Abstract
Target-mediated drug disposition (TMDD) describes drug binding with high affinity to a target such as a receptor. In application TMDD models are often over-parameterized and quasi-equilibrium (QE) or quasi-steady state (QSS) approximations are essential to reduce the number of parameters. However, implementation of such approximations becomes difficult for TMDD models with drug–drug interaction (DDI) mechanisms. Hence, alternative but equivalent formulations are necessary for QE or QSS approximations. To introduce and develop such formulations, the single drug case is reanalyzed. This work opens the route for straightforward implementation of QE or QSS approximations of DDI TMDD models. The manuscript is the first part to introduce DDI TMDD models with QE or QSS approximations.
Similar content being viewed by others
References
Levy G (1994) Pharmacologic target-mediated drug disposition. Clin Pharmacol Ther 56(3):248–252
Mager DE, Jusko WJ (2001) General pharmacokinetic model for drugs exhibiting target-mediated drug disposition. J Pharmacokinet Pharmacodyn 28(6):507–532
Mager DE, Krzyzanski W (2005) Quasi-equilibrium pharmacokinetic model for drugs exhibiting target-mediated drug disposition. Pharm Res 22(10):1589–1596
Dua P, Hawkins E, van der Graaf PH (2015) A tutorial on target-mediated drug disposition (TMDD) models. CPT Pharmacomet Syst Pharmacol 4(6):324–337
An G (2016) Small-molecule compounds exhibiting target-mediated drug disposition (TMMD): a minireview. J Clin Pharmacol. doi:10.1002/jcph.804
Gibiansky L, Gibiansky E (2010) Target-mediated drug disposition model for drugs that bind to more than one target. J Pharmacokinet Pharmacodyn 37(4):323–346
Gibiansky L, Gibiansky E (2014) Target-mediated drug disposition model and its approximations for antibody-drug conjugates. J Pharmacokinet Pharmacodyn 41(1):35–47
Krippendorff BF, Kuester K, Kloft C, Huisinga W (2009) Nonlinear pharmacokinetics of therapeutic proteins resulting from receptor mediated endocytosis. J Pharmacokinet Pharmacodyn 36(3):239–260
Cao Y, Jusko WJ (2014) Incorporating target-mediated drug disposition in a minimal physiologically-based pharmacokinetic model for monoclonal antibodies. J Pharmacokinet Pharmacodyn 41(4):375–387
Chen X, Jiang X, Jusko WJ, Zhou H, Wang W (2016) Minimal physiologically-based pharmacokinetic (mPBPK) model for a monoclonal antibody against interleukin-6 in mice with collagen-induced arthritis. J Pharmacokinet Pharmacodyn 43:291–304
Gibiansky L, Gibiansky E, Kakkar T, Ma P (2008) Approximations of the target-mediated drug disposition model and identifiability of model parameters. J Pharmacokinet Pharmacodyn 35(5):573–591
Peletier LA, Gabrielsson J (2012) Dynamics of target-mediated drug disposition: characteristic profiles and parameter identification. J Pharmacokinet Pharmacodyn 39(5):429–451
Peletier LA, Gabrielsson J (2013) Dynamics of target-mediated drug disposition: how a drug reaches its target. Comput Geosci 17:599–608
Marathe A, Krzyzanski W, Mager DE (2009) Numerical validation and properties of a rapid binding approximation of a target-mediated drug disposition pharmacokinetic model. J Pharmacokinet Pharmacodyn 36(3):199–219
Ma P (2012) Theoretical considerations of target-mediated drug disposition models: simplifications and approximations. Pharm Res 29(3):866–882
Patsatzis DG, Maris DT, Goussis DA (2016) Asymptotic analysis of a target-mediated drug disposition model: algorithmic and traditional approaches. Bull Math Biol 78(6):1121–1161
Yan X, Chen Y, Krzyzanski W (2012) Methods of solving rapid binding target-mediated drug disposition model for two drugs competing for the same receptor. J Pharmacokinet Pharmacodyn 39(5):543–560
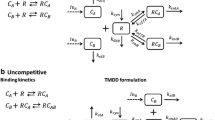
Koch G, Jusko WJ, Schropp J (2017) Target mediated drug disposition with drug-drug interaction, Part II: competitive and uncompetitive cases. J Pharmacokinet Pharmacodyn. doi:10.1007/s10928-016-9502-0
Fenichel N (1979) Geometric singular perturbation theory for ordinary differential equations. J Diff Equ 31:54–98
Vasileva AB (1963) Asymptotic behaviour of solutions to certain problems involving nonlinear differential equations containing a small parameter multiplying the highest derivatives. Russ Math Surv 18:13–83
Brenan KE, Campbell SL, Petzold LR (1996) Numerical solution of initial value problems in differential-algebraic equations. Classics in Applied Mathematics 14 SIAM
D’Argenio DZ, Schumitzky A, Wang X (2009) ADAPT 5 user’s guide: pharmacokinetic/pharmacodynamic systems analysis software. Biomedical Simulations Resource, Los Angeles
Beal S, Sheiner LB, Boeckmann A, Bauer RJ (2009) NONMEM user’s guides. Icon Development Solutions, Ellicott City
R Core Team (2014) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Release MATLAB (2014b) The MathWorks. MathWorks Inc., Natick
Hairer E, Wanner G (1991) Solving ordinary differential equations II. Stiff and differential-algebraic problems, 2nd edn in 1996., Springer Series in Computational Mathematics 14. Springer, New York
Goussis DA, Valorani M (2006) An efficient iterative algorithm for the approximation of the fast and slow dynamics of stiff systems. J Comput Phys 214(1):316–46
Kourdis PD, Goussis DA (2013) Glycolysis in saccharomyces cerevisiae: algorithmic exploration of robustness and origin of oscillation. Math Biosci 243(2):190–214
Acknowledgements
This work was supported in part by NIH Grant GM24211.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendices
Appendix 1: Short infusion and IV bolus
By construction and the use of Eqs. (12, 43–44) the final system (46–51) is equivalent to
We assume that the short infusion with infusion rate \(dose/(\varepsilon V)\) is given for \(t \in [t_a, t_a + \varepsilon ]\). We then obtain by integrating Eqs. (55–56) over \([t_a, t_a + \varepsilon ]\)
which leads to
Now letting \(\varepsilon \rightarrow 0\) in Eqs. (58–59) and using Eq. (57) we get
This means that \(\lim _{\varepsilon \rightarrow 0} C(t_a + \varepsilon )\), \(\lim _{\varepsilon \rightarrow 0} R(t_a + \varepsilon )\) satisfy Eqs. (53–54) and
is shown.
