Abstract
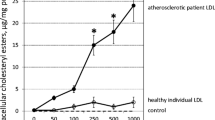
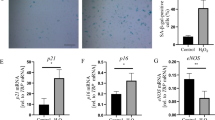
Cytosolic estrogen sulfotransferase (SULT1E1) mainly catalyzes the sulfoconjugation and deactivation of estrogens that are known to exert potent anti-atherogenic effects. However, it remains unknown about the connection between SULT1E1 and atherosclerosis. Recently, we reported that SULT1E1 is highly expressed in the aorta with plaques of high fat-fed ApoE knockout (KO) mice (mouse model of atherosclerosis), and interacts with oxidized low-density lipoprotein (Ox-LDL) known as a major component of atherosclerotic lesions. In this study, immunohistochemical staining for SULT1E1 in the aorta of high fat-fed ApoE KO mice showed that SULT1E1 is detected in vascular endothelial cells overlying atherosclerotic plaques. Results from Western blotting showed that Ox-LDL induces the protein expression of both SULT1E1 and peroxisome proliferator-activated receptor (PPAR) γ in human umbilical vein endothelial cells (HUVECs), and then that a PPARγ antagonist GW9662, but not a PPARα antagonist GW6471, inhibited the protein expression of SULT1E1 induced by Ox-LDL. Moreover, GW9662 significantly increased the proliferation of HUVECs induced by Ox-LDL. Our results suggest that SULT1E1 and PPARγ, both of which are increased by Ox-LDL, may interact with each other, and then may reduce cooperatively Ox-LDL-induced proliferation of vascular endothelial cells overlying atherosclerotic plaques, leading to against atherosclerosis.







Similar content being viewed by others
References
Glass CK, Witztum JL (2001) Atherosclerosis: the road ahead. Cell 104:503–516. https://doi.org/10.1016/S0092-8674(01)00238-0
Libby P, Ridker PM, Hansson GK (2011) Progress and challenges in translating the biology of atherosclerosis. Nature 47:317–325. https://doi.org/10.1038/nature10146
Ross R (1999) Mechanism of disease: atherosclerosis—an inflammatory disease. N Engl Med 340:115–126. https://doi.org/10.1056/NEJM199901143400207
Sternberg D, Parthasarathy S, Carew TE, Khoo JC, Witztum JL (1989) Beyond cholesterol: modification of low-density lipoprotein that increases its atherogenicity. N Engl J Med 320:915–924. https://doi.org/10.1056/NEJM198904063201407
Rosenfeld ME (1991) Oxidized LDL affects multiple atherogenic cellular responses. Circulation 83:2137–2140. https://doi.org/10.1161/01.cir.83.6.2137
Witztum JL (1993) Role of oxidised low-density lipoprotein in atherogenesis. Br Heart J 69:S12–S18. https://doi.org/10.1136/hrt.69.1_suppl.s12
Nishi K, Itabe H, Uno M, Kitazato KT, Horiguchi H, Shinno K, Nagahiro S (2002) Oxidized LDL in carotid plaques and plasma associates with plaque instability. Arterioscler Thromb Vasc Biol 22:1649–1654. https://doi.org/10.1161/01.atv.0000033829.14012.18
Sigala F, Kotsinas A, Savari P, Fillis K, Markantonis S, Iliodromitis EK, Gorgoulis VG, Andreadou I (2010) Oxidized LDL in human carotid plaques is related to symptomatic carotid disease and lesion instability. J Vasc Surg 52:704–713. https://doi.org/10.1016/j.jvs.2010.03.047
Falany C (1997) Enzymology of human cytosolic sulfotransferase. FASEB J 11:206–216. https://doi.org/10.1096/fasebj.11.4.9068609
Nagata K, Yamazoe Y (2000) Pharmacogenetics of sulfotransferase. Annu Rev Pharmacol Toxicol 40:159–176. https://doi.org/10.1146/annurev.pharmtox.40.1.159
Lindsay J, Wang LL, Li Y, Zhou SF (2008) Structure, function and polymorphism of human cytosolic sulfotransferases. Curr Drug Metab 9:99–105. https://doi.org/10.2174/138920008783571819
Alnouti Y, Klaassen CD (2006) Tissue distribution and ontogeny of sulfotransferase enzymes in mice. Toxicol Sci 93:242–255. https://doi.org/10.1093/toxsci/kfl050
Miki Y, Nakata T, Suzuki T, Darnel AD, Moriya T, Kaneko C, Hidaka K, Shiotsu Y, Kusaka H, Sasano H (2002) Systemic distribution of steroid sulfatase and estrogen sulfotransferase in human adult and fetal tissues. J Clin Endocrinol Metab 87:5760–5768. https://doi.org/10.1210/jc.2002-020670
Song WC (2001) Biochemistry and reproductive endocrinology of estrogen sulfotransferase. Ann N Y Acad Sci 948:43–50. https://doi.org/10.1111/j.1749-6632.2001.tb03985.x
