Abstract
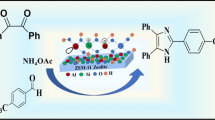
Biscoumarins, the bridge substituted dimers of 4-hydroxycoumarin, are distinguished derivatives of coumarin. In view of numerous pharmaceutical applications of biscoumarins, the construction of appropriate one-pot synthetic methods for their preparation has been a subject of great attention in recent years. In continuation of ongoing study for the construction of promising approaches for the preparation of biscoumarins, herein we report a zeolite-mediated methodology, in which zeolites, for example, NaY, HY, HZSM-5 and H-beta were employed as catalysts to investigate their catalytic potencies in the synthesis of biscoumarins. In this approach, aromatic aldehydes were treated with 4-hydroxycoumarin in the presence of zeolite catalyst in refluxing ethanol for 2–3 h to furnish biscoumarins in 80–96% yield. The yield of biscoumarins increased in the following order NaY < HZSM-5 < H-beta < HY. Aldehydes both with electron-donating and electron-withdrawing units underwent smooth conversion to their respective biscoumarins without the development of any side products. The cost-effective, eco-friendly and ready available zeolite catalyst could be recovered and reused several times. This methodology offers operational simplicity and avoids the use of toxic solvents and corrosive reagents. The present approach offers remarkable advantages such as high catalytic activity, straight-forward procedure, simple workup and low catalyst loading.



Similar content being viewed by others
References
M. Faisal, A. Saeed, D. Shahzad, T.A. Fattah, B. Lal, P.A. Channar, J. Mahar, S. Saeed, P.A. Mahesar, F.A. Larik, Eur. J. Med. Chem. 141, 386 (2017)
N.O. Mahmoodi, F. Ghanbari Pirbasti, Z. Jalalifard, J. Chin. Chem. Soc. 65, 383 (2018)
M.A. Musa, J.S. Cooperwood, M.O.F. Khan, Curr. Med. Chem. 15, 2664 (2008)
K.N. Venugopala, V. Rashmi, B. Odhav, BioMed Res. Int. (2013). https://doi.org/10.1155/2013/963248
D. Yu, M. Suzuki, L. Xie, S.L. Morris-Natschke, K.H. Lee, Med. Res. Rev. 23, 322 (2003)
P. Anand, B. Singh, N. Singh, Bioorg. Med. Chem. 20, 1175 (2012)
A. Saeed, M. Haroon, F. Muhammad, F.A. Larik, E.S. Hesham, P.A. Channar, J. Organomet. Chem. 834, 88 (2017)
A.K. Mahapatra, K. Maiti, P. Sahoo, P.K. Nandi, J. Lumin. 143, 349 (2013)
J. Li, Z. Hou, F. Li, Z.D. Zhang, Y. Zhou, X.X. Luo, M.K. Li, J. Mol. Struct. 1075, 509 (2014)
A.D. Gupta, S. Samanta, R. Mondal, A.K. Mallika, Bull. Korean Chem. Soc. 33, 4239 (2012)
R.S. Overman, M.A. Stahmann, C.F. Huebner, W.R. Sullivan, L. Spero, D.G. Doherty, M. Ikawa, L. Graf, S. Roseman, K.P. Link, J. Biol. Chem. 153, 5 (1944)
J. Lehmann, Lancet. 241, 611 (1943)
C.F. Huebner, W.R. Sullivan, M.A. Stahmann, K.P. Link, J. Am. Chem. Soc. 65, 2292 (1943)
