Abstract
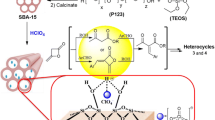
This work concerns the preparation and modification of mesoporous silica SBA-15 with different amine-copper complexes in order to study their impact on the catalytic behavior via Aza Michael addition. Firstly, SBA-15 was prepared hydrothermally, then functionalized by mono-, di- and tri-amine, and in the second step, copper (II) was anchored. The catalytic activity of these solids was evaluated at room temperature under liquid-phase conditions. Effects of catalyst nature, catalyst mass, reaction time and the starting reagents nature were investigated. Cu@SBA-15 (non-aminated) was shown to be efficient in the Aza Michael addition, but after its second re-use, a progressive decrease in the product yield was obtained due to copper leaching. The catalytic behavior of all prepared catalysts showed a selective reaction via the mono-Aza-Michael addition. Amongst all the catalysts, the one containing the monoamine-copper complex led to the highest yield. This catalyst was used in four consecutive cycles without significant loss of activity, which confirms its stability.










Similar content being viewed by others
References
B. Boukoussa, A. Hakiki, S. Moulai, K. Chikh, D.E. Kherroub, L. Bouhadjar, D. Guedal, K. Messaoudi, F. Mokhtar, R. Hamacha, J. Mater. Sci. 53, 7372–7386 (2018)
B. Boukoussa, R. Hamacha, A. Morsli, Arab. J. Chem. 10, S2160–S2169 (2017)
B. Boukoussa, S. Zeghada, G. Bentabed Ababsa, R. Hamachaa, A. Derdour, A. Bengueddach, F. Mongin, Appl. Catal. A 489, 131–139 (2015)
R. Ouargli-Saker, N. Bouazizi, B. Boukoussa, D. Barrimo, A.P. Nunes-Beltrao, A. Azzouz, Appl. Surf. Sci. 411, 476–486 (2017)
A. Hakiki, B. Boukoussa, H. Habib Zahmani, R. Hamacha, N.E.H. Hadj Abdelkader, F. Bekkar, F. Bettahar, A.P. Nunes-Beltrao, S. Hacini, A. Bengueddach, A. Azzouz, Mater. Chem. Phys. 212, 415–425 (2018)
D.X. Martínez-Vargas, J. Rivera De La Rosa, L. Sandoval-Rangela Jorge, L. Guzmán-Mar, M.A. Garza-Navarro, C.J. Lucio-Ortiz, D.A. De Haro-Del Río, Appl. Catal. A 547, 132–145 (2017)
Z. Talha, C. Bachir, S. Ziri, S. Bellahouel, A. Bengueddach, F. Villièras, M. Pelletier, P.G. Weidler, R. Hamacha, Catal. Lett. 147, 2116–2126 (2017)
I. Terrab, R. Ouargli, B. Boukoussa, K. Ghomari, R. Hamacha, R. Roy, A. Azzouz, A. Bengueddach, Res. Chem. Intermed. 43, 3775–3786 (2017)
X. Zhang, L. Zhang, H. Peng, X. You, C. Peng, X. Xu, W. Liu, X. Fang, Z. Wang, N. Zhang, X. Wang, Appl. Catal. B 224, 488–499 (2018)
A. Wach, M. Drozdek, B. Dudek, E. Szneler, P. Kuśtrowski, Catal. Commun. 64, 52–57 (2015)
L. Marcoux, J. Florek, F. Kleitz, Appl. Catal. A 504, 493–503 (2015)
Q. Xina, A. Papavasiliou, N. Boukos, A. Glisenti, J.P. Ho Li, Y. Yang, C.J. Philippopoulos, E. Poulakis, F.K. Katsaros, V. Meynen, P. Cool, Appl. Catal. B 223, 103–115 (2018)
X. Song, S. Li, K. Li, P. Ning, C. Wang, X. Sun, Y. Wang, Microporous Mesoporous Mater. 259, 89–98 (2018)
G.V. Shanbhag, T. Joseph, S.B. Halligudi, J. Catal. 250, 274–282 (2007)
M. Boutros, G. Moarbess, T. Onfroy, F. Launay, C. R. Chim. 21, 514–522 (2018)
G.R. Bardajee, R. Malakooti, F. Jami, Z. Parsaei, H. Atashin, Catal. Commun. 27, 49–53 (2012)
G. Anbarasu, M. Malathy, P. Karthikeyan, R. Rajavel, J. Solid State Chem. 253, 305–312 (2017)
P. Bhanja, R. Gomes, L. Satyanarayana, A. Bhaumik, J. Mol. Catal. A 415, 104–112 (2016)
