Abstract
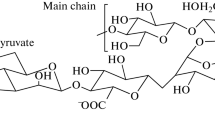
In this paper, we report acetylation of xanthan gum using acetic anhydride in an ionic liquid solvent, 1-butyl-3-methylimidazolium chloride (BMIMCl). Xanthan gum was dissolved with BMIMCl [2 % (w/w)] and the reaction was carried out in the presence of acetic anhydride (five equiv. for hydroxy groups in a repeating unit) with stirring the solution at elevated temperatures. The structures of xanthan gum acetates were confirmed by the 1H NMR and IR spectra. The degree of acetylation (DA) values determined by the 1H NMR analysis increased with the higher reaction temperatures. The thermal gravimetric analysis (TGA) indicated the enhancement of thermal stability by acetylation. Furthermore, the TGA as well as differential scanning calorimetric (DSC) analysis of the products suggested the presence of the highly and less acetylated segments in a xanthan gum chain. The DSC profile of the product with the high DA value also exhibited a small endothermic peak, which might potentially be ascribed to the melting temperature.





Similar content being viewed by others
References
Schuerch C (1986) Polysaccharides. In: Mark HF, Bilkales N, Overberger CG (eds) Encyclopedia of polymer science and engineering, vol 13, 2nd edn. Wiley, New York, pp 87–162
Rouilly A, Rigal L (2002) J Macromol Sci Part C 42:441
Klemm D, Heublein B, Fink H-P, Bohn A (2005) Angew Chem Int Ed 44:3358
Klemm D, Kramer F, Moritz S, Lindström T, Ankerfors M, Gray D, Dorris A (2011) Angew Chem Int Ed 50:5438
Stephen AM, Philips GO, Williams PA (1995) Food polysaccharides and their applications. Taylor & Francis, London
Jansson PE, Kenne L, Lindberg B (1975) Carbohydr Res 45:275
Melton LD, Mindt L, Rees DA (1976) Carbohydr Res 46:245
Kaura G, Mahajana M, Bassi P (2013) Int J Polym Mater Polym Biomater 62:475
Doelker E (1993) Adv Polym Sci 107:199
Kurita K (2001) Prog Polym Sci 26:1921
Morimoto M, Saimoto H, Shigemasa Y (2002) Trends Glycosci Glycotechnol 14:205
Sashiwa H, Aiba S (2004) Prog Polym Sci 29:887
Hamcerencu M, Desbrieres J, Popa M, Khoukh A, Riess G (2007) Polymer 48:1921
Fundador NG, Enomoto-Rogers Y, Takemura A, Iwata T (2012) Carbohydr Polym 87:170
Fundador NG, Enomoto-Rogers Y, Takemura A, Iwata T (2012) Polymer 53:3885
Enomoto-Rogers Y, Ohmoto Y, Iwata T (2013) Carbohydr Polym 92:1827
Liebert T, Heinze T (2008) Bioresources 3:576
Feng L, Chen Z-I (2008) J Mol Liq 142:1
Pinkert A, Marsh KN, Pang S, Staiger MP (2009) Chem Rev 109:6712
Zakrzewska ME, Lukasik EB, Lukasik RB (2010) Energy Fuels 24:737
Swatloski RP, Spear SK, Holbrey JD, Rogers RD (2002) J Am Chem Soc 124:4974
Wu J, Zhang J, Zhang H, He J, Ren Q, Guo M (2004) Biomacromolecules 5:266
Heinze T, Schwikal K, Barthel S (2005) Macromol Biosci 5:520
Barthel S, Heinze T (2006) Green Chem 8:301
Schlufter K, Schmauder HP, Dorn S, Heinze T (2006) Macromol Rapid Commun 27:1670
Mine S, Izawa H, Kaneko K, Kadokawa J (2009) Carbohydr Res 344:2263
Huang K, Wang B, Cao Y, Li H, Wang J, Lin W, Mu C, Liao D (2011) J Agric Food Chem 59:5376
Gibril ME, Yue Z, Da LX, Huan L, Xuan Z, Feng LH, Muhuo Y (2012) Int J Eng Sci Technol 4:3356
Izawa H, Kaneko Y, Kadokawa J (2009) J Mater Chem 19:6969
Izawa H, Kadokawa J (2010) J Mater Chem 20:5235
Wyatt NB, Liberatore MW (2009) J Appl Polym Sci 114:4076
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Endo, R., Setoyama, M., Yamamoto, K. et al. Acetylation of Xanthan Gum in Ionic Liquid. J Polym Environ 23, 199–205 (2015). https://doi.org/10.1007/s10924-014-0701-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-014-0701-8