Abstract
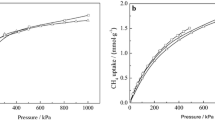
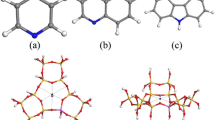
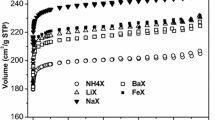
The adsorption of nitrogen, oxygen and argon has been studied in cadmium (II) cations exchanged zeolite X at 288.2 and 303.2 K. Experimentally measured adsorption isotherms are compared with theoretically calculated data using grand canonical Monte Carlo (GCMC) simulation. Nitrogen showed higher adsorption capacity and selectivity than oxygen and argon in these zeolite samples. The cadmium exchanged zeolite X was showed that increased adsorption capacity for nitrogen, oxygen, and argon with increase in Cd (II)-exchange levels, indicating as charge density increases adsorption capacity also increase. Isosteric heat of adsorption data showed stronger interactions of nitrogen molecules with cadmium cations in zeolite samples. These observations have been explained in terms of higher electrostatic interaction of nitrogen with extra framework zeolite cations. The selectivity of oxygen over argon is explained in terms of its higher interaction with Cd (II)-exchanged zeolites than argon molecules. The selectivity of N2/O2 of cadmium-exchanged zeolite X is better than only sodium containing zeolite-X. Heats of adsorption and adsorption isotherms were also calculated using GCMC simulation algorithm. Simulation studies expectedly show the proximity of nitrogen molecules to the locations of extra framework sodium and cadmium cations.








Similar content being viewed by others
References
R.T. Yang, Adsorbents: Fundamentals and Applications (Wiley-Interscience, New York, 2003)
W. Breck, Zeolites Molecular Sieves: Structure, Chemistry and Use (Wiley-Interscience, New York, 1974)
R.V. Jasra, N.V. Choudary, S.G.T. Bhat, Sep. Sci. Technol. 26, 885–930 (1991)
R.T. Yang, Gas Separation by Adsorption Processes (Imperial College Press, London, 1997)
D.M. Ruthven, S. Farooq, K.S. Knaebel, Pressure Swing Adsorption (Wiley-VCH, New York, 1994)
R.V. Jasra, N.V. Choudary, S.G.T. Bhat, Ind. Eng. Chem. Res. 35, 4221–4229 (1996)
C.C. Chao, U.S. Patent 4859217 (1989)
C.C. Chao, J.D. Sherman, J.T. Mullhaupt, C.M. Bolinger, U.S. Patent 5174979 (1992)
C.G. Coe, J.F. Kirner, R. Pierantozzi, T.R. White, U.S. Patent 5152813 (1992)
S.U. Rege, R.T. Yang, Ind. Eng. Chem. Res. 36, 5358–5365 (1997)
K.P. Prasanth, R.S. Pillai, H.C. Bajaj, R.V. Jasra, H.D. Chung, T.H. Kim, S.D. Song, Int. J. Hydrogen Energy 33, 735 (2008)
J. Sebastian, R.V. Jasra, Chem. Commun. 26, 8–269 (2003)
J. Sebastian, R.V. Jasra, U.S. Patent 6572838, PCT Patent WO 03/080236 A1, UK Patent 2,386,889 A (2003)
N. Chen, R.T. Yang, Ind. Eng. Chem. Res. 35, 4020–4027 (1996)
N.D. Hutson, D.A. Reisner, R.T. Yang, B.H. Toby, Chem. Mater. 12, 3020–3031 (2000)
J. Sebastian, S.A. Peter, R.V. Jasra, Langmuir 21, 11220–11225 (2005)
J. Sebastian, R.S. Pillai, S.A. Peter, R.V. Jasra, Ind. Eng. Chem. Res. 46, 6293–6302 (2007)
R.V. Jasra, C.D. Chudasama, U.S. Patent 0090380 A1 (2005)
A. Jayaraman, R.T. Yang, S.G.T. Bhat, N.V. Choudary, Adsorption 8, 271–278 (2002)
A.H. Fuchs, A.K. Cheetham, J. Phys. Chem. B 105, 7375 (2001)
D.M. Razmus, C.K. Hall, AIChE J. 37, 769–779 (1991)
K. Watanabe, N. Austin, M.R. Stapleton, Mol. Simul. 15, 197–221 (1995)
N.D. Hutson, S.C. Zajic, R.T. Yang, Ind. Eng. Chem. Res. 39, 1775–1780 (2000)
A.J. Richards, K. Watanabe, N. Austin, M.R. Stapleton, J. Porous Mat. 2, 43–49 (1995)
R.S. Pillai, S.A. Peter, R.V. Jasra, Langmuir 23, 8899–8908 (2007)
K.S.W. Sing, in Characterization of Powder Surfaces, ed. by G.D. Parfitt, K.S.W. Sing (Academic Press, London, 1976)
L. Zhu, K. Seff, J. Phys. Chem. B 103, 9512–9518 (1999)
J.H. Kwon, S.B. Jang, Y. Kim, K. Seff, J. Phys. Chem. 100, 13720–13724 (1996)
E. de Burchart, Studies on Zeolites: Molecular Mechanics, Framework Stability and Crystal Growth, Ph.D. Thesis Table I, Chapter XII (1992)
Cerius2 User Guide, Forcefield-Based Simulations (Molecular Simulations Inc., San Diego, 1997)
S. Murad, K.E. Gubbins, in Computer Modeling of Matter, ed. by P. Lykos. ACS Symposium Series 86 (American Chemical Society, Washington DC, 1978), p. 62
M.P. Allen, D.J. Tildesley, Computer Simulation of Liquids (Clarendon, Oxford, 1987)
G.J. Kramer, N.P. Farragher, B.W.H. van Beeset, R.A. van Santen, Phys. Rev. B 43, 5068 (1991)
G. Maurin, P. Llewellyn, T. Poyet, B. Kuchta, J. Phys. Chem. B 109, 125 (2005)
A.K. Rappe, C.J. Casewit, K.S. Colwell, W.A. Goddard III, W.M. Skiff, J. Am. Chem. Soc. 114, 10024 (1992)
I.A.M. Ahmed, S.D. Young, J.F.W. Mosselmans, N.M.J. Crout, E.H. Bailey, Geochim. Cosmochim. Acta 73, 1577–1587 (2009)
D.R. Lide, CRC Hand Book of Chemistry and Physics, 90th edn. (The Chemical Rubber Co., Cleveland, 2009)
Acknowledgments
We greatly admire the financial assistance and support from Council of Scientific and Industrial Research (CSIR), New Delhi. R. S. P. thanks to CSIR, New Delhi, India for financial assistance in the form of senior research fellowship. We also thank to Dr. Alexandra Simperler and Dr. James Wescott, Accerlys Inc. E. U.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pillai, R.S., Sebastian, J. & Jasra, R.V. Sorption of nitrogen, oxygen, and argon in Cd (II) exchanged zeolite X: volumetric equilibrium adsorption and grand canonical Monte Carlo study. J Porous Mater 18, 113–124 (2011). https://doi.org/10.1007/s10934-010-9362-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-010-9362-8