Abstract
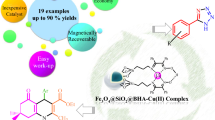
A highly efficient and sustainable ferrite supported palladium nanocatalyst (Pd(II)-Benz-Am-Fe3O4@SiO2) has been prepared and characterized by diverse microscopic and spectroscopic techniques. The Pd(II)-Benz-Am-Fe3O4@SiO2 was employed as magnetically recoverable nanocatalyst in the synthesis of symmetrical biaryls by reductive homocoupling of haloarenes (I, Br and Cl) using hydrazine hydrate (N2H4) as reducing agent. The Pd-Benz-Am-Fe3O4@SiO2 nanocatalyst exhibited high surface area and efficient magnetization as 45.25 m2/g and 34 emu/g, respectively. The Electron Dispersive X-ray (EDX) analysis confirmed 3.9 mol % loading of Pd in Pd-Benz-Am-Fe3O4@SiO2. Moverever, high turnover number (TON) and turnover frequency (TOF), operational simplicity and facial recyclability and reusability (up to eight runs) are the remarkable advantages of the nanocatalyst. The catalytic activity and stability of recycled Pd(II)-Benz-Am-Fe3O4@SiO2 catalyst was comprehensively studied from X- ray diffraction (XRD), Scanning Electron Microscopy (SEM), Transmission Electron Microscopic (TEM) and EDX analysis.
Graphical Abstract














Similar content being viewed by others
References
M. Szostak, N.J. Fazakerley, D. Parmar, D.J. Procter, Chem. Rev. 114, 5959 (2014)
A. Balanta, C. Godard, C. Claver, Chem. Soc. Rev. 40, 4973 (2011)
B. Yuan, Y. Pan, Y. Li, B. Yin, H. Jiang, Angew. Chem. Int. Ed. 49, 4054 (2010)
B. Karimi, H. Behzadnia, H. Vali, ChemCatChem 6, 745 (2014)
P. Puthiaraj, W.S. Ahn, Mol. Catal. 437, 73 (2017)
S.K. Movahed, M. Dabiri, A. Bazgir, Appl. Catal. Gen. 488, 265 (2014)
C. Sambiagio, S.P. Marsden, A.J. Blacker, P.C. McGowan, Chem. Soc. Rev. 43, 3525 (2014)
L. Guo, C. Huang, L. Liu, Z. Shao, Y. Tong, H. Hou, Y. Fan, Cryst. Growth Des. 16, 4926 (2016)
W.-W. Chen, Q. Zhao, M.-H. Xu, G.-Q. Lin, Org. Lett. 12, 1072 (2010)
H. Tran, T. McCallum, M. Morin, L. Barriault, Org. Lett. 18, 4308 (2016)
B.W. Crabbe, O.P. Kuehm, J.C. Bennett, G.L. Hallett-Tapley, Catal. Sci. Technol. 18, 4907 (2018)
D. Tyagi, C. Binnani, R.K. Rai, A.D. Dwivedi, K. Gupta, P.-Z. Li, Y. Zhao, S.K. Singh, Inorg. Chem. 55, 6332 (2016)
Q. Wu, Y. Han, Z. Shao, J. Li, H. Hou, Dalton Trans. 47, 8063 (2018)
M. Niakan, Z. Asadi, M. Emami, Catal. Lett. 150, 404 (2020)
P.K. Prajapati, S. Saini, S.L. Jain, J. Mater. Chem. A 8, 5246 (2020)
A. Kamal, V. Srinivasulu, B.N. Seshadri, N. Markandeya, A. Alarifi, N. Shankaraiah, Green Chem. 14, 2513 (2012)
K. Wang, L. Yang, W. Zhao, L. Cao, Z. Sun, F. Zhang, Green Chem. 19, 1949 (2017)
X. Gong, J. Wu, Y. Meng, Y. Zhang, L.-W. Ye, C. Zhu, Green Chem. 21, 995 (2019)
M. Busch, M.D. Wodrich, C. Corminboeuf, ACS Catal. 7, 5643 (2017)
C. Diner, M.G. Organ, Organometallics 38, 66 (2018)
M.J. Jin, D.H. Lee, Angew. Chem. Int. Ed. 49, 1119 (2010)
Y. Zhang, X. Gao, J. Li, G. Tu, J. Mater. Chem. C 3, 7463 (2015)