Appendix 2: Source codes
The matrix representation applied in Eqs. (46–50) is of the general form
Hence, performing matrix multiplication the right hand side of the differential equation reads
To implement the matrix multiplication in Eq. (46), we apply Eqs. (60)-(61) where \(H_1\),\(H_2\) correspond to DADT(1), DADT(2) in NONMEM (lines 125-126) and XP(1), XP(2) in ADAPT 5 (lines 224–225).
The lines of the code are numbered for referencing but are not part of the code implementation.
NONMEM control stream for single drug TMDD with infusion coded in the control stream
The $PK and $DES blocks of the control stream are presented. Additionally, the first lines of the data file is shown to present the IV infusion mechanism. Full control stream is available in the supplemental material.
100: $PK
101: KEL = THETA(1)*EXP(ETA(1))
102: KD = THETA(2)*EXP(ETA(2))
103: KINT = THETA(3)*EXP(ETA(3))
104: KSYN = THETA(4)*EXP(ETA(4))
105: KDEG = THETA(5)*EXP(ETA(5))
106: V = THETA(6)*EXP(ETA(6))
107: A_0(1) = 0
108: A_0(2) = KSYN/KDEG
109: $DES
110: EPSILON = 1e-4
111: ; Dose at T1 = 0
112: IN = 0
113: IF (T.GE.0.AND.T.LE.0+EPSILON) THEN
114: IN = 100*EPSILON**(-1)
115: ENDIF
116: C = A(1)/V
117: R = A(2)
118: DET = KD+R+C
119: G1 = IN - KEL*C - (KINT*C*R)/KD
120: G2 = KSYN - KDEG*R - (KINT*C*R)/KD
121: M11 = (1/DET)*(KD+C)
122: M12 = (1/DET)*(-C)
123: M21 = (1/DET)*(-R)
124: M22 = (1/DET)*(KD+R)
125: DADT(1) = M11*G1 + M12*G2
126: DADT(2) = M21*G1 + M22*G2
The first lines of the data file are:
#ID,TIME,DV,MDV
127: 1,0,.,1
128: 1,0.0001,.,1
129: 1,1,77.8812,0
ADAPT 5 source code for single drug TMDD
The subroutine DIFFEQ is presented. For full source code see supplemental material.
201: Subroutine DIFFEQ(T,X,XP)
202: Implicit None
203: Include ’globals.inc’
204: Include ’model.inc’
205: Real*8 T,X(MaxNDE),XP(MaxNDE)
206: Real*8 kel,KD,kint,ksyn,kdeg
207: Real*8 C,RR,RR0
208: Real*8 Det,M(2,2),g(2)
209: kel = P(1)
210: KD = P(2)
211: kint = P(3)
212: ksyn = P(4)
213: kdeg = P(5)
214: RR0 = ksyn/kdeg
215: C = X(1)
216: RR = X(2) + RR0
217: Det = RR + C + KD
218: g(1) = R(1) - kel*C - (kint*C*RR)/KD
219: g(2) = ksyn - kdeg*RR - (kint*C*RR)/KD
220: M(1,1) = (1/Det)*(KD+C)
221: M(1,2) = (1/Det)*(-C)
222: M(2,1) = (1/Det)*(-RR)
223: M(2,2) = (1/Det)*(KD+RR)
224: XP(1) = M(1,1)*g(1) + M(1,2)*g(2)
225: XP(2) = M(2,1)*g(1) + M(2,2)*g(2)
226: Return
227: End
Free concentration C(t) from the original TMDD model Eqs. (1–7) (dashed line) and the QE or QSS approximation Eqs. (46–50) (solid line) is shown. In panel a the QE and in panel b the QSS approximation without baseline for escalating doses (dose = 10, 100 and 1000) are presented. In panel c (QE) and in panel d (QSS) behavior for different baselines (\(C^0\) = 0.01, 0.1, 1 and 10) with a fixed dose (dose = 100) are visualized. In panels e (QE) and f (QSS) the effect for increasing \(k_{on} = 2.5\) and \(k_{off} = 0.1\) (by multiplying the factors 0.1, 1, 10 to both parameters with equal ratio \(K_D\) for QE) for a fixed dose (dose = 100) and baseline (\(C^0\) = 1) are presented. In panel e (QE) the original system converges to the QE approximation and in panel f (QSS) the original systems and its QSS approximations converge for increasing \(k_{on}\) and \(k_{off}\) values
Rights and permissions
About this article
Cite this article
Koch, G., Jusko, W.J. & Schropp, J. Target-mediated drug disposition with drug–drug interaction, Part I: single drug case in alternative formulations. J Pharmacokinet Pharmacodyn 44, 17–26 (2017). https://doi.org/10.1007/s10928-016-9501-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-016-9501-1