Abstract
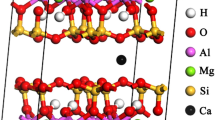
The dynamics and thermodynamics of adsorption of hexadecyl ammonium with different numbers of carbon chains in montmorillonite (Mt) with different layer charge density and the structure of hexadecyl ammonium/Na-Mt composites were studied in this paper. The dynamic results show that the adsorption process of hexadecyl ammonium in Na-Mt fits well with the quasi-second-order dynamics equation. The reaction rate constant k is correlated negatively with the numbers of carbon chains and the dosages of hexadecyl ammonium, but positively with the layer charge density of Na-Mt. The thermodynamic results show that the adsorption of hexadecyl ammonium in Na-Mt is a spontaneous and exothermic process, in which the entropy change ΔS first increases and then decreases. The adsorption efficiency has a positive correlation with the numbers of carbon chains and the layer charge density, but a negative correlation with the dosage of hexadecyl ammonium. Under the same dosage of hexadecyl ammonium, when the layer charge density of Na-Mt is lower, fewer carbon chains are conducive to the spontaneous reaction. On the contrary, when the layer charge density of Na-Mt is higher, a greater number of carbon chains are beneficial to the spontaneous reaction. The results of structural characterization of hexadecyl ammonium/Na-Mt show that with the increase of the numbers of carbon chains, hexadecyl ammonium dosage and Na-Mt layer charge density, the d(001) value and orderly degree of hexadecyl ammonium/Na-Mt increase, but its BET surface area, pore volume and most probable pore radius decrease.









Similar content being viewed by others
References
Y. Zheng, A. Zaoui, Mechanical behavior in hydrated Na-montmorillonite clay. Physica A Statal Mechanics Appl. 505, 582–590 (2018). https://doi.org/10.1016/j.physa.2018.03.093
G. Wang, S. Zhang, J. Wang, S. Ma, X. Lu, S. Komarneni, Synthesis of porous Al pillared montmorillonite after pre-intercalation with dodecylamine: textural and thermal properties. J. Porous Mater. 23(6), 1687–1694 (2016). https://doi.org/10.1007/s10934-016-0276-y
Q. Zhang, R. Jing, S. Zhao, M. Wu, X. Liu, Y. Shao, F. Lv, A. Liu, Z. Meng, Adsorption of cationic and anionic dyes on montmorillonite in single and mixed wastewater. J. Porous Mater. 26(6), 1861–1867 (2019). https://doi.org/10.1007/s10934-019-00782-2
Z. Sun, J. Xu, G. Wang, A. Song, S. Zheng, Hydrothermal fabrication of rectorite based biocomposite modified by chitosan derived carbon nanoparticles as efficient mycotoxins adsorbents. Appl. Clay Sci. 184, 105373 (2019). https://doi.org/10.1016/j.clay.2019.105373
J. Yang, K. Yu, C. Liu, Chromium immobilization in soil using quaternary ammonium cations modified montmorillonite: Characterization and mechanism. J Hazard Mater 321, 73–80 (2016). https://doi.org/10.1016/j.jhazmat.2016.09.003
J.L. Alves, P.D.T.V.E. Rosa, A.R. Morales, Evaluation of organic modification of montmorillonite with ionic and nonionic surfactants. Appl. Clay Sci. 150, 23–33 (2017)
Siyu Peng, Taoyan Mao, XuWu. Cheng Zheng, Yuan Wei, Polyhydroxyl gemini surfactant-modified montmorillonite for efficient removal of methyl orange—ScienceDirect. Colloids Surf. A Physicochem. Eng. Asp. 578, 123602–123602 (2019). https://doi.org/10.1016/j.colsurfa.2019.123602
E.E. Yalçınkaya, F.O. Pelit, İ Güney, H. Türkmen, Ionic liquid intercalated clay nanofillers for chromatographic applications. J. Porous Mater. 21(6), 1151–1158 (2014). https://doi.org/10.1007/s10934-014-9867-7
Y. Li, X. Hu, X. Liu, Y. Zhang, S. Tian, Adsorption behavior of phenol by reversible surfactant-modified montmorillonite: Mechanism, thermodynamics, and regeneration. Chem. Eng. J. 334 (2018)