S. Song, X. Wang, S. Li, Z. Wang, Q. Zhu, H. Zhang, Chem. Sci. 6, 6420 (2015)
X. Li, L. Zhou, Y. Wei, A.M. El-Toni, F. Zhang, D. Zhao, J. Am. Chem. Soc. 137, 5903 (2015)
L. He, K. Natte, J. Rabeah, C. Taeschler, H. Neumann, A. Bruckner, M. Beller, Angew. Chem. Int. Ed. 54, 4320 (2015)
H. Kaur, D. Shah, U. Pal, Catal. Commun. 12, 1384 (2011)
J. González-Rivera, I.R. Galindo-Esquivel, M. Onor, E. Bramanti, I. Longo, C. Ferrari, Green Chem. 16, 1417 (2014)
T. Kawasaki, Y. Araki, K. Hatase, K. Suzuki, A. Matsumoto, T. Yokoi, Y. Kubota, T. Tatsumi, K. Soai, Chem. Commun. 51, 8742 (2015)
M. Cai, Q. Xu, Y. Huang, J. Mol. Catal. A Chem. 271, 93 (2007)
M. Mazur, A. Barras, V. Kuncser, A. Galatanu, V. Zaitzev, K.V. Turcheniuk, P. Woisel, J. Lyskawa, W. Laure, A. Siriwardena, R. Boukherrouba, S. Szunerits, Nanoscale 5, 2692 (2013)
S. Singamaneni, V.N. Bliznyuk, C. Binek, E.Y. Tsymbal, J. Mater. Chem. 21, 16819 (2011)
M.B. Gawande, A.K. Rathi, I.D. Nogueira, R.S. Varma, P.S. Branco, Green Chem. 15, 1895 (2013)
M.B. Gawande, P.S. Branco, R.S. Varma, Chem. Soc. Rev. 42, 3371 (2013)
O. Baudoin, F. Gueritte, Stud. Nat. Prod. Chem. 29, 355 (2003)
G. Bringmann, C. Gunther, M. Ochse, O. Schupp, S. Tasler, in Progress in the Chemistry of Organic Natural Products, vol. 82 (Springer, New York, 2001), p. 1
F. Bellina, A. Carpita, R. Rossi, Synthesis 15, 2419 (2004)
D.A. Horton, G.T. Bourne, L.M. Smythe, Chem. Rev. 103, 893 (2003)
M. Kertesz, C.H. Choi, S. Yang, Chem. Rev. 105, 3448 (2005)
X. Mei, C.J. Wolf, J. Am. Chem. Soc. 128, 13326 (2006)
S. Lightowler, M. Hird, Chem. Mater. 22, 5538 (2005)
J.M. Brunel, Chem. Rev. 105, 857 (2005)
M. Berthod, G. Mignani, G. Woodward, M. Lemaire, Chem. Rev. 105, 1801 (2005)
I.P. Beletskaya, A.V. Cheprakov, Coord. Chem. Rev. 248, 2337 (2004)
F. Alonso, I.P. Beletskaya, M. Yus, Tetrahedron 64, 3047 (2008)
N.T.S. Phan, M. Van Der Sluys, C.W. Jones, Adv. Synth. Catal. 348, 609 (2006)
D. Alberico, M.E. Scott, M. Lautens, Chem. Rev. 107, 174 (2007)
W. Miao, T.H. Chan, Org. Lett. 5, 5003 (2003)
J.D. Revell, A. Ganesan, Org. Lett. 4, 3071 (2002)
P.N.W. Baxter, J. Org. Chem. 64, 1257 (2000)
K.M. Hossain, T. Shibata, K. Takagi, Synlett 8, 1137 (2000)
J. Huang, S.P. Nolan, J. Am. Chem. Soc. 121, 9889 (1999)
A.C. Spivey, T. Fekner, S.E. Spey, J. Org. Chem. 65, 3154 (2000)
F. Ullman, Chem. Ber. 36, 2382 (1903)
P.E. Fanta, Chem. Rev. 64, 613 (1964)
P.E. Fanta, Chem. Rev. 38, 139 (1946)
F.M. Moghaddam, R. Pourkaveh, A. Karimi, S. Ebrahim Ayati, Asian. J. Org. Chem. 7, 802 (2018)
J. Chen, L. Dai, J. Li, M. Mohammadnia, Appl. Organomet. Chem. 34, e5708 (2020)
A.V. Dubey, A. Vijay Kumar, Appl. Organomet. Chem. 34, e5570–e5571 (2020)
S. Mukhopadhyay, G. Rothenberg, H. Wiener, Y. Sasson, Tetrahedron 55, 14763 (1999)
M. Kuroboshi, Y. Waki, H. Tanaka, J. Org. Chem. 68, 3938 (2003)
K. Abiraj, G.R. Srinivasa, C.D. Gowda, Tetrahedron Lett. 45, 2081 (2004)
