Abstract
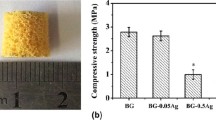
In this study, hexagonal boron nitride (hBN) nanoparticle- containing (0.1–2 wt%) polycaprolactone (PCL) and polylactic-co-glycolic acid (PLG)-coated 13-93B3 borate-based porous bioactive glass composite scaffolds were prepared by polymer foam replication method and their ability to use in bone tissue engineering applications was assessed. Morphological, mechanical properties, cytotoxicity and the drug release behavior of the prepared composite scaffolds were investigated. In vitro bioactivity was tested in simulated body fluid and results were analyzed using FTIR spectrometer and SEM. Results showed that both polymer coating and the existence of hBN nanoparticles in the polymeric matrix improved the compressive strength of the fabricated composite scaffolds. Incorporation of the hBN nanoparticles enhanced the in vitro hydroxyapatite forming ability of the glass composites. Results also revealed that prepared bioactive glass based composite scaffolds showed no toxicity to MC3T3-E1 cells under in vitro conditions up to 72 h and hBN-containing glass scaffolds showed higher gentamicin sulfate release rates compared to the bare polymer coated scaffolds. Manufactured bioactive glass scaffolds containing hBN nanoparticles are found to be promising for bone repair and regeneration.












Similar content being viewed by others
References
L.L. Hench, R.J. Splinter, W.C. Allen, T.K. Greenlee Jr., Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. 2, 117–141 (1971). https://doi.org/10.1002/jbm.820050611
L.L. Hench, J. Wilson, Surface active biomaterials. Science 226, 630–636 (1984). https://doi.org/10.1126/science.6093253
M.N. Rahaman, D.E. Day, B.S. Bal, Q. Fu, S.B. Jung, L.F. Bonewald, Bioactive glass in tissue engineering. Acta Biomater. 7(6), 2355–2373 (2011). https://doi.org/10.1016/j.actbio.2011.03.016
W. Liang, N.M. Rahaman, D. Day, N. Marion, G. Riley, J. Mao, Bioactive borate glass scaffold for bone tissue engineering. J. Non-Cryst. Solids 354, 1690–1696 (2008). https://doi.org/10.1016/j.jnoncrysol.2007.10.003
J. Ning, A. Yao, D. Wang, W. Huang, H. Fu, X. Liu, X. Jiang, X. Zhang, Synthesis and in vitro bioactivity of a borate-based bioglass. Mater. Lett. 61, 5223–5226 (2007). https://doi.org/10.1016/j.matlet.2007.04.089
W.-T. Jia, X. Zhang, S.-H. Luo, X. Liu, W.-H. Huang, N.M. Rahaman, D. Day, C.-Q. Zhang, Z.-P. Xie, J.-Q. Wang, Novel borate glass/chitosan composite as a delivery vehicle for teicoplanin in the treatment of chronic osteomyelitis. Acta Biomater. 6, 812–819 (2009). https://doi.org/10.1016/j.actbio.2009.09.011
R. Sergi, D. Bellucci, V. Cannillo, A review of bioactive glass/natural polymer composites: state of the art. Materials 13, 5560 (2002). https://doi.org/10.3390/ma13235560
A.M. Deliormanlı, Fabrication and characterization of poly caprolactone coated silicate and borate based bioactive glass composite scaffolds. J. Compos. Mater. 50(7), 917–928 (2016). https://doi.org/10.1177/0021998315583320
M. Türk, A.M. Deliormanlı, Graphene-containing PCL- coated porous 13–93B3 bioactive glass scaffolds for bone regeneration. Mater. Res. Express 5(4), 045406 (2018). https://doi.org/10.1088/2053-1591/aab87b
M. Jedrzejczak-Silicka, M. Trukawka, M. Dudziak, K. Piotrowska, E. Mijowska, Hexagonal boron nitride functionalized with Au nanoparticles—properties and potential biological applications. Nanomaterials 8, 605 (2018). https://doi.org/10.3390/nano8080605
A. Merlo, V.R.S.S. Mokkapati, S. Pandit, I. Mijakovic, Boron nitride nanomaterials: biocompatibility and bio-applications. Biomater. Sci. 6, 2298–2311 (2018). https://doi.org/10.1039/C8BM00516H
D. Golberg, Y. Bando, Y. Huang, T. Terano, M. Mitome, C. Tang, C. Zhi, Boron nitride nanotubes and nanosheets. ACS Nano 4, 2979–2993 (2010). https://doi.org/10.1021/nn1006495
