Abstract
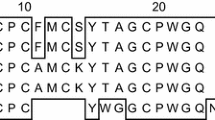
Sea anemones synthesize a variety of toxic peptides and proteins of biological interest. The Caribbean Sea anemone Stichodactyla helianthus, produces two pore-forming toxins, Sticholysin I (St I) and Stichloysin II (St II), with the ability to form oligomeric pores in cell and lipid bilayers characteristically lacking cysteine in their amino acid sequences. Recently, two mutants of a recombinant variant of Sticholysin I (rSt I) have been obtained with a Cys residue in functionally relevant regions for the pore-forming activity of the toxin: r St I F15C (in the amino terminal sequence) and r St I R52C (in the binding site). Aiming at characterizing the effects of oxidants in toxins devoid (r St I) or containing –SH moieties (r St I F15C and r St I R52C), we measured their hemolytic activity and pore forming capacity prior and after their incubation with peroxynitrite (ONOO−). At low ONOO−/Toxin ratios, nearly 0.8 Trp groups are modified by each added peroxynitrite molecule, and the toxin activity is reduced in ca. 20 %. On the other hand, in –SH bearing mutants only 0.5 Trp groups are modified by each peroxynitrite molecule and the toxin activity is only reduced in 10 %. The results indicated that Cys is the initial target of the oxidative damage and that Trp residues in Cys-containing toxins were less damaged than those in r St I. This relative protection of Trp groups correlates with a smaller loss of hemolytic activity and permeabilization ability in liposomes and emphasizes the relevance of Trp groups in the pore forming capacity of the toxins.









Similar content being viewed by others
Abbreviations
- DPPC:
-
1,2-Palmitoyl-sn-glycero-3-phosphatidylcholine
- SM:
-
Sphingomyelin
- DTNB:
-
5,5′-Dithiobis-(2-nitrobenzoic acid)
- HA:
-
Hemolytic activity
- H:
-
Hemolysis percentage
- E:
-
Permeation percentage
- RBC:
-
Red blood cells
- t20Hs:
-
Time required to produce 20 % hemolysis for non-treated toxins
- t20Hm:
-
Time required to produce 20 % hemolysis for treated toxins with peroxynitrite
- t20Es:
-
Time required to produce 20 % of liposomes with pores for non-treated toxins
- t20Em:
-
Time required to produce 20 % of liposomes with pores for treated toxins with peroxynitrite
References
Harvey HL (1990) In: Shier WT, Mebs D (eds) Handbook of toxinology. Marcel Dekker, New York, pp 1–66
Gutiérrez-Aguirre I, Barli A, Podlesek Z, Maček P, Anderluh G, González-Mañas JM (2004) Membrane insertion of the N-terminal α-helix of equinatoxin II, a sea anemone cytolytic toxin. Biochem J 384:421–428
Tejuca M, Dalla Serra M, Ferrreras M, Lanio ME, Menestrina G (1996) Mechanism of membrane permeabilization by Sticholysin I, a cytolysin isolated from the venom of the sea anemone Stichodactyla helianthus. Biochemistry 35:14947–14957
Kristan K, Podlesek Z, Hojnik V, Gutierrez-Aguirre I, Guncar G, Turk D, Gonzalez-Manas JM, Lakey JH, Macek P, Anderluh G (2004) Pore formation by equinatoxin, a eukaryotic pore-forming toxin, requires a flexible N-terminal region and a stable beta-sandwich. J Biol Chem 279:46509–46517
Mancheño JM, Martín-Benito J, Martínez-Ripoll M, Gavilanes JG, Hermoso JA (2003) Crystal and electron microscopy structures of Sticholysin II actinoporin reveal insights into the mechanism of membrane pore formation. Structure 11:1319–1328
Hong Q, Gutiérrez-Aguirre I, Barlič A, Malovrh P, Kristan K, Podlesek Z, Maček P, Turk D, González-Mañas JM, Lakey JH, Anderluh G (2002) Two-stepmembrane binding by Equinatoxin II, a pore-forming toxin from the sea anemone, involves an exposed aromatic cluster and a flexible helix. J Biol Chem 277:41916–41924
García-Linares S, Castrillo I, Bruix M, Menendez M, Alegre-Cebollada J, Martínez-del-Pozo A, Gavilanes J (2013) Three-dimensional structure of the actinoporin sticholysin I. Influence of long-distance effects on protein function. Arch Biochem Biophys 532:39–45
Valle AE, López-Castilla A, Pedrera L, Martínez D, Tejuca M, Campos J, Fando L, Lissi E, Álvarez C, Lanio ME, Pazos F, Schreier S (2011) Cys mutants in functional regions of Sticholysin I clarify the participation of these residues in pores formation. Toxicon 58:8–17
Groves JT (1999) Peroxynitrite: reactive, invasive and enigmatic. Curr Opin Chem Biol 3:226–235
Koppenol WH, Moreno JJ, Pryor WA (1992) Peroxynitrite, a cloaked oxidant formed by nitric oxide and superoxide. Chem Res Toxicol 5:834–842
