mixing and various temperature regimes of hydrothermal treatment (HT) has been performed. It has turned out that one-stage HT at 200 °C had not provided SAPO-11 crystallization in low H2O/EtOH medium, only non-porous aluminum phosphates had been obtained, while a two-stage HT at 200 and 120 °C on the first and second stages, respectively, had allowed us to synthesize the AEL phase. Whereas equimolar water–ethanol synthesis medium has resulted in a mixture of both microporous and non-porous phases. The last one is apparently the result of the unusual properties of water–ethanol crystallization system. SAPO-11 samples which were obtained in different media have possessed “screw-like”, “tubular-like”, and “spindle-like” morphologies. SAPO-11 synthesized in the medium with the predominance of ethanol has had significant changes in physicochemical properties compared with ones obtained in water.

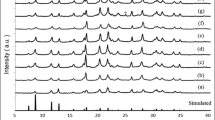
taken from open database http://www.crystallography.net

taken from open database http://www.crystallography.net








Similar content being viewed by others
Data availability
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Code availability
Not applicable.
References
C.S. Cundy, P.A. Cox, Chem. Rev. (2003). https://doi.org/10.1021/cr020060i
S.T. Wilson, B.M. Lok, C.A. Messina, T.R. Cannan, E.M. Flanigen, J. Am Soc. (1982). https://doi.org/10.1021/ja00368a062
B.M. Lok, C.A. Messina, R.L. Patton, R.T. Gajek, T.R. Cannan, E.M. Flanigen, J. Am. Chem. Soc. (1984). https://doi.org/10.1021/ja00332a063
R.M. Barrer, Zeolites (1981). https://doi.org/10.1016/S0144-2449(81)80001-2
T. Ennaert, Van Aelst, J. Dijkmans, R. De Clercq, W. Schutyser, M. Dusselier, D. Verboekend, B.F. Sels, Chem. Soc. Rev. (2016) https://doi.org/10.1039/c5cs00859j
M. Moliner, C. Martínez, A. Corma, Angew. Chemie - Int. Ed. (2015). https://doi.org/10.1002/anie.201406344
N. Rangnekar, N. Mittal, B. Elyassi, Caro, M. Tsapatsis, Soc. Rev. (2015) https://doi.org/10.1039/c5cs00292c
J. Coronas, Chem. Eng. J. (2010). https://doi.org/10.1016/j.cej.2009.11.006
M. Dusselier, M.E. Davis, Chem. Rev. (2018). https://doi.org/10.1021/acs.chemrev.7b00738
S. Mintova, J. Grand, V. Valtchev, Comptes Rendus Chim. (2016). https://doi.org/10.1016/j.crci.2015.11.005
C.S. Cundy, P.A. Cox, Micropor Mesopor Mat. (2005). https://doi.org/10.1016/j.micromeso.2005.02.016
D. Karami, S. Rohani, Rev. Chem. Eng. (2007). https://doi.org/10.1515/REVCE.2007.23.1.1
K.A. Sashkina, Z. Qi, W. Wu, A.B. Ayupov, A.I. Lysikov, E.V. Parkhomchuk, Micropor Mesopor Mat. (2017). https://doi.org/10.1016/j.micromeso.2017.02.060
S. Li, J. Li, M. Dong, S. Fan, T. Zhao, J. Wang, W. Fan, Chem. Soc. Rev. (2019). https://doi.org/10.1039/c8cs00774h
T. Sano, S. Wakabayashi, Y. Oumi, T. Uozumi, Micropor Mesopor Mat. (2001). https://doi.org/10.1016/S1387-1811(01)00285-2
S. Qiu, J. Yu, G. Zhu, O. Terasaki, Y. Nozue, W. Pang, R. Xu, Micropor Mesopor Mat. (1998). https://doi.org/10.1016/S1387-1811(98)00048-1
R. Roldán, M. Sánchez-Sánchez, G. Sankar, F.J. Romero-Salguero, C. Jiménez-Sanchidrián, Micropor Mesopor Mat. (2007). https://doi.org/10.1016/j.micromeso.2006.09.035
X. Ren, S. Komarneni, D.M. Roy, Zeolites (1991). https://doi.org/10.1016/0144-2449(91)80408-R
M. Alfonzo, J. Goldwasser, C.M. Lpez, F.J. Machado, M. Matjushin, B. Mendez, M.M.R. De Agudelo, J. Mol. Catal. A Chem. (1995). https://doi.org/10.1016/1381-1169(95)00004-6