H. Sediri, D. Pierucci, M. Hajlaoui, H. Henck, G. Patriarche, Y.J. Dappe, S. Yuan, B. Toury, R. Belkhou, M.G. Silly et al., Atomically sharp interface in an h-BN-epitaxial graphene van der Waals heterostructure. Sci. Rep. 5, 16465 (2015). https://doi.org/10.1038/srep16465
J. Wang, F. Ma, W. Liang, M. Sun, Electrical properties and applications of graphene, hexagonal boron nitride (h-BN), and graphene/h-BN heterostructures. Mater. Today Phys. 2, 6–34 (2017). https://doi.org/10.1016/j.mtphys.2017.07.001
J. Joy, E. George, P. Haritha, S. Thomas, S. Anas, An overview of boron nitride based polymer nanocomposites. J. Polym. Sci. 58(22), 3115–3141 (2020). https://doi.org/10.1002/pol.20200507
C. Zhi, B. Yoshio, W. Wang, C. Tang, K. Hiroaki, G. Dmitri, Mechanical and thermal properties of polymethyl methacrylate-BN nanotube composites. J. Nanomater. (2008). https://doi.org/10.1155/2008/642036
L. Hao, I.C. Chen, J.K. Oh, N. Nagabandi, F. Bassan, S. Liu, E. Scholar, L. Zhang, M. Akbulut, B. Jiang, Nanocomposite foam involving boron nitride nanoplatelets and polycaprolactone: porous structures with multiple length scales for oil spill cleanup. Ind. Eng. Chem. Res. 56, 14670 (2017). https://doi.org/10.1021/acs.iecr.7b03911
F. Bakan, M. Sezen, M. Geçgin, Y. Göncü, A.Y. Nuran, Structural and chemical analysis of hydroxyapatite (HA)-boron nitride (BN) nanocomposites sintered under different atmospheric conditions. Microsc. Microanal. 23, 1–9 (2017). https://doi.org/10.1017/S1431927617012405
S. Unal, N. Ekren, A.Z. Sengil, F.N. Oktar, S. Irmak, O. Oral, Y.M. Sahin, O. Kilic, S. Agathopoulos, O. Gunduz, Synthesis, characterization, and biological properties of composites of hydroxyapatite and hexagonal boron nitride. J. Biomed. Mater. Res. B 106(6), 2384–2392 (2018). https://doi.org/10.1002/jbm.b.34046
R. Saggar, H. Porwal, P. Tatarko, I. Dlouhý, M.J. Reece, Boron nitride nanosheets reinforced glass matrix composites. Adv. Appl. Ceram. 114(sup1), S26–S33 (2015). https://doi.org/10.1179/1743676115Y.0000000056
A.I. Rasel, T. Li, T.D. Nguyen, S. Singh, Y. Zhou, Y. Xiao, Y.T. Gu, Bio physical response of living cells to boron nitride nanoparticles: Uptake mechanism and bio-mechanical characterization. J. Nanopart. Res. 17, 441 (2015). https://doi.org/10.1007/s11051-015-3248-2
X. Chen, P. Wu, M. Rousseas, D. Okawa, Z. Gartner, A. Zettl, C.R. Bertozzi, Boron nitride nanotubes are noncytotoxic and can be functionalized for interaction with proteins and cells. J. Am. Chem. Soc. 131, 890–891 (2009). https://doi.org/10.1021/ja807334b
L. Horváth, A. Magrez, D. Golberg, C. Zhi, Y. Bando, R. Smajda, E. Horváth, L. Forró, B. Schwaller, In vitro investigation of the cellular toxicity of boron nitride nanotubes. ACS Nano 5, 3800–3810 (2011). https://doi.org/10.1021/nn200139h
T. Lu, L. Wang, Y. Jiang, Q. Liu, C. Huang, Hexagonal boron nitride nanoplates as emerging biological nanovectors and their potential applications in biomedicine. J. Mater. Chem. B 4(36), 6103–6110 (2016). https://doi.org/10.1039/C6TB01481J
K.Y. Gudz, E.S. Permyakova, A.T. Matveev, A.V. Bondarev, A.M. Manakhov, D.A. Sidorenko, S.Y. Filippovich, A.V. Brouchkov, D.V. Golberg, S.G. Ignatov, D.V. Shtansky, Pristine and antibiotic-loaded nanosheets/nanoneedles-based boron nitride films as a promising platform to suppress bacterial and fungal infections. ACS Appl. Mater. Interfaces 12(38), 42485–42498 (2020). https://doi.org/10.1021/acsami.0c10169
M. Ensoylu, A.M. Deliormanlı, H.A. İlhan, Tungsten disulfide nanoparticle-containing PCL and PLGA-coated bioactive glass composite scaffolds for bone tissue engineering applications. J. Mater. Sci. 56(33), 18650–18667 (2021). https://doi.org/10.1007/s10853-021-06494-w
T. Kokubo, H. Kushitani, S. Sakka, T. Kitsugi, T. Yamamuro, Solutions able to reproduce in vivo surface-structure change in bioactive glass-ceramic A-W. J Biomed. Mater. Res. 24, 721–734 (1990). https://doi.org/10.1002/jbm.820240607
E. Altun, E. Yuca, N. Ekren, D.M. Kalaskar, D. Ficai, G. Dolete, A. Ficai, O. Gunduz, Kinetic release studies of antibiotic patches for localtransdermal delivery. Pharmaceutics 13, 613 (2021). https://doi.org/10.3390/pharmaceutics13050613
