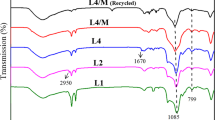
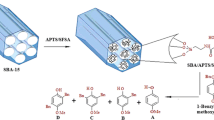
Abstract
The modified MCM-41 with aminopropyl groups was synthesized, characterized and its catalytic activity was evaluated in aza-Michael addition of various aryl tetrazoles to activated olefin esters under reflux condition. The synthesized new compounds were characterized using spectroscopic and physical methods. The catalyst was reused in five consecutive experiments without obvious loss of activity. The results showed with application of aminopropylated MCM-41 only N2 alkylation occurs on tetrazole rings. However, when triethylamine was used as the base, a mixture of N1 and N2 alkylation was observed. In addition, antimicrobial studies investigated and results indicated that the products did not show considerable antimicrobial activity.










Similar content being viewed by others
References
H.R. Meier, H. Heimgartner, in Methoden der Organischen Chemie (Houben-Weyl), Vol. E8d, ed by E. Schumann (Georg Thieme, Stuttgart, 1994)
R.J. Herr, Bioorg. Med. Chem. 10, 3379 (2002)
R.N. Butler, in Comprehensive Heterocyclic Chemistry, eds by A.R. Katritzky, C. W. Ress, E. F. V. Scriven (Pergamon, Oxford, 1996)
J. Zabrocki, G.D. Smith, J.B. Dunbar Jr, H. Iijima, G.R. Marshall, Am. Chem. Soc. 110, 5875 (1988)
M.A. Malik, Y.M. Wani, S.A. Al-Thabaiti, R.A. Shiekh, J. Incl. Phenom. Macrocycl. Chem. 78, 15 (2013)
S.J. Wittenberger, Org. Prep. Proced. Int. 26, 499 (1994)
G.-H. Tao, Y. Guo, Y.-H. Joo, B. Twamley, J.M. Shreeve, Mater. Chem. 18, 5524 (2008)
A. Razynska, A. Tempczyk, E. Maslinski, J. Szafranek, Z. Grzonka, P. Hermann, J. Chem. Soc. Perkin Trans. 2, 379 (1983)
R.N. Butler, V.C. Garvin, H. Lumbroso, C. Liegois, J. Chem. Soc. Perkin Trans. 2, 721 (1984)
W.M.F. Fabian, Z. Naturforsch. 45, 1328 (1990)
M.W. Wong, R. Leung-Toung, C. Wentrup, J. Am. Chem. Soc. 115, 2465 (1993)
A.P. Mazurek, N. Sadlej-Sosnowska, Chem. Phys. Lett. 330, 212 (2000)
N. Sadlej-Sosnowska, J. Org. Chem. 66, 8737 (2001)
A.N. Chermahini, M. Nasr-Esfahani, Z. Dalirnasab, A.H. Dabbagh, A. Teimouri, J. Mol. Struct. Theochem 820, 7 (2007)
G.I. Koldobskii, R.B. Kharbash, Russ. J. Org. Chem. 39, 453 (2003)
J. Roh, K. Vávrová, A. Hrabálek, Eur. J. Org. Chem. 2011, 6101 (2012)
B.V. Rokade, K. Gadde, K.R. Prabhu, Adv. Synth. Catal. 356, 946 (2014)
G. Aridoss, K.K. Laali, Eur. J. Org. Chem. 2012, 6343 (2011)
S. Kumar, A. Kumar, A. Agarwal, S.K. Awasthi, RSC Adv. 5, 21651 (2014)
G.M. Schroeder, S. Marshall, H. Wan, A.V. Purandare, Tetrahedron Lett. 51, 1404 (2010)
C.T. Kresge, M.E. Leonowicz, W.J. Roth, J.C. Vartuli, J.S. Beck, Nature 359, 710 (1992)
A.L. de Lima, A. Mbengue, R.A.S. San Gil, C.M. Ronconi, C.A. Mota, Catal. Today 226, 210 (2014)
R. Bal, K. Chaudhari, S. Sivasanker, Catal. Lett. 75, 75 (2000)
M.J. Climent, A. Corma, S. Iborra, L. Marti, ChemSusChem 7, 1177 (2014)
Q. Yang, M.P. Kapoor, N. Shirokura, M. Ohashi, S. Inagaki, J.N. Kondo, K. Domen, J. Mater. Chem. 15, 666 (2005)