G. Wang, X. Wang, S. Zhang, S. Ma, Y. Wang, J. Qiu, Adsorption of heavy metal and organic pollutant by organo-montmorillonites in binary-component system. J. Porous Mater. 27(5), 1515–1522 (2020). https://doi.org/10.1007/s10934-020-00927-8
L.L. Pluart, J. Duchet, H. Sautereau, J.F. Gérard, Tailored interfaces in nanocomposites. Macromol. Symp. 194(1), 155–160 (2003). https://doi.org/10.1002/masy.200390077
Z. Sun, C. Lian, C. Li, S. Zheng, Investigations on organo-montmorillonites modified by binary nonionic/zwitterionic surfactant mixtures for simultaneous adsorption of aflatoxin B1 and zearalenone. J. Colloid Interface Sci. 565, 11–22 (2020). https://doi.org/10.1016/j.jcis.2020.01.013
P.G. Slade, W.P. Gates, The swelling of HDTMA smectites as influenced by their preparation and layer charges. Appl. Clay Sci. 25(1–2), 93–101 (2004). https://doi.org/10.1016/j.clay.2003.07.007
S.Y. Lee, W.J. Cho, P.S. Hahn, M. Lee, Y.B. Lee, K.J. Kim, Microstructural changes of reference montmorillonites by cationic surfactants. Appl. Clay Sci. 30(3–4), 174–180 (2005). https://doi.org/10.1016/j.clay.2005.03.009
Y.Q. Liu, X.J. Lu, J. Qiu, Characterization of the Alkyl-Ammonium Adsorption amount in organomontmorillonite and its effect on the viscosity of organomontmorillonite Gel. Adv. Mater. Res. 158, 25–34 (2010). https://doi.org/10.4028/www.scientific.net/AMR.158.25
K. Taleb, I. Pillin, Y. Grohens, S. Saidi-Besbes, Gemini surfactant modified clays: effect of surfactant loading and spacer length. Appl. Clay Sci. 161, 48–56 (2018). https://doi.org/10.1016/j.clay.2018.03.015
K.J. Lin, U.S. Jeng, K.F. Lin, Adsorption and intercalation processes of ionic surfactants on montmorillonite associated with their ionic charge. Mater. Chem. Phys. 131(1–2), 120–126 (2011). https://doi.org/10.1016/j.matchemphys.2011.07.076
D. Chen, J. Chen, X. Luan, H. Ji, Z. Xia, Characterization of anion–cationic surfactants modified montmorillonite and its application for the removal of methyl orange. Chem Eng J 171(3), 1150–1158 (2011). https://doi.org/10.1016/j.cej.2011.05.013
J. Ma, Y. Lei, M.A. Khan, F. Wang, Y. Chu, W. Lei, M. Xia, S. Zhu, Adsorption properties, kinetics & thermodynamics of tetracycline on carboxymethyl-chitosan reformed montmorillonite. Int. J. Biol. Macromol. 124, 557–567 (2019). https://doi.org/10.1016/j.ijbiomac.2018.11.235
A. Czimerova, J. Bujdak, R. Dohrmann, Traditional and novel methods for estimating the layer charge of smectites. Appl. Clay Sci. 34(1–4), 2–13 (2006). https://doi.org/10.1016/j.clay.2006.02.008
Ö. AçışIı, S. Karaca, A. Gürses, Investigation of the alkyl chain lengths of surfactants on their adsorption by montmorillonite (Mt) from aqueous solutions. Appl. Clay Sci. 142, 90–99 (2016). https://doi.org/10.1016/j.clay.2016.12.009
S.J. Sekewael, K. Wijaya, T. Triyono, Chemical modification of montmorillonite K10 and its catalytic activity. Asian J. Chem. 32(3), 659 (2020). https://doi.org/10.14233/ajchem.2020.22216
X.J. Lu, J. Qiu, Y.Q. Liu, P. Chen, Effect of preparation parameters on the structure and property of Montmorillonite/Alkylammonium Complexes. Adv. Mater. Res. 1049 (2011)
E.M. Daoudi, Y. Boughaleb, L. El Gaini, I. Meghea, M. Bakasse, Modeling of alkyl quaternary ammonium cations intercalated into montmorillonite lattice. Mater. Res. Bull. 48(5), 1824–1829 (2013)
J. Qiu, D. Liu, Y. Wang, G. Chen, S. Jiang, G. Li, Y. Wang, W. Wang, P. Wu, X. Liu, G. Wang, X. Lyu, Comprehensive characterization of the structure and gel property of organo-montmorillonite: effect of layer charge density of montmorillonite and carbon chain length of alkyl ammonium. Minerals-Basel (2020). https://doi.org/10.3390/min10040378
