Abstract
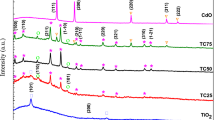
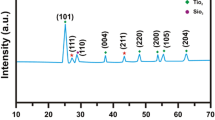
In this work, has been studied the effects of CdS nanoparticles at different concentrations on the microstructure and optical properties of TiO2/CdS nanocomposites (T-CdS). The structural properties of T-CdS samples were characterized by using X-ray diffraction (XRD), transmission electron microscopy (TEM). XRD analysis confirms the hexagonal wurtzite phase structure of CdS nanoparticles and the tetragonal anatase phase of TiO2 nanoparticles. TEM images appear the shapes and sizes of TiO2, CdS nanoparticles and T-CdS nanocomposite, where found in aggregates nanoparticles their sizes from 15 to 56 nm. By UV–vis diffuse reflectance spectrum (DRS) were determined Band-gap energies of the samples. The Photoluminescence (PL) analysis exposed, with the excitation wavelength of 285 nm, our samples exhibited the intensity of peak at 426 nm. Additionally, the FTIR spectroscopy of the samples exhibits the presence of functional groups. The thermal stability of T-CdS nanocomposite samples were determined by thermogravimetric and differential thermal analysis.










Similar content being viewed by others
References
M. Rafatullah, O. Sulaiman, R. Hashim, A. Ahmad, Adsorption of methylene blue on low-cost adsorbents: a review. J. Hazard Mater. 177, 70–80 (2010). https://doi.org/10.1016/j.jhazmat.2009.12.047
M.T. Yagub, T.K. Sen, S. Afroze, H.M. Ang, Dye and its removal from aqueous solution by adsorption: a review. Adv. Colloid Interface Sci. 209, 172–184 (2014). https://doi.org/10.1016/j.cis.2014.04.002
A. Houas, H. Lachheb, M. Ksibi, E. Elaloui, C. Guillard, J.M. Herrmann, Photocatalytic degradation pathway of methylene blue in water. Appl. Catal. B 31, 145–157 (2001). https://doi.org/10.1016/S0926-3373(00)00276-9
P. Zheng, J. Zhao, J. Zheng, G. Ma, Z. Zhu, Non-equilibrium partial oxidation of TiN surface for efficient visible-light-driven hydrogen production. J. Mater. Chem. 22, 12116–12120 (2012). https://doi.org/10.1039/c2jm30662j
Z. Xie, X. Liu, P. Zhan, W. Wang, Z. Zhang, Tuning the optical bandgap of TiO2-TiN composite films as photocatalyst in the visible light. AIP Adv. 3, 062129 (2013). https://doi.org/10.1063/1.4812702
A. Kaur, A. Umar, W.A. Anderson, S.K. Kansal, Facile synthesis of CdS/TiO2 nanocomposite and their catalytic activity for ofloxacin degradation under visible illumination. J. Photochem. Photobiol. A 360, 34–43 (2018). https://doi.org/10.1016/j.jphotochem.2018.04.021
F. Chen, S.-W. Wang, L. Yu, X. Chen, W. Lu, Control of optical properties of TiNxOy films and application for high performance solar selective absorbing coatings. Opt. Mater. Express 4, 1833 (2014). https://doi.org/10.1364/ome.4.001833
B. Li, Z. Zhao, F. Gao, X. Wang, J. Qiu, Mesoporous microspheres composed of carbon-coated TiO2 nanocrystals with exposed {001} facets for improved visible light photocatalytic activity. Appl. Catal. B 147, 958–964 (2014). https://doi.org/10.1016/j.apcatb.2013.10.027
R. Campostrini, M. Ischia, L. Palmisano, Pyrolysis study of sol-gel derived TiO2 powders. Part III. TiO2-anatase prepared by reacting titanium(IV) isopropoxide with acetic acid. J. Therm. Anal. Calorim. 75, 13–24 (2004). https://doi.org/10.1023/B:JTAN.0000017324.05515.b9