H. Zhao, N. Neamati, H. Hong, A. Mazumder, S. Wang, S. Sunder, G.W. Milne, Y. Pommier, T.R. Burke, J. Med. Chem. 40, 920 (1997)
E.B. Yang, Y.N. Zhao, K. Zhang, P. Mack, Biochem. Biophys. Res. Commun. 260, 682 (1999)
K. Franke, A. Porzel, J. Schmidt, Phytochem. 61, 873 (2002)
M. Riaz, A. Malik, Heterocycles 55, 769 (2001)
A. Mazumder, S. Wang, N. Neamati, M. Nicklaus, S. Sunder, J. Chen, G.W.A. Milne, W.G. Rice, Y. Pommier, J. Med. Chem. 40, 2472 (1996)
H. Mehrabi, H. Abusaidi, J. Iran. Chem. Soc. 7, 890 (2010)
K.M. Khan, S. Iqbal, M.A. Lodhi, G.M. Maharvi, M.I. Choudhary, S. Perveen, Bioorg. Med. Chem. 12, 1963 (2004)
K. Tabatabaeian, H. Heidari, A. Khorshidi, M. Mamaghani, N.O. Mahmoodi, J. Serb. Chem. Soc. 77, 407 (2012)
R. Gasparova, K. Kotlebova, M. Lacova, Nava Biotech. 3, 349 (2009)
B. Banerjee, Ultrason. Sonochem 35, 1 (2017)
A.M. Al-Kadasi, G.M. Nazeruddin, Int. J. Chem. Sci. 10, 324 (2012)
H.R. Shaterian, M. Honarmand, Chin. J. Chem. 27, 1795 (2009)
N. Hamdi, M.C. Puerta, P. Valerga, Eur. J. Med. Chem. 43, 2541 (2008)
B. Karmakar, A. Nayak, J. Banerji, Tetrahedron Lett. 53, 4343 (2012)
B. Sadeghi, T. Ziya, J. Chem. (2013). https://doi.org/10.1155/2013/179013
R. Karimian, F. Piri, A.A. Safari, S.J. Davarpanah, J. Nanostr. Chem. 3, 1 (2013)
J.F. Zhou, G.X. Gong, L.T. An, S.X. Jun, F.X. Zhu, Chin. J. Org. Chem. 29, 1988 (2009)
Z.N. Siddiqui, F. Farooq, Catal. Sci. Technol. 1, 810 (2011)
E. Sheikhhosseini, Trend Mod. Chem. 3, 34 (2012)
H. Hagiwara, K. Sato, T. uzuki, M. Ando, Heterocycles. 51, 497 (1999)
H. Hagiwara, N. Fujimoto, T. Suzuki, M. Ando, Heterocycles. 53, 549 (2000)
J.N. Sangshetti, N.D. Kokare, D.B. Shinde, Green. Chem. Lett. Rev. 2, 233 (2009)
M. Kidwai, V. Bansal, P. Mothsra, S. Saxena, R.K. Somvanshi, S. Dey, T.P. Singh, J. Mol. Catal. A. Chem. 268, 76 (2007)
P. Singh, P. Kumar, A. Katyal, R. Kalra, S.K. Dass, S. Prakash, R. Chandra, Catal. Lett. 134, 303 (2010)
K. Parvanak Boroujeni, P. Ghasemi, Z. Rafienia, Monatsh. Chem. 145, 1023 (2014)
A.R. Kiasat, L. Hemat-Alian, Res. Chem. Intermed. 41, 873 (2015)
S.C. Azimi, K. Rad-Moghadam, Iran. Chem. Commun. 23, 356 (2015)
J.M. Khurana, S. Kumar, Monatsh. Chem. 141, 561 (2010)
F. Shirini, M. Abedini, S. Abroon Kiaroudi, Phos. Sul. Sili. 189, 1279 (2014)
M.A. Zolfigol, A.R. Moosavi-Zare, M. Zarei, C. R. Chim. 17, 1264 (2014)
R. Rezaei, F. Moezzi, M.M. Doroodmand, Chin. Chem. Lett. 25, 183 (2014)
J.M. Khurana, S. Kumar, Tetrahedron Lett. 50, 4125 (2009)
M. Esmaeilpour, J. Javidi, F. Dehghani, F.N. Dodeji, RSC Adv. 5, 26625 (2015)
M.M. Heravi, B. Alimadadi Jani, F. Derikvand, F.F. Bamoharram, H.A. Oskooie, Catal. Commun. 10, 272 (2008)
T. Frising, P. Leflaive, Microporous Mesoporous Mater. 114, 27 (2008)
