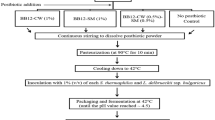
Abstract
Brewery yeast cell wall residues were used as a matrix for the development of bioactive films with refined liquid smoke extract. The extract and bioactive films were fully characterised in terms of their functional properties. Refined liquid smoke extract is a commercial additive used as a preservative agent in various food products. Minimum Inhibitory Concentration (MIC) values for foodborne pathogens of the refined liquid smoke extract ranged from 0.403 to 1.612 mg/mL. Its total phenolic content, as determined by the Folin-Ciocalteu method, was 450 mg/L, and its antioxidant capacity, as determined by IC50 (ABTS), was 593 mg/L. The functional properties of the bioactive films depended on the concentration of liquid smoke extract added. At low concentrations, the extract retarded the thermal degradation and improved the mechanical properties by increasing the tensile strength due to crosslinking between the carbonyl groups of the liquid smoke extract and the amino groups of the residual yeast cell wall film. However, high concentrations of extract functioned as a plasticiser and increased the hydration of the samples. The bioactive films containing 10% liquid smoke extract achieved the highest radical scavenging activity (RSA > 90%) among the tested samples. The findings indicated that incorporating liquid smoke extract into residual yeast cell wall films could represent a promising and innovative substitute for bioactive materials in the field of food packaging development.







Similar content being viewed by others
References
Ashrafi A, Jokar M, Nafchi AM (2018) Preparation and characterization of biocomposite film based on chitosan and kombucha tea as active food packaging. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2017.12.028
Mendiburu-Valor E, Larraza I, Echeverria-Altuna O, Harismendy I, Peña-Rodriguez C, Eceiza A (2023) Thermoset polyurethanes from biobased and recycled components. J Polym Environ. https://doi.org/10.1007/s10924-023-02891-1
Peltzer M, Delgado JF, Salvay AG, Wagner JR (2018) β-Glucan, a promising polysaccharide for bio-based films developments for food contact materials and medical applications. Curr Org Chem. https://doi.org/10.2174/1385272822666171129153633
Gomez-Caturla J, Tejada-Oliveros R, Ivorra-Martinez J, Garcia-Sanoguera D, Balart R, Garcia D (2023) Development and characterization of new environmentally friendly polylactide formulations with terpenoid-based plasticizers with improved ductility. J Polym Environ. https://doi.org/10.1007/s10924-023-03000-y
Bos HL, Meesters KP, Conijn SG, Corré WJ, Patel MK (2012) Accounting for the constrained availability of land: a comparison of bio-based ethanol, polyethylene, and PLA with regard to non-renewable energy use and land use. Biofuel Bioprod Biorefin. https://doi.org/10.1002/bbb.1320
Cottet C, Salvay AG, Peltzer MA, Fernández-García M (2021) Incorporation of poly (Itaconic acid) with quaternized thiazole groups on gelatin-based films for antimicrobial-active food packaging. Polym. https://doi.org/10.3390/polym13020200
Delgado JF, Sceni P, Peltzer MA, Salvay AG, de la Osa O, Wagner JR (2016) Development of innovative biodegradable films based on biomass of Saccharomyces cerevisiae. Food Sci Technol. https://doi.org/10.1016/j.ifset.2016.06.002
Cottet C, Ramirez-Tapias YA, Delgado JF, de la Osa O, Salvay AG, Peltzer MA (2020) Biobased materials from microbial biomass and its derivatives. Materials. https://doi.org/10.3390/ma13061263
Peltzer MA, Salvay AG, Delgado JF, de la Osa O, Wagner JR (2018) Use of residual yeast cell wall for new biobased materials production: Effect of plasticization on film properties. Food Bioproc Tech. https://doi.org/10.1007/s11947-018-2156-8
Rezzani GD, Choque E, Salvay AG, Mathieu F, Peltzer MA (2022) New antioxidant active packaging films based on yeast cell wall and naphtho-γ-pyrone extract. Polymers. https://doi.org/10.3390/polym14102066
Delgado JF, Peltzer MA, Salvay AG, de la Osa O, Wagner JR (2018) Characterization of thermal, mechanical and hydration properties of novel films based on saccharomyces cerevisiae biomass. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2018.06.017