H. Ghassemi, W. Harlow, O. Mashtalir, M. Beidaghi, M.R. Lukatskaya, Y. Gogotsi, M.L. Taheri, In situ environmental transmission electron microscopy study of oxidation of two-dimensional Ti3C2 and formation of carbon-supported TiO2. J. Mater. Chem. A 2, 14339–14343 (2014). https://doi.org/10.1039/c4ta02583k
C.X. Tian, J.Q. Du, X.H. Chen, W.P. Ma, Z.Q. Luo, X.Z. Cheng, H.F. Hu, D.J. Liu, Influence of hydrolysis in sulfate process on titania pigment producing. Trans. Nonferr. Metals Soc. China (Engl. Ed.) 19, s829–s833 (2009). https://doi.org/10.1016/S1003-6326(10)60160-4
S.Y. Chae, M.K. Park, S.K. Lee, T.Y. Kim, S.K. Kim, W.I. Lee, Preparation of size-controlled TiO2 nanoparticles and derivation of optically transparent photocatalytic films. Chem. Mater. 15, 3326–3331 (2003). https://doi.org/10.1021/cm030171d
Y.F. You, C.H. Xu, S.S. Xu, S. Cao, J.P. Wang, Y.B. Huang, S.Q. Shi, Structural characterization and optical property of TiO2 powders prepared by the sol-gel method. Ceram. Int. 40, 8659–8666 (2014). https://doi.org/10.1016/j.ceramint.2014.01.083
H. Xu, J. Jia, S. Zhao, P. Chen, Q. Xia, J. Wu, P. Zhu, Hydrophobic TiO2-SiO2 aerogel composites for fast removal of organic pollutants. ChemistrySelect 3, 10483–10490 (2018). https://doi.org/10.1002/slct.201801646
V.G. Parale, H.N.R. Jung, W. Han, K.Y. Lee, D.B. Mahadik, H.H. Cho, H.H. Park, Improvement in the high temperature thermal insulation performance of Y2O3 opacified silica aerogels. J. Alloys Compd. 727, 871–878 (2017). https://doi.org/10.1016/j.jallcom.2017.08.189
I. Lázár, J. Kalmár, A. Peter, A. Szilágyi, E. Gyori, T. Ditrói, I. Fábián, Photocatalytic performance of highly amorphous titania-silica aerogels with mesopores: the adverse effect of the in situ adsorption of some organic substrates during photodegradation. Appl. Surf. Sci. 356, 521–531 (2015). https://doi.org/10.1016/j.apsusc.2015.08.113
G. Zu, J. Shen, W. Wang, L. Zou, Y. Lian, Z. Zhang, Silica-titania composite aerogel photocatalysts by chemical liquid deposition of titania onto nanoporous silica scaffolds. ACS Appl. Mater. Interfaces 7, 5400–5409 (2015). https://doi.org/10.1021/am5089132
Y.N. Kim, G.N. Shao, S.J. Jeon, S.M. Imran, P.B. Sarawade, H.T. Kim, Sol-gel synthesis of sodium silicate and titanium oxychloride based TiO2TiO2-SiO2 aerogels and their photocatalytic property under UV irradiation. Chem. Eng. J. 231, 502–511 (2013). https://doi.org/10.1016/j.cej.2013.07.072
S. Cao, N. Yao, K.L. Yeung, Synthesis of freestanding silica and titania-silica aerogels with ordered and disordered mesopores. J. Sol-Gel Sci. Technol. 46, 323–333 (2008). https://doi.org/10.1007/s10971-008-1701-8
N. Yao, S. Cao, K.L. Yeung, Mesoporous TiO2-SiO2 aerogels with hierarchal pore structures. Microporous Mesoporous Mater. 117, 570–579 (2009). https://doi.org/10.1016/j.micromeso.2008.08.020
T.C. Dang, D.L. Pham, H.C. Le, V.H. Pham, TiO2/CdS nanocomposite films: fabrication, characterization, electronic and optical properties. Adv. Nat. Sci. 1, 1689–1699 (2010). https://doi.org/10.1088/2043-6254/1/1/015002
B. Gomathi Thanga Keerthana, P. Murugakoothan, Synthesis and characterization of CdS/TiO2 nanocomposite: methylene blue adsorption and enhanced photocatalytic activities. Vacuum. 159, 476–481 (2019). https://doi.org/10.1016/j.vacuum.2018.10.082