R.V. Crieken, J.A. Melero, G. Morales, J. Mol. Catal. A 256, 29 (2006)
J.A. Posada, C.A. Cardona, O. Giraldo, Mater. Chem. Phys. 121, 215 (2010)
G.Q. Silveira, M.D. Vargas, C.M. Ronconi, J. Mater. Chem. 21, 6034 (2011)
X. Wang, S. Cheng, Catal. Commun. 7, 689 (2006)
D. Macquarrie, D. Jackson, Chem. Commun. 1781 (1997)
N. Shadjou, M. Hasanzadeh, RSC Adv. 4, 18117 (2014)
G. Sartori, F. Bigi, R. Maggi, R. Sartorio, D. Macquarrie, M. Lenarda, L. Storaro, S. Coluccia, G. Martra, J. Catal. 222, 410 (2004)
M. Sánchez-Roselló, J.L. Aceña, A. Simón-Fuentesa, C. del Pozo, Chem. Soc. Rev. 43, 7430 (2014)
M. Sánchez-Roselló, C. Mulet, M. Guerola, C. del Pozo, S. Fustero, Chem. Eur. J. 20, 15697 (2014)
P. Chauhan, D. Enders, Angew. Chem. Int. Ed. 53, 1485 (2014)
J. Cheng, Z. Huang, Y.R. Chi, Angew. Chem. Int. Ed. 52, 8592 (2013)
B.M. Reddy, M.K. Patil, B.T. Reddy, Catal. Lett. 126, 413 (2008)
S. Boncel, K. Saletra, B. Hefczyc, K.Z. Walczak, Beilstein J. Org. Chem. 7, 173 (2011)
U. Uria, J.L. Vicario, D. Badía, L. Carrillo, Chem. Commun. 2509 (2007)
J.M. Xu, C. Qian, B.K. Liu, Q. Wu, X.F. Lin, Tetrahedron 63, 986 (2007)
L. Yang, L.W. Xu, C.G. Xia, Tetrahedron Lett. 46, 3279 (2005)
G. Bartoli, M. Bartolacci, A. Giuliani, E. Marcantoni, M. Massaccesi, E. Torregiani, J. Org. Chem. 70, 169 (2005)
J. Lv, H. Wu, Y. Wang, Eur. J. Org. Chem. 11, 2073 (2010)
M. Gandelman, E.N. Jacobsen, Angew. Chem. Int. Ed. 44, 2393 (2005)
F. Han, L. Yang, Z. Li, C. Xia, Org. Biomol. Chem. 10, 346 (2012)
H. Dziklinska, S. Dzierzgowski, A. Jezewski, J. Plenkiewicz, Bull. Soc. Chim. Belg. 98, 277 (1989)
A. Alami, A. El Hallaoui, A. Elachqar, S. El Hajji, M.L. Roumestant, Ph Viallefont, Prep. Biochem. Biotechnol. 28, 167 (1998)
A.N. Chermahini, A. Teimouri, F. Momenbeik, A. Zarei, Z. Dalirnasab, A. Ghaedi, M. Roosta, J. Heterocycl. Chem. 47, 913 (2010)
A.N. Chermahini, A. Teimouri, A. Moadeli, Heteroatom Chem. 22, 168 (2011)
A. Teimouri, A.N. Chermahini, Polyhedron 30, 2606 (2011)
D. Kumar, K. Schumacher, C. du Fresne von Hohenesche, M. Grun, K.K. Unger, Coll. Surf. A Physicochem. Eng. Aspects 187–188, 109 (2001)
G. Romanelli, G. Pasquale, Á. Sathicqa, H. Thomas, J. Autino, P. Vázquez, J. Mol. Cat. A 340, 24 (2011)
V. Antochshuk, M. Jaroniec, Chem. Commun. 3, 258 (2002)
L.T. Gibson, Chem. Soc. Rev. 43, 5163 (2014)
G.I. Koldobskii, V.A. Ostrovskii, Russ. Chem. Rev. 63, 797 (1981)
H. Singh, A.S. Chawla, V.K. Kapoor, D. Paul, R.K. Malhorta in Progress in Medicinal Chemistry, vol 17, eds by G.P. Ellis, G.B. (West 1980), p. 151
Acknowledgments
This work was financially supported by the Ms.C. Programs Foundation of Yasouj University and Isfahan University of Technology.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Najafi Chermahini, A., Azadi, M., Tafakori, E. et al. Amino-functionalized mesoporous silica as solid base catalyst for regioselective aza-Michael reaction of aryl tetrazoles. J Porous Mater 23, 441–451 (2016). https://doi.org/10.1007/s10934-015-0098-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-015-0098-3