Pryor WA, Squadrito GL (1995) The chemistry of peroxynitrite: a product from the reaction of nitric oxide with superoxide. Am J Phys 268:L699–L722
Rugeles MT, Patiño PJ (2009) Inmunología. Una ciencia activa.2.ª edición, Ed universidad de Antioquina
Alvarez B, Radi R (2003) Peroxynitrite reactivity with amino acids and proteins. Amino Acids 25:295–311
Castro L, Rodriguez M, Radi R (1994) Aconitase is readily inactivated by peroxynitrite, but not by its precursor, nitric oxide. J Biol Chem 269:29409–29415
Pucciarelli S, Spina M, Montequia F, Lupidi G, Eleuteri AM, Fioretti E, Angeletti M (2005) Peroxynitrite-mediated oxidation of the C85S/C152E mutant of dihydrofolate reductase from Escherichia coli: functional and structural effects. Arch Biochem Biophys 434:221–231
Van der Vliet A, Halliwell B, Cross CE, Kaur H (1994) Aromatic hydroxylation and nitration of phenylalanine and tyrosine by peroxynitrite: evidence for hydroxyl radical production from peroxynitrite. FEBS Lett 339:89–92
Kem WR, Dunn BM (1988) Separation and characterization of four different aminoacids sequence variants of a sea anemone (Stichodactyla heliantus) protein cytolysin. Toxicon 26:997–1008
Alvarez C, Lanio ME, Tejuca M, Pazos F, Acevedo AM, Campos AM, Encinas MV, Pertinhez T, Schreier S, Lissi EA (1998) The role of the ionic strength in the enhancement of the hemolytic activity of sticholysin I, a cytolysin from Stichodactyla heliantus. Toxicon 36:165–178
Celedon G, Venegas F, Campos AM, Lanio ME, Martinez D, Soto C, Alvarez C, Lissi E (2005) Role of endogenous channels in red blood cells response to their exposure to the pore forming toxin Sticholysin II. Toxicon 46:297–307
Celedón G, Gonzales G, Barrientos D, Pino J, Venegas F, Lissi E, Soto C, Martínez D, Alvarez C, Lanio ME (2008) Stycholysin II, a cytolysin from the sea anemone Stichodactyla helianthus promotes higher hemolysis in aged red blood cells. Toxicon 51:1383–1390
Mayer LD, Hope MJ, Cullis PR (1986) Vesicles of variable sizes produced by a rapid extrusion procedure. Biochim Biophys Acta 858:161–168
Pazos F, Martínez D, Tejuca M, Valle A, del Pozo A, Álvarez C, Lanio ME, Lissi E (2003) Comparison of pore-forming ability in membranes of a native and a recombinant variant of Sticholysin II from Stichodactyla helianthus. Toxicon 42:571–578
Martínez D, Otero A, Alvarez C, Pazos F, Tejuca M, Lanio ME, Gutiérrez-Aguirre I, Barlič A, Iloro I, Rodríguez JL, González-Mañas JM, Lissi E (2007) Effect of sphingomyelin and cholesterol in the interaction of St II with lipidic interfaces. Toxicon 49:68–81
Beckman JS, Beckman TW, Chen J, Marshall PA, Freeman BA (1990) Apparent hydroxyl radical production by peroxynitrite: implication for endothelial injury from nitric oxide and superoxide. Proc Natl Acad Sci USA 87:1620–1624
Lanio ME, Morera V, Alvarez C, Tejuca M, Gomez T, Pazos F (2001) Purification and characterization of two hemolysins from Stichodactyla helianthus. Toxicon 39:187–194
Lacowickz JR (2006) Principles of fluorescence spectroscopy, 3rd edn. Springer, Singapore
Tibi S, Koppenol WH (2000) Reactions of peroxynitrite with phenolic and carbonyl compounds: flavonoids are not scavengers of peroxynitrite. Helv Chim Acta 83:2412–2424
Campos AM, Lissi E, Vergara C, Lanio ME, Alvarez C, Pazos I, Morera V, García Y, Martinez D (1999) Kinetics and mechanism of the St I modification by peroxyl radicals. Int J Protein Chem 18:297–305
Pazos IF, Alvarez C, Lanio ME, Martinez D, Morera V, Lissi E, Campos AM (1998) Modification of sticholysin II hemolytic activity by free radicals. Toxicon 36:1383–1393
Martinez D, Campos AM, Pazos F, Alvarez C, Lanio ME, Casallanovo F, Schreier S, Salinas RK, Vergara C, Lissi E (2001) Properties of St I and St II, two isotoxins isolated from Stichodactyla helianthus: a comparison. Toxicon 39:1547–1560
Carballal S, Alvarez B, Turell L, Botti H, Freeman BA, Radi R (2007) Sulfenic acid in human serum albumin. Amino Acids 32:543–551
Acknowledgments
Thanks are given to FONDECYT (Project 10702859). LL also acknowledges a doctoral Fellowship. This work was supported by the Iberian American Network CYTED 212RT0467 Biotox.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
León, L., Lissi, E.A., Celedón, G. et al. Inactivation of the Pore-Forming Toxin Sticholysin I by Peroxynitrite: Protection by Cys Groups Incorporated in the Toxin. Protein J 33, 493–501 (2014). https://doi.org/10.1007/s10930-014-9582-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-014-9582-x