J. Moon, H. Nam, J. Ju, M. Jeong, S. Lee, Chem. Lett. 36, 1432 (2007)
N. Ma, Z. Duan, Y. Wu, J. Organomet. Chem. 691, 5697 (2006)
R.N. Ram, V. Singh, Tetrahedron Lett. 47, 7625 (2006)
A. Monopoli, V. Calo, F. Ciminale, P. Cotugno, C. Angelici, N. Cioffi, A. Nacci. J. Org. Chem. 75, 3908 (2010)
S.P. Vibhute, P.M. Mhaldar, S.N. Korade, D.S. Gaikwad, R.V. Shejawal, D.M. Pore, Tetrahedron Lett. 41, 3643 (2018)
S.P. Vibhute, P.M. Mhaldar, R.V. Shejawal, D.M. Pore, Tetrahedron Lett. 11, 151594 (2020)
P.M. Mhaldar, S.P. Vibhute, G.S. Rashinkar, D.M. Pore, React. Funct. Polym. 152, 104586 (2020)
S.P. Vibhute, P.M. Mhaldar, D.S. Gaikwad, R.V. Shejwal, D.M. Pore, Monatsh. Chem. 151, 87 (2020)
B. Polshettiwar, R.S. Baruwati, Varma. Green Chem. 11, 127 (2009)
Q. Zhang, H. Su, J. Luo, Y. Wei, Green Chem. 14, 201 (2012)
C. Pereira, A.M. Pereira, P. Quaresma, P.B. Tavares, E. Pereira, J.P. Araújo, C. Freire, Dalton Trans. 39, 2842 (2010)
N.T.S. Phan, H.V. Le, J. Mol. Catal. A Chem. 334, 130 (2011)
V. Polshettiwar, R.S. Varma, Org. Biomol. Chem. 7, 37 (2009)
Z. Zhang, F. Zhang, Q. Zhu, W. Zhao, B. Ma, Y. Ding, J. Colloid Interf. Sci. 360, 189 (2011)
M. Yamaura, R.L. Camilo, L.C. Sampaio, M.A. Macedo, M. Nakamurad, H.E. Toma, J. Magn. Magn. Mater. 279, 210 (2004)
J. Wang, S. Zheng, Y. Shao, J. Liu, Z. Xu, D. Zhu, J. Colloid Interfaces Sci. 349, 293 (2010)
M.M. Farahani, N. Tayyebi, J. Mol. Catal. A Chem. 348, 83 (2011)
H. Veisi, P. Sarachegol, S. Hemmati, Tetrahedron 156, 30571 (2018)
K. Manjunatha, T.S. Koley, V. Kandathil, R.B. Dateer, G. Balakrishna, B.S. Sasidhar, S.A. Patil, S.A. Patil, Appl. Organomet. Chem. 32, e4266 (2018)
K. Vishal, B.D. Fahlman, B.S. Sasidhar, S.A. Patil, S.A. Pati, Catal. Lett. 147, 900 (2017)
Y. Liu, D. Tang, K. Cao, L. Yu, J. Han, Q. Xu, J. Catal. 360, 250 (2018)
V. Cal, A. Nacci, A. Monopoli, P. Cotugno, Chem. Eur. J. 15, 1272 (2009)
D.D. Hennings, T. Iwama, V.H. Rawal, Org. Lett. 8, 1205 (1999)
L. Shaoa, Y. Dub, M. Zenga, X. Lia, W. Shena, S. Zuoa, Y. Lua, X.-M. Zhanga, C. Qia, Appl. Organomet. Chem. 24, 421 (2010)
M.A. Rasouli, P.R. Ranjbar, Z. Nat. 68B, 946 (2013)
L. Lv, Z. Qiu, J. Li, M. Liu, C.-J. Li, Nat. Commun. 9, 4739 (2018)
J. Wang, Y. Li, P. Li, G. Song, Monatsh. Chem. 144, 1159 (2013)
Acknowledgements
One of the authors, Pradeep M. Mhaldar, is grateful to Council of Scientific and Industrial Research (CSIR), New Delhi, Government of India, for the award of the Junior Research Fellowship (File no. 09/816(0040)/2017-EMR-I).
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mhaldar, P.M., Patil, M.V., Rashinkar, G.S. et al. Magnetically Recoverable Palladium Nanocatalyst [Pd(II)-Benz-Am-Fe3O4@SiO2] for Ullmann Type Homocoupling of Aryl halides with N2H4 as an Efficient Reductant. J Inorg Organomet Polym 32, 3053–3066 (2022). https://doi.org/10.1007/s10904-022-02340-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-022-02340-x