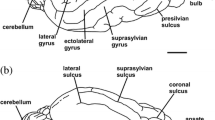
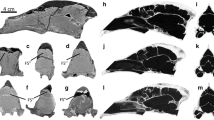
Abstract
Pinnipeds are semiaquatic carnivorans that spend most of their lives in water. Although there is currently a better understanding of the phylogenetic relationships among pinniped species, there is a lack of neurobiological studies of their brains conducted in an evolutionary framework. We reconstructed virtual 3D brain endocast of 29 skulls corresponding to adult and subadult specimens, both male and female, belonging to 18 species of pinnipeds. Data collection involved mostly newly acquired computer tomography scan and µCT scan data. In the Otariidae species studied, the olfactory bulbs are proportionally more developed than in other pinnipeds. Individuals of these species spend longer periods on land than phocids and have a social structure in which olfactory information/cues play an important role. The olfactory bulb of phocids is smaller than that of the otariids, with the exception of Hydrurga leptonyx and the males of Mirounga leonina, in which they significantly even larger than those of otariids. The frontal cortex is more extended in otariids and odobenids than in phocids, probably due to a functional relation to a complex social structure. Concerning the temporal cortex, which is related to hearing/orientation, we recorded an enlargement of the posterior zone in phocids, probably related to the need for orientation at great depths in the absence of light, whereas in otariids and odobenids this extension is similar to that of terrestrial carnivorans. The occipital cortex, linked to vision, is well developed in Ommatophoca rossii and Hydrurga leptonyx, and slightly less so in Mirounga leonina and Leptonychotes weddellii.








Similar content being viewed by others
Availability of data and materials
All data are included in the text and Openline Resource 1 file.
Change history
16 September 2023
A Correction to this paper has been published: https://doi.org/10.1007/s10914-023-09686-0
References
Arsznov BM, Lundrigan BL, Holekamp KE, Sakai ST (2010) Sex and the frontal cortex: A developmental CT study in the spotted hyena. Brain Behav Evol 76:185–197. https://doi.org/10.1159/000321317
Arsznov BM, Sakai ST (2012) Pride diaries: Sex, brain size and sociality in the African lion (Panthera leo) and cougar (Puma concolor). Brain Behav Evol 79:275–289. https://doi.org/10.1159/000338670
Berta A (2009) Pinnipedia, overview. In: Perrin WF, Wursig B, Thewissen JGM (eds) Encyclopedia of Marine Mammals, 2nd edn. Academic Press, San Diego, California, pp 878–885
Berta A, Adam PJ (2001) Evolutionary biology of pinnipeds. In: Mazin J-M, de Buffrénil V (eds) Secondary Adaptation of Tetrapods to Life in Water. Verlag Dr. Friedrich Pfeil, Munich, pp 235–260
Berta A, Churchill M, Boessenecker, R W (2018) The origin and evolutionary biology of pinnipeds: seals, sea lions, and walruses. Ann Rev Earth Planet Sci 46:203-228
Berta A, Sumich J, Kovacs K (2005) Marine Mammals: Evolutionary Biology, 2nd ed. Elsevier. https://doi.org/10.1016/B978-0-12-088552-7.X5000-0
Bertrand OC, San Martin-Flores G, Silcox MT (2019) Endocranial shape variation in the squirrel-related clade and their fossil relatives using 3D geometric morphometrics: contributions of locomotion and phylogeny to brain shape. J Zool 308:197–211. https://doi.org/10.1111/jzo.12665
Bertrand OC, Silcox MT (2016) First virtual endocasts of a fossil rodent: Ischyromys typus (Ischyromyidae, Oligocene) and brain evolution in rodents. J Vertebr Paleontol 36:e1095762.