P. Liu, J. Ren, Y. Sun, J. Fuel Chem. Technol. (2008). https://doi.org/10.1016/S1872-5813(08)60035-3
P. Liu, J. Ren, Y. Sun, Catal. Commun. (2008). https://doi.org/10.1016/j.catcom.2008.01.030
D. Fan, P. Tian, S. Xu, D. Wang, Y. Yang, J. Li, Q. Wang, M. Yang, Z. Liu, New J. Chem. (2016). https://doi.org/10.1039/C5NJ02351C
G. Liu, P. Tian, J. Li, D. Zhang, F. Zhou, Z. Liu, Micropor Mesopor Mat. (2008). https://doi.org/10.1016/J.MICROMESO.2007.07.023
E. Dumitriu, A. Azzouz, V. Hulea, D. Lutic, H. Kessler, Micropor Mater. (1997). https://doi.org/10.1016/S0927-6513(96)00107-1
H. Weyda, H. Lechert, Zeolites (1990). https://doi.org/10.1016/0144-2449(94)90136-8
M. Razavian, R. Halladj, S. Askari, Rev. Adv. Mater. Sci. (2011)
Z. Chen, W. Song, S. Zhu, W. Lai, X. Yi, W. Fang, RSC Adv. (2017). https://doi.org/10.1039/C6RA26522G
Z. Chen, X. Li, Y. Xu, Y. Dong, W. Lai, W. Fang, X. Yi, Catal. Commun. (2018). https://doi.org/10.1016/j.catcom.2017.09.002
Z. Chen, Y. Dong, S. Jiang, W. Song, W. Lai, X. Yi, W. Fang, J. Mater. Sci. (2017). https://doi.org/10.1007/s10853-016-0692-z
M. Luo, D. Wang, Y. Fu, G. Mao, B. Wang, Eur. J. Inorg. Chem. (2018). https://doi.org/10.1002/ejic.201800393
A.K. Sinha, S. Seelan, Appl. Catal. A Gen. (2004). https://doi.org/10.1016/j.apcata.2004.05.013
Z. Wang, Z. Tian, G. Wen, F. Teng, Y. Xu, Z. Xu, L. Lin, React. Kinet. Catal. Lett. (2006). https://doi.org/10.1556/RKCL.88.2006.1.11
Z. Liu, W. Xu, G. Yang, R. Xu, Micropor Mesopor Mat. (1998). https://doi.org/10.1016/S1387-1811(98)00105-X
I.A. Tiuliukova, N.A. Rudina, A.I. Lysikov, S.V. Cherepanova, E.V. Parkhomchuk, Mater. Lett. (2018). https://doi.org/10.1016/J.MATLET.2018.05.118
X.T. Xu, J.P. Zhai, I.L. Li, J.N. Tang, S.C. Ruan, Micropor Mesopor Mat. (2012). https://doi.org/10.1016/j.micromeso.2011.08.003
S.H. Jhung, Y.K. Hwang, J.S. Chang, S.E. Park, Micropor Mesopor Mat. (2004). https://doi.org/10.1016/j.micromeso.2003.10.012
A.A. Chernov, Modern crystallography III Crystal growth (Springer, Verlag, Berlin, Heidelberg, New York, Tokyo, 1984)
P. Cubillas, M.A. Holden, M.W. Anderson, Cryst. Growth Des. (2011). https://doi.org/10.1021/cg200407h
Y. Liu, Y. Lyu, X. Zhao, L. Xu, S. Mintova, Z. Yan, X. Liu, Chem. Commun. (2018). https://doi.org/10.1039/C8CC05952G
J. Grand, H. Awala, S. Mintova, CrystEngComm (2016). https://doi.org/10.1039/C5CE02286J
M. Niederberger, H. Cölfen, Phys. Chem. Chem. Phys. (2006). https://doi.org/10.1039/B604589H
D. Tian, W. Yan, X. Cao, J. Yu, R. Xu, Chem. Mater. (2008). https://doi.org/10.1021/cm703317c
Z. Chen, S. Zhu, P. Li, X. Li, Y. Xu, Y. Dong, W. Song, X. Yi, W. Fang, CrystEngComm (2017). https://doi.org/10.1039/c7ce01132f
I.V. Grenev, V.Y. Gavrilov, Micropor Mesopor Mat. (2020). https://doi.org/10.1016/j.micromeso.2019.109906
G. Sastre, D.W. Lewis, C.R.A. Catlow, J. Mol. Catal. A Chem. (1997). https://doi.org/10.1016/S1381-1169(96)00498-0
J.A. Martens, P.J. Grobet, P.A. Jacobs, J. Catal. (1990). https://doi.org/10.1016/0021-9517(90)90068-U
L. Guo, X. Bao, Y. Fan, G. Shi, H. Liu, D. Bai, J. Catal. (2012). https://doi.org/10.1016/j.jcat.2012.07.016
M. Popova, C. Minchev, V. Kanazirev, Appl. Catal. A Gen. (1998). https://doi.org/10.1016/S0926-860X(98)00003-9
G.V. Echevsky, Q. Weixing, A.V. Toktarev, W. Wei, Pet. Chem. (2016). https://doi.org/10.1134/S096554411603004X
J. Yang, O.V. Kikhtyanin, W. Wu, Y. Zhou, A.V. Toktarev, G.V. Echevsky, R. Zhang, Micropor Mesopor Mat. (2012). https://doi.org/10.1016/j.micromeso.2011.09.020