M. Madani, K. Omri, N. Fattah, A. Ghorbal, X. Portier, Influence of silica ratio on structural and optical properties of SiO2/TiO2 nanocomposites prepared by simple solid-phase reaction. J. Mater. Sci. 28, 12977–12983 (2017). https://doi.org/10.1007/s10854-017-7129-6
N. Alonizan, S. Rabaoui, K. Omri, R. Qindeel, Microstructure and luminescence properties of ZnO:Mn nano-particles and ZnO:Mn/TiO2 nano-composite synthesized by a two-step chemical method. Appl. Phys. A 124, 710 (2018). https://doi.org/10.1007/s00339-018-2127-y
K. Omri, I. Najeh, LEl. Mir, Influence of annealing temperature on the microstructure and dielectric properties of ZnO nanoparticles. Ceram. Int. 42, 8940–8948 (2016). https://doi.org/10.1016/j.ceramint.2016.02.151
K. Deka, M.P.C. Kalita, Microstructure analysis of chemically synthesized wurtzite-type CdS nanocrystals. Pramana 86, 1119–1126 (2016). https://doi.org/10.1007/s12043-015-1132-3
D. Wang, B. Yu, F. Zhou, C. Wang, W. Liu, Synthesis and characterization of anatase TiO2 nanotubes and their use in dye-sensitized solar cells. Mater. Chem. Phys. 113, 602–606 (2009). https://doi.org/10.1016/j.matchemphys.2008.08.011
V. Loryuenyong, N. Jarunsak, T. Chuangchai, A. Buasri, The photocatalytic reduction of hexavalent chromium by controllable mesoporous anatase TiO2 nanoparticles. Adv. Mater. Sci. Eng. (2014). https://doi.org/10.1155/2014/348427
K. Omri, N. Alonizan, Effects of ZnO/Mn concentration on the micro-structure and optical properties of ZnO/Mn–TiO2 nano-composite for applications in photo-catalysis. J. Inorg. Organomet. Polym. Mater. 29, 203–212 (2019). https://doi.org/10.1007/s10904-018-0979-4
S. Saravanan, M. Balamurugan, T. Soga, Synthesis of titanium dioxide nanoparticles with desired ratio of anatase and rutile phases and the effect of high temperature annealing. Trans. Mater. Res. Soc. Jpn. 43, 255–261 (2018). https://doi.org/10.14723/tmrsj.43.255
T. Homann, T. Bredow, K. Jug, Adsorption of small molecules on the anatase (1 0 0) surface. Surf. Sci. 555, 135–144 (2004). https://doi.org/10.1016/j.susc.2003.12.039
J. Pei, W. Ma, R. Li, Y. Li, H. Du, Preparation and photocatalytic properties of TiO2-Al2O3 composite loaded catalysts. J. Chem. (2015). https://doi.org/10.1155/2015/806568
S.Y. Ha, H.J. Kim, S. Nam, S.Y. Oh, C. Lim, I.G. Kim, D.S. Yoo, S.Y. Park, One-step synthesis of TiO2/CdS nanocomposites by using microwave irradiation of a TiO2 + Cd2+-mercaptopropionic acid aqueous solution. J. Korean Phys. Soc. 64, 436–442 (2014). https://doi.org/10.3938/jkps.64.436
A.L. Patterson, The scherrer formula for X-ray particle size determination. Phys. Rev. 56, 978–982 (1939). https://doi.org/10.1103/PhysRev.56.978
G.K. Williamson, W.H. Hall, X-ray line broadening from filed aluminium and wolfram. Acta Metall. 1, 22–31 (1953). https://doi.org/10.1016/0001-6160(53)90006-6
A. Khorsand Zak, W.H. Abd, M.E. Majid, R. Abrishami, Yousefi, X-ray analysis of ZnO nanoparticles by Williamson-Hall and size-strain plot methods. Solid State Sci. 13, 251–256 (2011). https://doi.org/10.1016/j.solidstatesciences.2010.11.024
