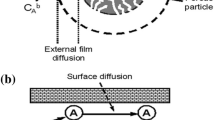
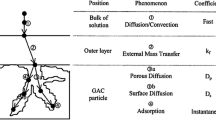
Abstract
In this paper, we have developed an accurate and efficient Haar wavelet method to solve film-pore diffusion model. Film-pore diffusion model is widely used to determine study the kinetics of adsorption systems. To the best of our knowledge, until now rigorous wavelet solution has been not reported for solving film-pore diffusion model. The use of Haar wavelets is found to be accurate, simple, fast, flexible, convenient, and computationally attractive. The power of the manageable method is confirmed. It is shown that film-pore diffusion model satisfactorily describes the kinetics of methylene blue adsorption onto three low-cost adsorbents, Gauva, teak and gulmohar plant leaf powders, used in this study.
Similar content being viewed by others
Abbreviations
- As :
-
Total surface area of all the particles, m2dm−3
- Bt:
-
Biot number (Bt = k fdp/Deff), −
- Ct :
-
Bulk concentration at time t, mg dm−3
- Cs :
-
Surface concentration, mg dm−3
- Co :
-
Initial bulk concentration, mg dm−3
- dp :
-
Diameter of particle, m
- Deff :
-
Internal effective diffusivity, m2 s−1
- KL :
-
Langmuir adsorption constant, dm3mg−1
- r:
-
Radial position in the particle, m
- R:
-
Radius of the particle, m
- qe :
-
Solid phase dye concentration at equilibrium, mg g−1
- qi :
-
Solid phase dye concentration at grid i at time t, mg g−1
- t:
-
Time, s or min
- V:
-
Volume of solution, dm3
- ε :
-
Particle porosity, –
- ρ p :
-
Particle density, kg m−3
- kf :
-
External film transfer coefficient, m s−1
References
Ponnusami V., Rajan K.S., Srivastava S.N.: Application of film-pore diffusion model, for methyline blue adsorption onto plant leaf powders. Chem. Eng. J. 163(3), 236–242 (2010)
Ponnusami V., Gunasekar V., Srivastava S.N.: Kinetics of methylene blue removal from aqueous solution using gulmohar (Delonix regia) plant leaf powder: multivariate regression analysis. J. Hazard. Mater. 169, 119–127 (2009)
Ponnusami V., Krithika V., Madhuram R., Srivastava S.N.: Biosorption of reactive dye using acid-treated rice husk: factorial design analysis. J. Hazard. Mater. 142, 397–403 (2007)
Cheung C.W., Chan C.K., Porter J.F., McKay G.: Film-pore diffusion control for the batch sorption of cadmium ions from effluent onto bone char. J. Colloid Interface Sci. 234(2), 328–336 (2001)
Hameed B.H.: Removal of cationic dye from aqueous solution using jackfruit peel as non-conventional low-cost adsorbent. J. Hazard. Mater. 162(1), 344–350 (2009)
Hameed B.H., Krishni R.R., Sata S.A.: A novel agricultural waste adsorbent for the removal of cationic dye from aqueous solutions. J. Hazard. Mater. 162(1), 305–311 (2009)
Gupta N., Kushwaha A.K., Chattopadhyaya M.C.: Adsorption studies of cationic dyes onto Ashoka (Saraca asoca) leaf powder. J. Taiwan Inst. Chem. Eng. 43(4), 604–613 (2012)
Hameed B.H., Mahmoud D.K., Ahmad A.L.: Sorption of basic dye from aqueous solution by pomelo (Citrus grandis) peel in a batch system. Colloids Surf. A Physicochem. Eng. Aspects 316(1–3), 78–84 (2008)
Çelekli A., Ilgün G., Bozkurt H.: Sorption equilibrium, kinetic, thermodynamic, and desorption studies of reactive red 120 on Chara contraria. Chem. Eng. J. 191, 228–235 (2012)
S. Kumar, V. Gunasekar, V. Ponnusami, Removal of methylene blue from aqueous effluent using fixed bed of groundnut shell powder. J. Chem. (in press). doi:10.1155/2013/259819
B.H. Hameed, M.I. El-Khaiary, Sorption kinetics and isotherm studies of a cationic dye using agricultural waste: broad bean peels. J. Hazard. Mater. 154(1–3)15, 639–648 (2008)
Chen C.F., Hsiao C.H.: Haar wavelet method for solving lumped and distributed-parameter systems. IEEE Proc. Pt. D 144(1), 87–94 (1997)
C.H. Hsiao, State analysis of linear time delayed systems via Haar wavelets. Math. Comput. Simul. 44(5) 457–470 (1997)
Z. Shi, T. Liu, B. Gao, International Conference on Computer Application and System Modeling (ICCASM 2010), IEEE Proceeding. Haar wavelet method for solving wave equation (2010)
Fazal-I-Haq, , Aziz I., Aziz I.: A Haar wavelets based numerical method for eight-order boundary problems. Int. J. Math. Comput. Sci. 6(1), 25–31 (2010)
Wu J.L: A wavelet operational method for solving fractional partial differential equations numerically. Appl. Math. Comput. 214(1), 31–40 (2009)
Z. Shi, Y.-Y. Cao, Q. -J. Chen, Solving 2D and 3D Poisson equations and biharmonic equations by the Haar wavelet method. Appl. Math. Model. 36(11), 5143–5161 (2012)
W. Geng, Y. Chen, Y. Li, D. Wang, Wavelet method for nonlinear partial differential equations of fractional order, Comput. Inf. Sci. 4(5) 28–35 (2011)
Lepik U.: Numerical solution of evolution equations by the Haar wavelet method. Appl. Math. Comput. 185, 695–704 (2007)
Lepik U.: Numerical solution of differential equations using Haar wavelets. Math. Comput. Simul. 68, 127–143 (2005)
U. Lepik, Application of the Haar wavelet transform to solving integral and differential equations, Proc. Estonian Acad. Sci. Phys. Math. 56(1), 28–46 (2007)
G. Hariharan, K. Kannan, Haar wavelet method for solving some nonlinear parabolic equations. J. Math. Chem. 48(4), 1044–1061 (2010)
G. Hariharan, K. Kannan, A comparative study of a Haar Wavelet method and a restrictive Taylor’s series method for solving convection-diffusion equations. Int. J. Comput. Methods Eng. Sci. Mech. 11(4), 173–184 (2010)
Hariharan G.: Haar wavelet method for solving Sine-Gordon and Klein-Gordon equations. Int. J. Nonlinear Sci. 9(2), 1–10 (2010)
Hariharan G., Kannan K.: Haar wavelet method for solving fitz Hugh-Nagumo equation. Int. J. Math. Stat. Sci. 2, 2 (2010)
Porter D.C.K., McKay G.: Film-pore diffusion model for the fixed-bed sorption of copper and cadmium ions onto bone char. Water Res. 35, 3876–3886 (2001)
Ho Y.S., McKay G.: A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf. Environ. Prot. 76(B4), 332–340 (1998)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hariharan, G., Ponnusami, V. & Srikanth, R. Wavelet method to film-pore diffusion model for methylene blue adsorption onto plant leaf powders. J Math Chem 50, 2775–2785 (2012). https://doi.org/10.1007/s10910-012-0063-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10910-012-0063-1