J.A. Rabo, Zeolite Chemistry and Catalysis. (American Chemical Society, Washington DC, 1976), p 171
C.S. Cundy, Coll. Czechoslovak chem. Comm. 63, 1699 (1998)
J. Pérez-Ramírez, C.H. Christensen, K. Egeblad, C.H. Christensen, J.C. Groen, Chem. Soc. Rev. 37, 2542 (2008)
M. Hunger, Solid State Nucl. Magn. Res. 6, 1 (1996)
M. Faisal, S. Shahid, S.A. Ghumro, A. Saeed, F.A. Larik, Z. Shaheen, P.A. Channar, T.A. Fattah, S. Rasheed, P.A. Mahesar, Synth. Commun. 48, 462 (2018)
M. Faisal, D. Shahzad, A. Saeed, B. Lal, S. Saeed, F.A. Larik, P.A. Channar, P.A. Mahesar, J. Mahar, Tetrahedron: Asymmetry 28, 1145 (2017)
M. Karthik, A.K. Tripathi, N.M. Gupta, M. Palanichamy, V. Murugesan, Catal. Commun. 5, 371 (2004)
M. Karthik, C.J. Magesh, P.T. Perumal, M. Palanichamy, B. Arabindoo, V. Murugesan, Appl. Catal. A 286, 137 (2005)
H. Hassan, B.H. Hameed, Desalination 276, 45 (2011)
D.I. Petkowicz, R. Brambilla, C. Radtke, C. Da Silva, Z.N. Da Rocha, S.B.C. Pergher, J.H.Z. Dos Santos, Appl. Catal. A 357, 125 (2009)
W. Cheng-Cai, L. Chung-Kung, L. Meng-Du, J. Lain-Chuen, Dyes Pigments 76, 817 (2008)
M.A. Aramendía, J.C. Colmenares, S. López-Fernández, A. Marinas, J.M. Marinas, F.J. Urbano, Catal. Today 129, 102 (2007)
X. Liu, K.K. Iu, J.K. Thomas, Chem. Phys. Lett. 195, 163 (1992)
M. Anpo, H. Yamashita, Y. Ichihashi, Y. Fujii, M. Honda, J. Phys. Chem. B. 101, 2632 (1997)
H.G. Karge, E. Geidel, Vibrational spectroscopy. Mol. Sieves 4, 1–200 (2004)
H.G. Karge, Microporous Mesoporous Mater. 22, 547 (1998)
S. Khabtou, T. Chevreauand, J.C. Lavalley, Microporous Mater. 3, 133 (1994)
Y.W.L. Lim, Y. Tang, Y.H. Cheng, Z. Chen, Nanoscale 2, 12 (2010)
P. Andy, J. Garcia-Martinez, G. Lee, H. Gonzalez, C.W. Jones, M.E. Davis, J. Catal. 192, 215 (2000)
P.J. Kunkeler, D. Moeskops, H. van Bekkum, Microporous Mater. 11, 313 (1997)
D. Završnik, S. Muratoviæ, D. Makuc, J. Plavec, M. Cetina, A. Nagl, E.D. Clercq, J. Balzarini, M. Mintas, Molecules 16, 6023 (2011)
I. Manolov, C.M. Moessmer, N. Danchev, Eur. J. Med. Chem. 41, 882 (2006)
A. Tzani, A. Douka, A. Papadopoulos, E.A. Pavlatou, E. Voutsas, A. Detsi, ACS Sustain. Chem. Eng. 1, 1180 (2013)
S.K. Patil, D.V. Awale, M.M. Vadiyar, S.A. Patil, S.C. Bhise, S.S. Kolekar, Res. Chem. Int. 43, 5365 (2017)
A.D. Gupta, S. Samanta, R. Mondal, A.K. Mallik, Bull. Korean Chem. Soc. 33, 4239 (2012)
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Faisal, M., Larik, F.A. & Saeed, A. A highly promising approach for the one-pot synthesis of biscoumarins using HY zeolite as recyclable and green catalyst. J Porous Mater 26, 455–466 (2019). https://doi.org/10.1007/s10934-018-0625-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-018-0625-0