Campagna C, Piola AR, Marin MR, Lewis M, Zajaczkovski U, Fernández T, (2007) Deep divers in shallow seas: Southern elephant seals on the Patagonian shelf. Deep Res Part I Oceanogr Res Pap 54:1792–1814. https://doi.org/10.1016/j.dsr.2007.06.006
Campagna C, Quintana F, Boeuf BJL, Blackwell SB, Crocker DE (1995) Diving behaviour and foraging location of female southern elephant seals from Patagonia. Aquat Mamm 24:1–11. https://doi.org/10.1111/j.1469-7998.1995.tb01784.x
Campagna C, Rivas AL, Marin MR (2000) Temperature and depth profiles recorded during dives of elephant seals reflect distinct ocean environments. J Mar Syst 24:299–312. https://doi.org/10.1016/S0924-7963(99)00091-3
Carreira LM, Ferreira A (2015) Anatomical variations in the pseudosylvian fissure morphology of brachy-, dolicho-, and mesaticephalic dogs. Anat Rec 298:1255–1260. https://doi.org/10.1002/ar.23171
Claude M (2013) Log-shape ratios, Procrustes superimposition, elliptic Fourier analysis: Three worked examples in R. Hystrix, Ital J Mammal 24:94–102. https://doi.org/10.4404/hystrix-24.1-6316
Duque A, McCormick DA (2010) Circuit-based localization of ferret prefrontal cortex. Cereb Cortex 20:1020–1036. https://doi.org/10.1093/cercor/bhp164
Eisert R, Potter CW, Oftedal OT (2013) Brain size in neonatal and adult Weddell seals: Costs and consequences of having a large brain. Mar Mammal Sci 30:184–205. https://doi.org/10.1111/mms.12033
Farris JS (1970) Methods for computing Wagner trees. Syst Zool 19(1):83–92. https://doi.org/10.2307/2412028
García N, Santos E, Arsuaga JL, Carretero JM (2007) Endocranial morphology of the Ursus deningeri von Reichenau 1904 from the sima de los huesos (sierra de Atapuerca) Middle Pleistocene site. J Vertebr Paleontol 27(4):1007-1017
Glezer II (2002) Neural morphology. In: Rus Hoelzel A (ed) Marine Mammal Biology: An Evolutionary Approach. 1st edn. pp 98–115.
Haddow Brown NK (2015) Descripción anatómica de encéfalos de crías de lobo fino austral (Arctocephalus australis Zimmerman 1783) de Isla Guafo, Región de Los Lagos, Chile. Dissertation, Facultad de Ciencias Veterinarias, Universidad Austral de Chile
Hamuy TP, Bromiley RB, Woolsey C N (1956). Somatic afferent areas I and II of dog's cerebral cortex. J Neurophysiol 19(6):485-499
Holekamp KE, Sakai ST, Lundrigan BL (2007) Social intelligence in the spotted hyena (Crocuta crocuta). Philos Trans R Soc B Biol Sci 362:523–538. https://doi.org/10.1098/rstb.2006.1993
Isler K, Christopher Kirk E, Miller JMA, Albrecht GA, Gelvin BR, Martin RD (2008) Endocranial volumes of primate species: scaling analyses using a comprehensive and reliable data set. J Hum Evol 55:967–978. https://doi.org/10.1016/j.jhevol.2008.08.004
Jeffery N, Ryan TM, Spoor F (2008) The primate subarcuate fossa and its relationship to the semicircular canals part II: adult interspecific variation. J Hum Evol 55(2):326-339.
Jeffery N, Spoor F (2006). The primate subarcuate fossa and its relationship to the semicircular canals part I: prenatal growth. J Hum Evol 51(5): 537-549.