M. Sedighi, J. Towfighi, A. Mohamadalizadeh, Powder Technol. (2014). https://doi.org/10.1016/j.powtec.2014.03.045
A. Gedanken, Ultrason. Sonochem. (2004). https://doi.org/10.1016/J.ULTSONCH.2004.01.037
I.A. Shamanaeva, E.V. Parkhomchuk, Pet. Chem. (2019). https://doi.org/10.1134/S0965544119080218
L. Xin, H. Sun, R. Xu, W. Yan, Sci. Rep. (2015). https://doi.org/10.1038/srep14940
J. Yu, R. Xu, Chem. Soc. Rev. (2006). https://doi.org/10.1039/b505856m
M.R. Agliullin, Z.R. Khairullina, A.V. Faizullin, B.I. Kutepov, Pet. Chem. (2019). https://doi.org/10.1134/S0965544119030010
A.K. Sinha, S. Sivasanker, P. Ratnasamy, Ind. Eng. Chem. Prod. Res. Dev. (1998). https://doi.org/10.1021/ie9707228
G. Liu, P. Tian, Z. Liu, Chinese. J. Catal. (2012). https://doi.org/10.1016/S1872-2067(11)60325-2
D. Fan, P. Tian, S. Xu, Q. Xia, X. Su, L. Zhang, Y. Zhang, Y. He, Z. Liu, J. Mater. Chem. (2012). https://doi.org/10.1039/c2jm15281a
Y. Wang, S.-L. Chen, Y.-J. Jiang, Y.-Q. Cao, F. Chen, W.-K. Chang, Y.-L. Gao, RSC Adv. (2016). https://doi.org/10.1039/C6RA23048B
D. Wang, P. Tian, D. Fan, M. Yang, B. Gao, Y. Qiao, C. Wang, Z. Liu, J. Colloid Interface Sci. (2015). https://doi.org/10.1016/J.JCIS.2014.12.029
W. Yan, X. Song, R. Xu, Micropor Mesopor Mat. (2009). https://doi.org/10.1016/j.micromeso.2009.03.023
F. Franks, D.J.G. Ives, Q. Rev, Chem. Soc. (1966). https://doi.org/10.1039/qr9662000001
A.K. Soper, L. Dougan, J. Crain, J.L. Finney, J. Phys. Chem. B (2006). https://doi.org/10.1021/jp054556q
I.S. Khattab, F. Bandarkar, M.A.A. Fakhree, A. Jouyban, Korean J. Chem. Eng. (2012). https://doi.org/10.1007/s11814-011-0239-6
A. Wakisaka, K. Matsuura, J. Mol. Liq. (2006). https://doi.org/10.1016/j.molliq.2006.08.010
P. Petong, R. Pottel, U. Kaatze, J. Phys. Chem. A. (2000). https://doi.org/10.1021/jp001393r
N. Nishi, S. Takahashi, M. Matsumoto, A. Tanaka, K. Muraya, T. Takamuku, T. Yamaguchi, J. Phys. Chem. (2005). https://doi.org/10.1021/j100001a068
S. Burikov, T. Dolenko, S. Patsaeva, Y. Starokurov, V. Yuzhakov, Mol. Phys. (2010). https://doi.org/10.1080/00268976.2010.516277
M. Matsumoto, N. Nishi, T. Furusawa, M. Saita, T. Takamuku, M. Yamagami, T. Yamaguchi, Bull. Chem. Soc. Jpn. (1995)
Q. Dong, C. Yu, L. Li, L. Nie, D. Li, H. Zang, Acta Part A Mol. Biomol. Spectros (2019). https://doi.org/10.1016/J.SAA.2019.117183
K. Mizuno, Y. Miyashita, Y. Shindo, H. Ogawa, J. Phys. Chem. (1995). https://doi.org/10.1021/j100010a037
K. Egashira, N. Nishi, J. Phys. Chem. B. (1998). https://doi.org/10.1021/jp980635
Acknowledgements
The authors are grateful to Alekseeva N.A. (XRD patterns), Rudina N.A. (SEM images obtaining), Lysikov A.I. (TPD-NH3 measurements) and Leonova A.A. (texture measurements). This work was supported by the Ministry of Science and Higher Education of the Russian Federation within the governmental order for Boreskov Institute of Catalysis (project AAAA-A21-121011490008-3).
Funding
This work was supported by the Ministry of Science and Higher Education of the Russian Federation within the governmental order for Boreskov Institute of Catalysis (project AAAA-A21-121011490008–3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shamanaeva, I.A., Parkhomchuk, E.V. Variability of molecular sieve SAPO-11 crystals: acidity, texture, and morphology. J Porous Mater 29, 481–492 (2022). https://doi.org/10.1007/s10934-021-01177-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-021-01177-y