M. Manickam, V. Ponnuswamy, C. Sankar, R. Mariappan, R. Suresh, Influence of substrate temperature on the properties of cobalt oxide thin films prepared by nebulizer spray pyrolysis (NSP) technique. Silicon 8, 351–360 (2016). https://doi.org/10.1007/s12633-015-9316-5
N. Serpone, D. Lawless, R. Khairutdinov, Size effects on the photophysical properties of colloidal anatase TiO2 particles: size quantization or direct transitions in this indirect semiconductor? J. Phys. Chem. 99, 16646–16654 (1995). https://doi.org/10.1021/j100045a026
H. Tang, F. Lévy, H. Berger, P.E. Schmid, Urbach tail of anatase TiO2. Phys. Rev. B. 52, 7771–7774 (1995). https://doi.org/10.1103/PhysRevB.52.7771
V.D.-M.Ž Barbarić-Mikočević, K. Itrić, Kubelka-Munk theory in describing optical properties of paper (I). Tech. Gaz. 18, 117–124 (2011)
G.H. Meetent, P. Wood, Optical fibre methods for measuring the diffuse reflectance of fluids. Eng. Opt. 6, 331–336 (1993)
Z. Jiang, K. Qian, C. Zhu, H. Sun, W. Wan, J. Xie, H. Li, P.K. Wong, S. Yuan, Carbon nitride coupled with CdS-TiO2 nanodots as 2D/0D ternary composite with enhanced photocatalytic H2 evolution: a novel efficient three-level electron transfer process. Appl. Catal B 210, 194–204 (2017). https://doi.org/10.1016/j.apcatb.2017.03.069
G. He, Y. Zhang, Q. He, MoS2 /CdS heterostructure for enhanced photoelectrochemical performance under visible light. Catalysts 9, 19–21 (2019). https://doi.org/10.3390/catal9040379
K. Omri, A. Bettaibi, K. Khirouni, L. El, Mir, The optoelectronic properties and role of Cu concentration on the structural and electrical properties of Cu doped ZnO nanoparticles. Physica B 537, 167–175 (2018). https://doi.org/10.1016/j.physb.2018.02.025
G. Rajakumar, A.A. Rahuman, S.M. Roopan, V.G. Khanna, G. Elango, C. Kamaraj, A.A. Zahir, K. Velayutham, Fungus-mediated biosynthesis and characterization of TiO2 nanoparticles and their activity against pathogenic bacteria. Spectrochim. Acta A 91, 23–29 (2012). https://doi.org/10.1016/j.saa.2012.01.011
P.C. Dey, R. Das, Ligand free surface of CdS nanoparticles enhances the energy transfer efficiency on interacting with Eosin Y dye – helping in the sensing of very low level of chlorpyrifos in water. Spectrochim. Acta A 207, 156–163 (2019). https://doi.org/10.1016/j.saa.2018.09.014
N.S. Kumar, S.K.N. Kumar, L. Yesappa, Structural, optical and conductivity study of hydrothermally synthesized TiO2 nanorods. Mater. Res. Exp. 7, 0–11 (2020). https://doi.org/10.1088/2053-1591/ab691f
M.L. Hu, M.H. Fang, C. Tang, T. Yang, Z.H. Huang, Y.G. Liu, X.W. Wu, X. Min, The effects of atmosphere and calcined temperature on photocatalytic activity of TiO2 nanofibers prepared by electrospinning. Nanoscale Res. Lett. 8, 1–9 (2013). https://doi.org/10.1186/1556-276X-8-548
Acknowledgements
This study is a part of a project from The Deanship of Scientific Research, Imam Abdulrahman Bin Faisal University, Saudi Arabia (To Dr Norah Alonizan, Grant No: 2019-083-Sc).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alonizan, N. Effects of CdS Nanoparticles on the Physical Properties of T-CdS Nanocomposite Materials. J Inorg Organomet Polym 31, 1086–1094 (2021). https://doi.org/10.1007/s10904-020-01722-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-020-01722-3