Jamróz E, Kopel P, Tkaczewska J, Dordevic D, Jancikova S, Kulawik P, Adam V (2019) Nanocomposite furcellaran films—The influence of nanofillers on functional properties of furcellaran films and effect on linseed oil preservation. Polymers. https://doi.org/10.3390/polym11122046
Balbinot-Alfaro E, Craveiro DV, Lima KO, Costa HLG, Lopes DR, Prentice C (2019) Intelligent packaging with pH indicator potential. Food Eng Rev 11:235–244
Dirpan A, Latief R, Syarifuddin A, Rahman ANF, Putra RP, Hidayat SH (2018) The use of colour indicator as a smart packaging system for evaluating mangoes arummanis (Mangifera indica L. var. Arummanisa) freshness. IOP Conf Ser: Earth Environ Sci 157:012031
Montazeri N, Himelbloom BH, Oliveira AC, Leigh MB, Crapo CA (2013) Refined liquid smoke: a potential antilisterial additive to cold-smoked sockeye salmon (Oncorhynchus nerka). J Food Prot. https://doi.org/10.4315/0362-028x.jfp-12-368
Montazeri N, Oliveira AC, Himelbloom BH, Leigh MB, Crapo CA (2013) Chemical characterization of commercial liquid smoke products. Food Sci Nutr 1(1):102–115. https://doi.org/10.1002/2Ffsn3.9
Xin X, Bissett A, Wang J, Gan A, Dell K, Baroutian S (2021) Production of liquid smoke using fluidised-bed fast pyrolysis and its application to green lipped mussel meat. Food Control. https://doi.org/10.1016/j.foodcont.2021.107874
Soazo M, Pérez LM, Piccirilli GN, Delorenzi NJ, Verdini RA (2016) Antimicrobial and physicochemical characterization of whey protein concentrate edible films incorporated with liquid smoke. LWT- Food Sci Technol. https://doi.org/10.1016/j.lwt.2016.04.027
Lingbeck JM, Cordero P, O’Bryan CA, Johnson MG, Ricke SC, Crandall PG (2014) Functionality of liquid smoke as an all-natural antimicrobial in food preservation. Meat Sci. https://doi.org/10.1016/j.meatsci.2014.02.003
Toledo RT (2007) Wood smoke components and functional properties. Smoked seafood safety. 55. https://doi.org/10.4027/isscp.2008.12
Moore TVL & Moore DK Jr (1998). Protective film peelably adhered to edible film enclosing product. United State Patent US 5741533 A
Wang W, Li C, Zhang H, Ni Y (2016) Using liquid smoke to improve mechanical and water resistance properties of gelatin films. J Food Sci. https://doi.org/10.1111/1750-3841.13282
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16(3):144–158
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med. https://doi.org/10.1016/s0891-5849(98)00315-3
Clinical and Laboratory Standards Institute. (2015). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard. CLSI document M07-A10, 10th ed. ISBN 1–56238–987–4
Balouiri M, Sadiki M, Ibnsouda SK (2016) Methods for in vitro evaluating antimicrobial activity: a review. J Pharm Anal. https://doi.org/10.1016/j.jpha.2015.11.005
Coma ME, Peltzer MA, Delgado JF, Salvay AG (2019) Water kefir grains as an innovative source of materials: study of plasticiser content on film properties. Eur Polym J. https://doi.org/10.1016/j.eurpolymj.2019.109234
Guggenheim EA (1966) Applications of statistical mechanics. Claredon Press, Oxford
Salvay AG, Colombo MF, Grigera JR (2003) Hydration effects on the structural properties and haem–haem interaction in haemoglobin. Phys Chem Chem Phys. https://doi.org/10.1039/B209560B
Delgado JF, Peltzer MA, Wagner JR, Salvay AG (2018) Hydration and water vapour transport properties in yeast biomass-based films: a study of plasticizer content and thickness effects. Eur Polym J. https://doi.org/10.1016/j.eurpolymj.2017.11.051
Delgado JF, Peltzer MA, Salvay AG (2022) Water vapour transport in biopolymeric materials: effects of thickness and water vapour pressure gradient on yeast biomass-based films. J Polym Environ. https://doi.org/10.1007/s10924-022-02412-6
Rezzoug M, Bakchiche B, Gherib A, Roberta A, Kilinçarslan Ö, Mammadov R, Bardaweel SK (2019) Chemical composition and bioactivity of essential oils and ethanolic extracts of Ocimum basilicum L. and thymus algeriensis boiss. & reut. from the algerian saharan atlas. BMC Complement Altern Med. https://doi.org/10.1186/s12906-019-2556-y
Al-Hassan AA, Norziah MH (2012) Starch–gelatin edible films: water vapor permeability and mechanical properties as affected by plasticizers. Food Hydrocoll. https://doi.org/10.1016/j.foodhyd.2011.04.015