Zhang H, Varmalova O, Vargas FM, Falany CN, Leyh TS (1998) Surfuryl transfer: the catalytic mechanism of human estrogen sulfotransferase. J Biol Chem 273:10888–10892. https://doi.org/10.1074/jbc.273.18.10888
Sato A, Yamazaki M, Watanabe H, Sakurai E, Ebina K (2020) Human estrogen sulfotransferase and its related fluorescently labeled decapeptides specifically interact with oxidized low-density lipoprotein. J Pept Sci 26:e3274. https://doi.org/10.1002/psc.3274
Sato A, Watanabe H, Yamazaki M, Sakurai E, Ebina K (2021) Interaction of native- and oxidized-low-density lipoprotein with human estrogen sulfotransferase. Protein J 40:1–13. https://doi.org/10.1007/s10930-021-09971-y
Nakamura Y, Miki Y, Suzuki T, Nakata T, Darnel D, Moriya T, Tazawa C, Saito H, Ishibashi T, Takahashi S, Yamada S, Sasano H (2003) Steroid sulfatase and estrogen sulfotransferase in the atherosclerotic human aorta. Am J Pathol 163:1329–1339. https://doi.org/10.1016/S0002-9440(10)63492-X
Li Y, Xu Y, Li X, Qin Y, Hu R (2013) Effects of PPAR-α agonist and IGF-1 on estrogen sulfotransferase in human vascular endothelial and smooth muscle cells. Mol Med Rep 8:133–139. https://doi.org/10.3892/mmr.2013.1483
Xu Y, Yang X, Wang Z, Li M, Ning Y, Chen S, Yin L, Li X (2013) Estrogen sulfotransferase (SULT1E1) regulates inflammatory response and lipid metabolism of human endothelial cells via PPARγ. Mol Cell Endocrinol 369:140–149. https://doi.org/10.1016/j.mce.2013.01.020
Havel RJ, Eder HA, Bragdon JH (1955) The distribution and chemical composition of ultracentrifugally separated lipoproteins in human serum. J Clin Investig 34:1345–1353. https://doi.org/10.1172/JCI103182
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Aboud OA, Wettersten HI, Weiss RH (2013) Inhibition of PPARα induces cell cycle arrest and apoptosis, and synergizes with glycolysis inhibition in kidneys cancer cells. PLoS ONE 8:e71115. https://doi.org/10.1371/journal.pone.0071115
Xu HE, Stanley TB, Montana VG, Lambert MH, Shearer BG, Cobb JE, McKee DD, Galardi CM, Plunket KD, Nolte RT, Parks DJ, Moore JT, Kliewer SA, Willson TM, Stimmel JB (2002) Structural basis for antagonist-mediated recruitment of nuclear co-repressors by PPARalpha. Nature 415:813–817. https://doi.org/10.1038/415813a
Leesnitzer LM, Parks DJ, Bledsoe RK, Cobb JE, Collins JL, Consler TG, Davis RG, Hull-Ryde EA, Lenhard JM, Patel L, Plunket KD, Shenk JL, Stimmel JB, Therapontos C, Willson TM, Blanchard SG (2002) Functional consequences of cysteine modification in the ligand binding sites of peroxisome proliferator activated receptors by GW9662. Biochemistry 41:6640–6650. https://doi.org/10.1021/bi0159581
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85. https://doi.org/10.1016/0003-2697(85)90442-7
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Hamblin M, Chang L, Fan Y, Zhang J, Chen YE (2009) PPARs and the cardiovasucular system. Antioxid Redox Signal 11:1415–1452. https://doi.org/10.1089/ars.2008.2280
Xu Y, Lin X, Xu J, Jing H, Qin Y, Li Y (2018) SULT1E1 inhibits cell proliferation and invasion by activating PPARγ in breast cancer. J Cancer 9:1078–1087. https://doi.org/10.7150/jca.23596
Barbosa ACS, Feng Y, Yu C, Huang M, Xie W (2019) Estrogen sulfotransfertase in the metabolism of estrogenic drugs and in the pathogenesis of diseases. Expert Opin Drug Metab Toxicol 15:329–339. https://doi.org/10.1080/17425255.2019.1588884
Mercer KE, Apostolov EO, Gamboa DCG, Yu X, Lang P, Roberts DW, Davis W, Basnakian AG, Kadlubar FF, Kadlubar SA (2010) Expression of sulfotransferase isoform 1A1 (SULT1A1) in breast cancer cells significantly increases 4-hydroxytamoxifen-induced apoptosis. Int J Mol Epidemiol Genet 1:92–103
Pasqualini JR (2009) Estrogen sulfotransferases in breast and endometrial cancers. Ann N Y Acad Sci 1155:88–98. https://doi.org/10.1111/j.1749-6632.2009.04113.x
Khor VK, Dhir R, Yin X, Ahima RS, Song WC (2010) Estrogen sulfotransferase regulates body fat and glucose homeostasis. Am J Physiol Endocrinol Metab 299:E657–E664. https://doi.org/10.1152/ajpendo.00707.2009