H. Veisi, M. Hamelian, S. Hemmati, A. Dalvand, Tetrahedron Lett. 58, 4440–4446 (2017)
Z. Dong, Z. Dong, J. Ren, J. Jin, P. Wang, J. Jiang, R. Li, J. Ma, Microporous Mesoporous Mater. 135, 170–177 (2010)
M. Yadavi, A. Badiei, G. Mohammadi Ziarani, Appl. Surf. Sci. 279, 121–128 (2013)
H. Golchin Hosseini, E. Doustkhah, M.V. Kirillova, S. Rostamnia, G. Mahmoudi, A.M. Kirillov, Appl. Catal. A 548, 96–102 (2017)
N. Malumbazo, S.F. Mapolie, J. Mol. Catal. A 312, 70–77 (2009)
S. Roy, T. Chatterjee, M. Pramanik, A. Singha Roy, A. Bhaumik, Sk.M. Islam, J. Mol. Catal. A 386, 78–85 (2014)
Y. Yang, J. Guan, P. Qiu, Q. Kan, Appl. Surf. Sci. 256, 3346–3351 (2010)
Y. Yang, Y. Zhang, S. Hao, J. Guan, H. Ding, F. Shang, P. Qiu, Q. Kan, Appl. Catal. A 381, 274–281 (2010)
Y. Yang, Y. Zhang, S. Hao, Q. Kan, Chem. Eng. J. 171, 1356–1366 (2011)
J. Mondal, S. Sreejith, P. Borah, Y. Zhao, ACS Sustain. Chem. Eng. 3, 934 (2014)
A. Narani, R.K. Marella, P. Ramudu, K.S. Rama Rao, D.Raju Burri, RSC Adv. 4, 3774 (2014)
D. An, Q. Zhang, Y. Wang, Catal. Today 157, 143–148 (2010)
A.Y. Rulev, Russ. Chem. Rev. 80, 197 (2011)
B. Das, N. Chowdhury, J. Mol. Catal. A 263, 212–215 (2007)
P.A. Esteves, M. Esloa, L.M. Rodriguez, A. Olivia-compost, H. Radim, Tetrahedron Lett. 48, 9040–9043 (2007)
M. Dewan, A. De, S. Mozumdar, Inorg. Chem. Commun. 53, 92–96 (2015)
L. Li, Z. Liu, Q. Ling, X. Xing, J. Mol. Catal. A 353–354, 178–184 (2012)
L. Dai, Y. Zhang, Q. Dou, X. Wang, Y. Chen, Tetrahedron. 69, 1712–1716 (2013)
M.L. Kantam, M. Roy, S. Roy, B. Sreedhar, R.L. De, Chem. Commun. 9, 2226–2230 (2008)
L.T.L. Nguyen, T.T. Nguyen, K.D. Nguyen, N.T.S. Phan, Appl. Catal. A 425–426, 44–52 (2012)
Y. Wang, Y.Q. Yuan, S.R. Guo, Molecules. 14, 4779–4789 (2009)
S.L. Xie, Y.H. Hui, X.J. Long, C.C. Wang, Z.F.C. Xie, Chem. Lett. 24, 28–30 (2013)
M.L. Kantam, S. Laha, J. Yadav, S. Jha, Tetrahedron Lett. 50, 4467–4469 (2009)
P. Kalita, C.D. Pegu, P. Dutta, P.K. Baruah, J. Mol. Catal. A 394, 145–150 (2014)
H. Sekkiou, B. Boukoussa, R. Ghezini, Z. Khenchoul, A. Ouali, R. Hamacha, A. Bengueddach, Mater. Res. Express. 3, 085501 (2016)
H. Hadj Mokhtar, B. Boukoussa, R. Hamacha, A. Bengueddach, D.El Abed, RSC Adv. 5, 93438 (2015)
S. Das, T. Maity, S. Koner, Appl. Catal. A 513, 53–66 (2016)
M.L. Kantam, B. Neelima, C.V. Reddy, J. Mol. Catal. A 241, 147–150 (2005)
M.R. Saidi, Y. Pourshojaei, F. Aryanasab, Int. J. Rapid Commun. Synth. Org. Chem. 39, 1109–1119 (2009)
S. Kim, S. Kang, G. Kim, Y. Lee, J. Org. Chem. 81, 4048–4057 (2016)
V. Choudhary, D. Dumbre, S. Patil, RSC Adv. 2, 7061–7065 (2012)
S. Rostamnia, H. Alamgholiloo, Catal. Lett. 148, 2918–2928 (2018)
M.L. Kantam, V. Neeraja, B. Kavita, B. Neelima, K. Mihir, S. Hussain, Adv. Synth. Catal. 347, 763–766 (2005)
S.M. Sarkar, T. Sultana, T.K. Biswas, M.L. Rahman, M.M. Yusoff, New J. Chem. 40, 497–502 (2016)
L. Zhen-Xing, L. Dan, L. Ming-Ming, X. Xiao-Fei, M. Zheng-Zheng, X. Hao, Catalysts 7, 219 (2017)
K. Rajender Reddy, S.N. Kumar, Synlett. 2246–2250 (2006)
Y. Yamamoto, J. Org. Chem. 72, 7817 (2007)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hakiki, A., Kerbadou, R.M., Boukoussa, B. et al. Catalytic behavior of copper–amine complex supported on mesoporous silica SBA-15 toward mono-Aza-Michael addition: role of amine groups. J Inorg Organomet Polym 29, 1773–1784 (2019). https://doi.org/10.1007/s10904-019-01139-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-019-01139-7