Abstract
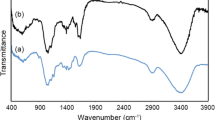
Modified cellulose from the pellicle produced by Rhodococcus sp. MI 2 was more efficient at removing Fe(III) and Cu(II) from aqueous solution than similarly modified cellulose from Komagataeibar xylinus TISTR 998. This study first describes the modification of the cellulose to produce mercerized bacterial cellulose (MBC), phosphorylated bacterial cellulose (PBC), acid–base cellulose and diethylenetriamine bacterial cellulose (EABC). Their efficacy as adsorbents to adsorb Fe(III) and Cu(II) was then determined. In aqueous solution at pH 4 at initial Fe(III) concentration of 20 mg/L, PBC reached adsorption equilibrium within 195 min. At pH 5 in an initial Cu(II) concentration of 75 mg/L, EABC reached adsorption equilibrium within 210 min. Molecular structures and chemical bonds were examined by Fourier transform infrared spectroscopy (FT-IR) and physical morphologies by scanning electron microscopy. The adsorption kinetics of MBC, PBC and EABC showed good agreement with the proposed pseudo-second order model and the adsorption isotherm was best described by the Freundlich model. Our study determined optimal conditions, molecular structures, physical morphologies and adsorption kinetics. The cellulose produced by the new strain Rhodococcus sp. MI 2 was highly efficient at adsorbing and removing metal ions from aqueous solution.








Similar content being viewed by others
References
Fergusson JE (1990) The heavy elements: chemistry, environmental impact and health effects. Pergamon Press, Oxford
Bradl H (2002) Heavy metals in the environment: origin, interaction and remediation. Academic Press, London
He ZL, Yang XE, Stoffella PJ (2005) J Trace Elem Med Biol 19(2–3):125–140
Shallari S, Schwartz C, Hasko A (1998) Sci Total Environ 19209:133–142
Nriagu JO (1989) Nature 338:47–49
WHO/FAO/IAEA (1996) Trace elements in human nutrition and health. World Health Organization, Geneva
Stern BR (2010) Toxicol Environ Health A 73(2):114–127
Harvey LJ, McArdle HJ (2008) Br J Nutr 99(S3):S10–S13
ATSDR (Agency for Toxic Substances and Disease Registry) (2002) Toxicological profile for copper. Centers for Disease Control, Atlanta
Vuori K-M (1995) Annal Zoo Fennici 32:317–329
Tchounwou P, Newsome C, Williams J (2008) Met Ions Biol Med 10:285–290
USEPA (US Environmental Protection Agency) (2015) Regulated drinking water contaminants. USEPA, Washington, DC
ATSDR (Agency for Toxic Substances and Disease Registry) (2015) Toxicological profiles, toxic substances portal. Department of Health and Human Services, Atlanta
EPA US (American Public Health Assoc US) (1993) Standard methods for the examination of water and waste-water. Environmental Protection Agency, Washington, DC
Phippen B, Horvath C, Nordin R (2008) Ambient water quality guidelines for iron: overview. Ministry of Environment Province of British Columbia, Nanaimo
Becker M, Asch F (2005) J Plant Nutr Soil Sci 168:558–573
Zhu MX, Lee L, Wang HH, Wang Z (2007) J Hazard Mater 149(3):735–741
Bayramoglu G, Altintas B, Arica MY (2009) Chem Eng J 152(2/3):339–346
Shu HY, Chang M-C, Yu H-H (2007) J Colloid Interface Sci 314(1):89–97
Yola M, Eren T, Atar N, Wang S (2013) Chem Eng J 242:333–340
Chen S, Zou Y, Yan Z (2009) J Hazard Mater 161:1355–1359
Li N, Bai RB (2005) Sep Purif Technol 42(3):237–247
Oshima T, Kondo K, Ohto K (2008) Funct Polym 68:376–383
Shen W, Chen S, Shi S (2009) Carbohydr Polym 75:110–114
Tanskul S, Amornthatree K, Jaturonlak N (2013) Carbohydr Polym 92:421–428
Tanskul S, Damthongsen T, Jaturonlak N (2018) Biosci J 34(3):666–673
Chen S, Shen W, Yu F (2010) J Appl Polym Sci 117:8–15
Lagergren S (1898) K Sven Vetenskapsakad Handl 24(4):1–39
Basurco J, Torem ML (2010) Chem Eng J 161(1):1–8
Vijayakumar G, Tamilarasan R, Dharmendirakumar M (2012) J Mater Environ Sci 3(1):157–170
Takagai Y, Shibata A, Kiyokawa S, Takase T (2011) J Colloid Interface Sci 353(2):593–597
Hokkanen S, Repo E, Sillanpää M (2013) Chem Eng J 223:40–47
Nakamoto K (1977) Infrared and Raman spectra of inorganic and coordination compounds, 3rd edn. Wiley-Interscience, New York
Hameed BH (2008) J Hazard Mater 154(1–3):204–212
Acknowledgements
This work was supported by the government budget of Prince of Songkla University, Thailand. The authors would like to thank Mr. Thomas Duncan Coyne and Ms. Anna Chatthong for assistance with the English.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yingkong, P., Tanskul, S. Adsorption of Iron(III) and Copper(II) by Bacterial Cellulose from Rhodococcus sp. MI 2. J Polym Environ 27, 1948–1958 (2019). https://doi.org/10.1007/s10924-019-01480-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-019-01480-5