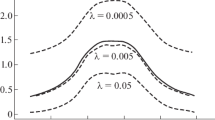
In this paper we combine atomic and molecular data, which are displayed in their periodic systems, in such a way as to take the discrete gradient. Then we act on the resulting vector field with the discrete vector divergence. The curl is obviously zero; the scalar field is conservative. We act on the original data with the discrete Laplacian operator (the iterated average utilizing only data on a border which contains the extreme values). The properties considered are atomic electronegativity, ionization potential and radius; and diatomic-molecular dissociation potential and internuclear separation. The calculus should work well to highlight the local energy minima of the nuclear valley of stability.
Similar content being viewed by others
References
Slater J.C., Frank N.H. (1947). Mechanics. McGraw-Hill, New York
Harnwell G.P. (1949). Principles of Electricity and Electromagnetism. McGraw-Hill, New York
Young H.D., Freedman R.A. (2000). University Physics. Addison Wesley Longman, San Francisco
Hefferlin R.A., Zhuvikin G.V., Caviness K.E., and Duerksen P.J., J. Quant. Sperctrosc. Radiat. Transfer 32 (1984) 257.
Hefferlin R. (1994). Chem J.. Inf. Comput. Sci. 34: 314
Periodic Table of the elements (chart), Sargent-Welch, New York, 1969.
http://www.lenntech.com/Periodic-chart-elements/electronegativity.htm
Handbook of Chemistry and Physics, Vol. 54 (Chemical Rubber Corporation Publishers, 1964).
Hefferlin R.A., Field Theory for Chemical Spaces, AMS/IP Studies in Advanced Mathematics, Vol. 13, 1999.
Fraga S., Karwowski J. (1976). Handbook of Atomic Data. Elsevier Scientific, Amsterdam
Latysheva V.A., Hefferlin R. (2004). J. Chem. Inf. Comput. Sci. 44: 1202
Hefferlin R., Numerical Solutions of the Laplace Equation in Chemical Spaces, DIMACS Series in Discrete Mathematics and Theoretical Computer Sciences, Vol. 51, 2000 (the caption of figure 1 is wrong and the section headings should refer to “Atoms”).
Campbell J.A. (1970). Chemical Systems. Freeman, San Francisco
Huber K.P., Herzberg G. (1979). Constants of Diatomic Molecules. Van Nostrand Reihold, New York
Hefferlin R. (2000). J. Mol. Structure (Theochem) 506: 71
Hefferlin R., Knoll J. (2000). J. Math. Chem. 28: 169
Hefferlin R., Kutzner M. (1981). J. Chem. Phys. 75: 1035
Hefferlin R., Periodic Systems of Molecules and their Relation to the Systematic Analysis of Molecular Data (Edwin Mellen Press, Lewiston, New York, 1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hefferlin, R. Discrete vector calculus on periodic systems of atoms and molecules. J Math Chem 43, 386–394 (2008). https://doi.org/10.1007/s10910-007-9251-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10910-007-9251-9