Wada T, Ihunnah CA, Gao J, Chai X, Zeng S, Philips BJ, Rubin JP, Marra KG, Xie W (2011) Estrogen sulfotransferase inhibits adipocyte differentiation. Mol Endocrinol 25:1612–1623. https://doi.org/10.1210/me.2011-1089
Akune T, Ohba S, Kamekura S, Yamaguchi M, Chung U, Kubota N, Terauchi Y, Harada Y, Azuma Y, Nakamura K, Kadowaki T, Kawaguchi H (2004) PPARγ insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J Clin Investig 113:846–855. https://doi.org/10.1172/JCI19900
Reddy AT, Lakshmi SP, Reddy RC (2016) PPARγ as a novel therapeutic target in lung cancer. PPAR Res 2016:8972570. https://doi.org/10.1155/2016/8972570
Wang G, Cao R, Quan G, Dan HC, Jiang W, Ju L, Wu M, Xiao Y, Wang X (2016) Simvastatin induces cell cycle arrest and inhibits proliferation of bladder cancer cells via PPARγ signalling pathway. Sci Rep 6:35783. https://doi.org/10.1038/srep35783
Bonofiglio D, Gabriele S, Aquila S, Qi H, Belmonte M, Catalano S, Andò S (2009) Peroxisome proliferator-activated receptor gamma activates fas ligand gene promoter inducing apoptosis in human breast cancer cells. Breast Cancer Res Treat 113:423–434. https://doi.org/10.1007/s10549-008-9944-1
Catalano S, Mauro L, Bonofiglio D, Pellegrino M, Qi H, Rizza P, Vizza D, Bossi G, Andò S (2011) In vivo and in vitro evidence that PPARγ ligands are antagonists of leptin signaling in breast cancer. Am J Pathol 179:1030–1040. https://doi.org/10.1016/j.ajpath.2011.04.026
Zheng J, Lu C (2020) Oxidized LDL causes endothelial apoptosis by inhibiting mitochondria autophagy. Front Cell Dev Biol 8:600950. https://doi.org/10.3389/fcell.2020.600950
Galle J, Heinloth A, Wanner C, Heermeier K (2001) Dual effect of oxidized LDL on cell cycle in human endothelial cells through oxidative stress. Kidney Int Suppl 78:S120-123. https://doi.org/10.1046/j.1523-1755.2001.59780120.x
Delimaris I, Faviou E, Antonakos G, Stathopoulou E, Zachari A, Dionyssiou-Asteriou A (2007) Oxidized LDL, serum oxidizability and serum lipid levels in patients with breast or ovarian cancer. Clin Biochem 40:1129–1134. https://doi.org/10.1016/j.clinbiochem.2007.06.007
Nakhjavani M, Khalilzadeh O, Khajeali L, Esteghamati A, Morteza A, Jamali A, Dadkhahipour S (2010) Serum oxidized-LDL is associated with diabetes duration independent of maintaining optimized levels of LDL-cholesterol. Lipids 45:321–327. https://doi.org/10.1007/s11745-010-3401-8
Prescott SM, Zimmerman GA, McIntyre TM (1990) Platelet-activating factor. J Biol Chem 265:17381–17384
Shindou H, Shimizu T (2009) Acyl-CoA: Lysophospholipid acyltransferases. J Biol Chem 284:1–5. https://doi.org/10.1074/jbc.R800046200
Nigam S, Benedetto SM (1989) Elevated plasma levels of platelet-activating factor (PAF) in breast cancer patients with hypercalcemia. J Lipid Mediat 1:323–328
Ayala A, Chaudry IH (1996) Platelet activating factor and its role in trauma, shock, and sepsis. New Horiz 4:265–275
Fang X, Gaudette D, Furui T, Mao M, Estrella V, Eder A, Pustilnik T, Sasagawa T, Lapushin R, Yu S, Jaffe RB, Wiener JR, Erickson JR, Mills GB (2000) Lysophospholipid growth factors in the initiation, progression, metastases, and management of ovarian cancer. Ann N Y Acad Sci 905:188–208. https://doi.org/10.1111/j.1749-6632.2000.tb06550.x
Runge-Morris M, Kocarek TA, Falany CN (2013) Regulation of the cytosolic sulfotransferases by nuclear receptors. Drug Metab Rev 45:15–33. https://doi.org/10.3109/03602532.2012.748794
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-profit sectors. We thank Tokushima Molecular Pathology Institute, Inc (Tokushima, Japan) for forming tissue slice by microtome. We thank Editage by Cactas Communications Inc. (Tokyo, Japan) for providing assistance with the English language.
Author information
Authors and Affiliations
Contributions
AS, ES, and KE conceived and designed the experiments. AS, HW, and MY performed the experiments and analyzed the data. AS wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sato, A., Watanabe, H., Yamazaki, M. et al. Estrogen Sulfotransferase is Highly Expressed in Vascular Endothelial Cells Overlying Atherosclerotic Plaques. Protein J 41, 179–188 (2022). https://doi.org/10.1007/s10930-022-10042-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-022-10042-z