J. Qiu, K. Cui, G. Chen, Y. Wang, X. Lyu, Micro-structure and gel performance of octadecyl trimethyl ammonium chloride intercalated montmorillonite. Colloids Surf. A Physicochem. Eng. Asp. 610, 125710 (2020). https://doi.org/10.1016/j.colsurfa.2020.125710
J. Qiu, S. Jiang, Y. Wang, G. Chen, D. Liu, X. Liu, G. Wang, P. Wu, X. Lyu, Crystal chemistry characteristics and dispersion performance of Ca-montmorillonite with different layer charge density. Mater. Res. Express 7(7), 075505 (2020). https://doi.org/10.1088/2053-1591/aba803
L. Janovák, J. Varga, L. Kemény, I. Dékány, Swelling properties of copolymer hydrogels in the presence of montmorillonite and alkylammonium montmorillonite. Appl. Clay Sci. 43(2), 260–270 (2009). https://doi.org/10.1016/j.clay.2008.08.002
M. Ghavami, Q. Zhao, S. Javadi, J.S.D. Jangam, J.B. Jasinski, N. Saraei, Change of organo bentonite interlayer microstructure induced by sorption of aromatic and petroleum hydrocarbons A combined study of laboratory characterization and molecular dynamics simulations. Colloids Surf. A 520, 324–334 (2017). https://doi.org/10.1016/j.colsurfa.2017.01.038
Q. Yang, M. Gao, W. Zang, Comparative study of 2,4,6-trichlorophenol adsorption by montmorillonites functionalized with surfactants differing in the number of head group and alkyl chain. Colloids Surf. A 520, 805–816 (2017). https://doi.org/10.1016/j.colsurfa.2017.02.057
P. Praus, M. Turicová, S. Študentová, M. Ritz, Study of cetyltrimethylammonium and cetylpyridinium adsorption on montmorillonite. J. Colloid Interface Sci. 304(1), 29–36 (2006). https://doi.org/10.1016/j.jcis.2006.08.038
H. Zhang, J. Ma, F. Wang, Y. Chu, M. Xia, Mechanism of carboxymethyl chitosan hybrid montmorillonite and adsorption of Pb(II) and Congo red by CMC-MMT organic-inorganic hybrid composite. Int. J. Biol. Macromol. 149, 1161–1169 (2020). https://doi.org/10.1016/j.ijbiomac.2020.01.201
H. Xing, Y. Liang, G. Liu, Y. Zhang, X. Liu, W. Fu, Organically treating montmorillonite with dual surfactants to modify bitumen. Constr. Build. Mater. (2020). https://doi.org/10.1016/j.conbuildmat.2020.120705
H. Zhang, F. Zhao, M. Xia, F. Wang, Microscopic adsorption mechanism of montmorillonite for common ciprofloxacin emerging contaminant: molecular dynamics simulation and Multiwfn wave function analysis. Colloids Surf. A Physicochem. Eng. Asp. 614, 126186 (2021). https://doi.org/10.1016/J.COLSURFA.2021.126186
T. Undabeytia, U. Shuali, S. Nir, B. Rubin, Applications of chemically modified clay minerals and clays to water purification and slow release formulations of herbicides. Minerals-Basel 11(1), 9 (2020). https://doi.org/10.3390/MIN11010009
G. Tang, C. Jia, G. Wang, P. Yu, X. Jiang, Adsorption mechanism of bacteria onto a Na-montmorillonite surface with organic and inorganic calcium. bioRxiv. (2020). https://doi.org/10.1101/2020.10.08.332536
W. Xiao, M. Zhan, Z. Li, Organically modifying and modeling analysis of montmorillonites. Mater. Des. 24(6), 455–462 (2003). https://doi.org/10.1016/s0261-3069(03)00064-5
Acknowledgements
This work was supported by grants from the National Nature Science Foundation of China: “Design of structure and gelling performance of Montmorillonite/Alkyl ammonium based on the adsorption properties of Alkyl ammonium on Montmorillonite” (No: 51774200).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qiu, J., Wang, Y., Wu, P. et al. Adsorption characteristics of hexadecyl ammonium with different numbers of carbon chains in montmorillonite and the structure of the prepared composites. J Porous Mater 28, 1675–1687 (2021). https://doi.org/10.1007/s10934-021-01114-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-021-01114-z