Liu CK, Fishman ML, Hicks KB (2007) Composite films from pectin and fish skin gelatin or soybean flour protein. J Agric Food Chem. https://doi.org/10.1021/jf062612u
Pranoto Y, Lee CM, Park HJ (2007) Characterizations of fish gelatin films added with gellan and κ-carrageenan. Food Sci Technol. https://doi.org/10.1016/j.lwt.2006.04.005
Lozano-Navarro JI, Díaz-Zavala NP, Velasco-Santos C, Melo-Banda JA, Páramo-García U, Paraguay-Delgado F, Zapién-Castillo S (2018) Chitosan-starch films with natural extracts: physical, chemical, morphological and thermal properties. Materials. https://doi.org/10.3390/ma11010120
Azeredo HM, Waldron KW (2016) Crosslinking in polysaccharide and protein films and coatings for food contact–A review. J Food Sci Technol. https://doi.org/10.1016/j.tifs.2016.04.008
Choque E, Rezzani GD, Salvay AG, Mathieu F, Peltzer MA (2021) Impact of fungal extracts on the physical and antioxidant properties of bioactive films based on enzymatically hydrolyzed yeast cell wall. J Polym Environ. https://doi.org/10.1007/s10924-020-02004-2
Ullah K, Khan SA, Murtaza G, Sohail M, Manan A, Afzal A (2019) Gelatin-based hydrogels as potential biomaterials for colonic delivery of oxaliplatin. Int J Pharm. https://doi.org/10.1016/j.ijpharm.2018.12.020
Rahmasari Y, Yemiş GP (2022) Characterization of ginger starch-based edible films incorporated with coconut shell liquid smoke by ultrasound treatment and application for ground beef. Meat Sci. https://doi.org/10.1016/j.meatsci.2022.108799
Yu C, Chen H, Du D, Lv W, Li S, Li D, Liu D (2021) β-Glucan from Saccharomyces cerevisiae alleviates oxidative stress in LPS-stimulated RAW264. 7 cells via Dectin-1/Nrf2/HO-1 signaling pathway. Cell Stress and Chaperones. https://doi.org/10.1007/s12192-021-01205-5
Jaehrig SC, Rohn S, Kroh LW, Wildenauer FX, Lisdat F, Fleischer LG, Kurz T (2008) Antioxidative activity of (1→3), (1→6)-β-d-glucan from saccharomyces cerevisiae grown on different media. Food Sci Technol. https://doi.org/10.1016/j.lwt.2007.06.004
Gülçin I (2007) Comparison of in vitro antioxidant and antiradical activities of L-tyrosine and L-dopa. Amino Acids 32:431–438
van Overveld FW, Haenen GR, Rhemrev J, Vermeiden JP, Bast A (2000) Tyrosine as important contributor to the antioxidant capacity of seminal plasma. Chem Biol Interact. https://doi.org/10.1016/S0009-2797(00)00179-4
Ramírez Tapias YA, Peltzer MA, Delgado JF, Salvay AG (2020) Kombucha tea by-product as source of novel materials: formulation and characterization of films. Food Bioproc Tech. https://doi.org/10.1007/s11947-020-02471-4
Ramírez Tapias YA, Di Monte MV, Peltzer MA, Salvay AG (2022) Bacterial cellulose films production by Kombucha symbiotic community cultured on different herbal infusions. Food Chem. https://doi.org/10.1016/j.foodchem.2021.131346
Mali S, GrossmannGarcı́a MVEMA, Martino MN, Zaritzky NE (2004) Barrier, mechanical and optical properties of plasticized yam starch films. Carbohydr Polym. https://doi.org/10.1016/j.carbpol.2004.01.004
Acknowledgements
The authors would like to express their gratitude to Kerry Group for providing liquid smoke extract, the National Council for Scientific and Technical Research (CONICET), the National University of Quilmes for their financial support, and Dr. Vanesa Ludemann for assisting with the antimicrobial testing.
Funding
This work has been possible thanks to the support of the Agencia Nacional de Promoción Científica y Tecnológica (Argentina) through PICT-2021–0092 and the National University of Quilmes (UNQ, Argentina) through R&D program PUNQ R 990/19 Expte: 827–1300/19.
Author information
Authors and Affiliations
Contributions
GDR: Methodology, investigation, data curation and writing-original draft preparation. AGS: Methodology, investigation, data curation, writing-original draft preparation, writing-review and editing, supervision. MAP: Investigation, writing-original draft preparation, writing-review and editing, supervision and funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezzani, G.D., Salvay, A.G. & Peltzer, M.A. Bioactive Materials from Brewery Yeast Cell Wall Residues with the Addition of Refined Liquid Smoke: Development and Study of Functional Properties. J Polym Environ (2024). https://doi.org/10.1007/s10924-023-03165-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s10924-023-03165-6