Kooyman G (1966) Maximum diving capacities of the Weddell seal, Leptonychotes weddelli. Science 151:1553–1554. https://doi.org/10.1126/science.151.3717.1553
Larsell O (1970) The Comparative Anatomy and Histology of the Cerebellum: From Monotremes through Apes. University of Minnesota Press, Minneapolis
Loza CM (2016a) Morfología comparada y ontogenia del oído medio e interno en Pinnípedos (Otariidae y Phocidae, Carnivora) de la Argentina y Antártida: Aspectos ecomorfológicos. Dissertation, Facultad de Ciencias Naturales y Museo, Uiversidad Nacioanl de La Plata
Loza CM (2016b) Nomenclatura osteológica y accidentes de la región ótica de pinnípedos, y sinónimos. Rev Mus La Plata 1:117–158. https://doi.org/10.24215/25456377e009
Loza CM, Latimer AE, Sánchez-Villagra MR, Carlini AA (2017) Sensory anatomy of the most aquatic of carnivorans: The Antarctic Ross seal, and convergences with other mammals. Biol Lett 13. https://doi.org/10.1098/rsbl.2017.0489
Lyras GA, van der Geer AAE (2003) External brain anatomy in relation to the phylogeny of Caninae (Carnivora: Canidae). Zool J Linn Soc 138:505–522. https://doi.org/10.1046/j.1096-3642.2003.00067.x
Lyras GA, van der Geer A E, Werdelin L (2022) Paleoneurology of Carnivora. In: Dozo MT, Paulina-Carabajal A, Macrini TE, Walsh S (eds) Paleoneurology of Amniotes, New Directions in the Study of Fossil Endocasts. Springer, Cham, Switzerland, pp 681–710
Macrini TE, Rowe T, VandeBerg JL (2007) Cranial endocasts from a growth series of Monodelphis domestica (Didelphidae, Marsupialia): A study of individual and ontogenetic variation. J Morphol 268:844–865. https://doi.org/10.1002/jmor.10556
Maddison WP, Maddison DR (2019) Mesquite: A Modular System for Evolutionary Analysis. Version 3.61. http://mesquiteproject.org
Manger PR (2006) An examination of cetacean brain structure with a novel hypothesis correlating thermogenesis to the evolution of a big brain. Biol Rev Camb Philos Soc 81:293–338. https://doi.org/10.1017/S1464793106007019
Marino L, Mcshea DW, Uhen MD (2004) Origin and evolution of large brains in toothed whales. Anat Rec A Discov Mol Cell Evol Biol 281(2):1247–1255. https://doi.org/10.1002/ar.a.20128
Marino L, Murphy TL, Deweerd AL, Morris JA, Fobbs AJ, Humblot N, Ridgway SH, Johnson JI (2001a) Anatomy and three-dimensional reconstructions of the brain of the white whale (Delphinapterus leucas) from magnetic resonance images. Anat Rec 262:429–439. https://doi.org/10.1002/ar.10018
Marino L, Sudheimer KD, Murphy TL, Davis KK, Pabst DA, McLellan WA, Rilling JK, Johnson JI (2001b) Anatomy and three-dimensional reconstructions of the brain of a bottlenose dolphin (Tursiops truncatus) from magnetic resonance images. Anat Rec 264:397–414. https://doi.org/10.1002/ar.10018
Marino L, Sudheimer K, Pabst DA, McLellan WA, Johnson JI (2003a) Magnetic resonance images of the brain of a dwarf sperm whale (Kogia simus). J Anat 203:57–76. https://doi.org/10.1046/j.1469-7580.2003.00199.x
Marino L, Uhen MD, Pyenson ND, Frohlich B (2003b) Reconstructing cetacean brain evolution using computed tomography. Anat Rec B New Anat 272(1):107–117. https://doi.org/10.1002/ar.b.10018
McIntyre T, Tosh CA, Plötz J, Bornemann H, Bester MN (2010) Segregation in a sexually dimorphic mammal: a mixed-effects modelling analysis of diving behaviour in southern elephant seals. Mar Ecol Prog Ser 412:293–304. https://doi.org/10.3354/meps08680
Miller EK, Freedman DJ, Wallis JD (2002) The prefrontal cortex: Categories, concepts and cognition. Philos Trans R Soc B Biol Sci 357:1123–1136. https://doi.org/10.1098/rstb.2002.1099
Milne AO, Muchlinski MN, Orton LD, Sullivan MS, and Grant R A (2022) Comparing vibrissal morphology and infraorbital foramen area in pinnipeds. Anat Rec 305:556–567.
Mittermeier RA, Wilson, DE (2014) Handbook of the Mammals of the World, Vol. 4: Sea Mammals. Lynks Edicions, Barcelona
Montie EW, Pussini N, Schneider GE, Battey TWK, Dennison S, Barakos J, Gulland F (2009) Neuroanatomy and volumes of brain structures of a live California sea lion (Zalophus californianus) from magnetic resonance images. Anat Rec 292:1523–1547. https://doi.org/10.1002/ar.20937
Morgane PJ, Jacobs MS, Galaburda A (1986) Evolutionary morphology of the dolphin brain. In: Schusterman RJ, Thomas JA, Wood F (eds) Dolphin Cognition and Behavior: A Comparative Approach. Lawrence Erlbaum Associates, Inc. Publishers, Hillsdale, New Jersey, pp 5–30.
