Abstract
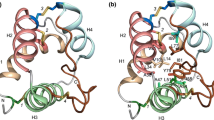
Plant non-specific lipid transfer proteins (ns-LTPs) comprise two families, LTP1s and LTP2s, all structurally stabilized by four native disulfide bonds. Solution and crystal structures of both LTP1s and LTP2s from various plants have been determined. Despite the similarities of their biological function and backbone folds, the biophysical properties of LTP1s and LTP2s differ significantly. In this report, the mechanisms of unfolding and refolding of rice LTP1 and LTP2 have been investigated using the technique of disulfide bonds scrambling. LTP1 is shown to unfold and refold via predominant species of partially structured intermediates. Four isomers of partly unfolded and extensively unfolded LTP1 were identified, isolated and their disulfide structures were determined. By contrast, unfolding and refolding of LTP2 adopt a (close to) two-state mechanism, and undergo a reversible conversion between the native and a single extensively unfolded isomer without accumulation of any significant intermediate.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lin, CH., Li, L., Lyu, PC. et al. Distinct Unfolding and Refolding Pathways of Lipid Transfer Proteins LTP1 and LTP2. Protein J 23, 553–566 (2004). https://doi.org/10.1007/s10930-004-7882-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10930-004-7882-2