S. Dash, P.N. Murthy, L. Nath, P. Chowdhury, Kinetic modeling on drug release from controlled drug delivery systems. Acta Poloniae Pharmaceutica Drug Res. 67(3), 217–223 (2010)
D.W. Bourne, Pharmacokinetics, in Modern Pharmaceutics, 4th edn., ed. by G.S. Banker, C.T. Rhodes (Marcel Dekker Inc, New York, 2002)
C. Du, H. Ma, M. Ruo, Z. Zhang, X. Yu, Y. Zeng, An experimental study on the biomechanical properties of the cancellous bones of distal femur. Biomed. Mater. Eng. 16, 215–222 (2006)
D. Lahiri, F. Rouzaud, T. Richard, A.K. Keshri, S.R. Bakshi, L. Kos, A. Agarwal, Boron nitride nanotube reinforced polylactide–polycaprolactone copolymer composite: mechanical properties and cytocompatibility with osteoblasts and macrophages in vitro. Acta Biomater. 6(9), 3524–3533 (2010). https://doi.org/10.1016/j.actbio.2010.02.044
S. Nagarajan, H. Belaïd, C. Pochat-Bohatier, C. Teyssier, I. Iatsunskyi et al., Design of boron nitride/gelatin electrospun nanofibers for bone tissue engineering. ACS Appl. Mater. Interfaces 9(39), 33695–33706 (2017). https://doi.org/10.1021/acsami.7b13199
R.F. Brown, M.N. Rahaman, A.B. Dwilewicz, W. Huang, D.E. Day, Y. Li, B.S. Bal, Effect of borate glass composition on its conversion to hydroxyapatite and on the proliferation of MC3T3-E1 cells. J. Biomed. Mater. Res. Part A (2007). https://doi.org/10.1002/jbm.a.31679
J. Pawlik, K. Jukowicz, K. Cholewa-Kowalska, A.M. Osyczka, New insights into the PLGA and PCL blending: physico-mechanical properties and cell response. Mater. Res. Express 6, 085344 (2019). https://doi.org/10.1088/2053-1591/ab2823
S.J. Gadaleta, E.P. Paschalis, F. Betts, R. Mendelsohn, A.L. Boskey, Fourier transform infrared spectroscopy of the solution-mediated conversion of amorphous calcium phosphate to hydroxyapatite: New correlations between X-ray diffraction and infrared data. Calcif. Tissue Int. 58, 9–16 (1996). https://doi.org/10.1007/BF02509540
S. Gunasekaran, G. Anbalagan, S. Pandi, Raman and infrared spectra of carbonates of calcite structure. J. Raman Spectrosc. 37, 892–899 (2006). https://doi.org/10.1002/jrs.1518
K.W. Chan, H.M. Wong, K.W.K. Yeung, S.C. Tjong, Polypropylene biocomposites with boron nitride and nano-hydroxyapatite reinforcements. Materials 8, 992–1008 (2015). https://doi.org/10.3390/ma8030992
D. Lahiri, V. Singh, A.K. Keshri, S. Seal, A. Agarwal, Apatite formability of boron nitride nanotubes. Nanotechnology 22, 205601 (2011). https://doi.org/10.1088/0957-4484/22/20/205601
H.-L. Nguyen, Z. Hanif, S.-A. Park, B.G. Choi, T.H. Tran, D.S. Hwang, J. Park, S.Y. Hwang, D.X. Oh, Sustainable boron nitride nanosheet-reinforced cellulose nanofiber composite film with oxygen barrier without the cost of color and cytotoxicity. Polymers 10(5), 501 (2018). https://doi.org/10.3390/polym10050501
S.M. Sharker, Hexagonal boron nitrides (white graphene): a promising method for cancer drug delivery. Int. J. Nanomed. 14, 9983–9993 (2019). https://doi.org/10.2147/IJN.S205095
E. Melis, S. Özlem, T.I. Çulha, Ç. Mustafa, Synthesis, functionalization, and bioapplications of two-dimensional boron nitride nanomaterials. Front. Bioeng. Biotechnol. 7, 363 (2019). https://doi.org/10.3389/fbioe.2019.00363
D.R. Paul, Elaborations on the Higuchi model for drug delivery. Int. J. Pharm. 418, 13–17 (2010). https://doi.org/10.1016/j.ijpharm.2010.10.037
Acknowledgements
The financial support for this research was provided by the Scientific and Technical Research Council of Turkey (TUBITAK), Short‐Term R&D Funding Program, Grant No.: 119M935.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ensoylu, M., Deliormanlı, A.M. & Atmaca, H. Hexagonal Boron Nitride/PCL/PLG Coatings on Borate Bioactive Glass Scaffolds for Bone Regeneration. J Inorg Organomet Polym 32, 1551–1566 (2022). https://doi.org/10.1007/s10904-022-02246-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-022-02246-8