Mosimann JE (1970) Size allometry: Size and shape variables with characterizations of the lognormal and generalized gamma distributions. J Am Stat Assoc 65:930–945. https://doi.org/10.1080/01621459.1970.10481136
Mosimann JE, James FC (1979) New statistical methods for allometry with application to Florida red-winged blackbirds. Evolution 33:444–459.
Muchlinski MN (2008) The relationship between the infraorbital foramen, infraorbital nerve, and maxillary mechanoreception: implications for interpreting the paleoecology of fossil mammals based on infraorbital foramen size. Anat Rec 291:1221–1226.
Muchlinski MN, Wible JR, Corfe I, Sullivan M, Grant RA. 2020. Good vibrations: The evolution of whisking in small mammals. Anat Rec 303:89–99.
NAV (2017) Nomina Anatomica Veterinaria, 6th ed. Editorial Committee Hanover (Germany), Ghent (Belgium), Columbia, MO (U.S.A.), Rio de Janeiro (Brazil). https://doi.org/10.1111/j.1751-0813.1971.tb02174.x
Oelschlager HHA, Ridgway SH, Knauth M (2010) Cetacean brain evolution: Dwarf sperm whale (Kogia sima) and common dolphin (Delphinus delphis). An investigation with high-resolution 3D MRI. Brain Behav Evol 75:33–62. https://doi.org/10.1159/000293601
Pellegrino FC (2000) Revisión anatómica del neopalio del perro. Rev Chil Anat 18:1–22. https://doi.org/10.4067/s0716-98682000000100005
Perini FA, Macrini TE, Flynn JJ, Bamba K, Ni X, Croft DA, Wyss AR (2022) Comparative endocranial anatomy, encephalization, and phylogeny of Notoungulata (Placentalia, Mammalia). J Mamm Evol 29:369–394. https://doi.org/10.1007/s10914-021-09583-4
Petrovic V, Sabol M, Šurka J, Pyszko M, Stehlik L (2018) External brain morphology of juvenile cave hyena (Crocuta crocuta spelaea) from the jasovská jaskyna cave (Slovakia) revealed by x-ray computed tomography. Acta Geol Slovaca 10:133–142.
Radinsky LB (1968) Evolution of somatic sensory specialization in otter brains. J Comp Neurol 134:495-505.
Radinsky L (1971) An example of parallelism in carnivore brain evolution. Evolution 25:518–522. https://doi.org/10.2307/2407350
Radinsky L (1975) Primate brain evolution: Comparative studies of brains of living mammal species reveal major trends in the evolutionary development of primate brains, and analysis of endocasts from fossil primate braincases suggests when these specializations occurred. Am Sci 63:656–663.
Riedman M (1990) The Pinnipeds: Seals, Sea lions, and Walruses. University of California Press, Berkeley
Reep RL, O’Shea TJ (1990) Regional brain morphometry and lissencephaly in the Sirenia. Brain Behav Evol 35:185–194.
Reep RL, Marshall CD, Stoll ML, Whitak DM (1998) Distribution and innervation of facial bristles and hairs in the Florida manatee (Trichechus manatus latirostris). Mar Mammal Sci 14(2):257–273. https://doi.org/10.1111/j.1748.7692.1998.tb00715.x
Ridgway SH (1986) Physiological observations on dolphin brains. In: Schusterman RJ, Thomas JA, Wood FG (eds) Dolphin Cognition and Behavior: A Comparative Approach. Lawrence Erlbaum Associates, Inc. Publishers, Hillsdale, New Jersey, pp 31–59
Ridgway SH, Harrison R (1993) Handbook of Marine Mammals: The First Book of Dolphins. Academic Press, San Diego, California
Sakai ST, Arsznov BM, Hristova AE, Yoon EJ, Lundrigan BL (2016) Big cat coalitions: A comparative analysis of regional brain volumes in Felidae. Front Neuroanat 10:1–12. https://doi.org/10.3389/fnana.2016.00099
Sakai ST, Arsznov BM, Lundrigan BL, Holekamp KE (2011) Brain size and social complexity: A computed tomography study in Hyaenidae. Brain Behav Evol 77:91–104. https://doi.org/10.1159/000323849
Sakai ST, Whitt B, Arsznov BM, Lundrigan BL (2018) Endocranial development in the coyote (Canis latrans) and gray wolf (Canis lupus): A computed tomographic study. Brain Behav Evol 91(2):65-81. https://doi.org/10.1159/000487427
Sawyer EK, Turner EC, Kaas JH (2016) Somatosensory brainstem, thalamus, and cortex of the California sea lion (Zalophus californianus). J Comp Neurol 524:1957–1975. https://doi.org/10.1002/cne.23984
Sienkiewicz T, Sergiel A, Huber D, Maślak R, Wrzosek M, Podgórski P, Reljić S, Paśko Ł (2019) The brain anatomy of the brown bear (Carnivora, Ursus arctos L., 1758) compared to that of other carnivorans: A cross-section study using MRI. Front Neuroanat 13:79. https://doi.org/10.3389/fnana.2019.00079
Thiery G, Ducrocq S (2015) Endocasts and brain evolution in Anthracotheriidae (Artiodactyla, Hippopotamoidea). J Anat 227:277-285. https://doi.org/10.1111/joa.12348
Turner EC, Sawyer EK, Kaas JH (2017) Optic nerve, superior colliculus, visual thalamus, and primary visual cortex of the northern elephant seal (Mirounga angustirostris) and California sea lion (Zalophus californianus). J Comp Neurol 525:2109-2132
Turner W (1890) The convolutions of the brain: a study in comparative anatomy. J Anat Physiol 25:105–153.
Welker WI, Campos GB (1963) Physiological significance of sulci in somatic sensory cerebral cortex in mammals of the family Procyonidae. J Comp Neurol 120(1):19-36.
Welker WI, Johnson JI Jr, Pubols BH Jr (1964) Some morphological and physiological characteristics of the somatic sensory system in raccoons. Am Zool 75–94.
Welker W I, Seidenstein S (1959) Somatic sensory representation in the cerebral cortex of the racoon (Procyon lotor). J Comp Neurol 111(3):469-501.
Woolsey CN (1958) Organization of somatic sensory and motor areas of the cerebral cortex. In: Harlow HF, Woolsey CN (eds) Biological and Biochemical Bases of Behavior. University Wisconsin Press, Madison, pp 63–81
Acknowledgements
We are grateful to Drs. Diego Verzi and Itatí Olivares of the Vertebrate Zooology Division of Museo de La Plata (MLP) (Argentina), Dr. Pablo Teta and Mr. Sergio Lucero of Museo Argentino de Ciencias Naturales, Bernardino Rivadavia (MACN) (Argentine), Dr. Javier Negrete of Instituto Antártico Argentino (IAA), and Dr. Alexandra Wegmann Vertebrate Collection, Department of Palaeontology at Universityof Zurich (Switzerland), for providing access to the specimens under their care; to the technicians at CIMED La Plata for their assistance during CT scanning; to Dr. J. Maisano (Digimorph.org), T. Rowe's NSF Digital Libraries Grant, and Morphosurce (https://www.morphosource.org) for the Odobenus rossmarus, Callorhinus ursinus, and Zalophus californianus scans; and to Alexandra Wegmann for the high resolution CT use at the University of Zurich, to Dr. Christy A. Hipsley for the reading and comments on an early version of the ms. We thank two anonymous reviewers and the editors at JME for valuable comments used to improve the manuscript.
Funding
This work was partially funded by the Consejo Nacional de Investigaciones Científicas (PIP-0798) and Universidad Nacional de La Plata (N-871) to AAC, Argentina.
Author information
Authors and Affiliations
Contributions
CML, and AAC: conceived and designed research. CML: conducted macroscopic observations, 3D reconstructions. CML, and MR: described the samples. MS-V performed the mapping of characters. CML, MR, and AAC: designed and performed the figures. ACS: performed the statistical analysis. CML, MS-V, CGB, and AAC analyzed data and wrote the first draft of the manuscript. CML, MS-V, and AAC: wrote the final version of the manuscript. The results presented in this study resulted from a collaborative effort between the authors. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
The original online version of this article was revised: There was a small omission in the Author Information (Authors' Contributions)
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Loza, C.M., Sánchez-Villagra, M.R., Scarano, A.C. et al. The brain of fur seals, seals, and walrus (Pinnipedia): A comparative anatomical and phylogenetic study of cranial endocasts of semiaquatic mammals. J Mammal Evol 30, 1011–1028 (2023). https://doi.org/10.1007/s10914-023-09679-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-023-09679-z