Abstract
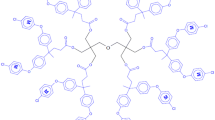
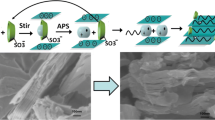
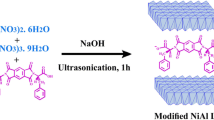
In the quest for new materials, intercalated nanocomposites consisting of molybdenum disulfide (MoS2) and an organometallic dendrimer were synthesized by exploiting the exfoliating/restacking properties of lithiated MoS2 (LiMoS2). By changing the molar ratio of the dendrimer to the LiMoS2, the amount of dendrimer intercalated within the restacked MoS2 was controlled. The molar ratio of the dendrimer to the LiMoS2 influenced the interlayer spacing of the intercalated material. For instance, we found an increase in the interlayer spacing of the nanocomposite when the mole ratio of dendrimer to LiMoS2 in the reaction mixture was changed from 1:1 to 2:1. In addition, the average size of the crystallite decreased as the mole ratio of the dendrimer to LiMoS2 was increased. Specifically, a change from 1:2 to 2:1 resulted in a noticeable decrease from 313 to 175 Å. The reported nanocomposites are unique being the first system derived from MoS2 and an organometallic dendrimer.






Similar content being viewed by others
References
R. Bissessur, B.D. Wagner, R. Brüning, J. Mater. Sci. 39(1), 119–125 (2004).
D.J. Harris, T.J. Bonagamba, K. Schmidt-Rohr, Macromolecules 32, 6718–6724 (1999)
R. Bissessur, W. White, Mater. Chem. Phys. 99, 214–219 (2006)
R. Bissessur, R.I. Haines, R. Brüning, J. Mater. Chem. 13, 44–49 (2003)
X. Wang, X. Shen, Z. Wang, R. Yu, L. Chen, ACS Nano 8, 11394–11400 (2014)
M.A. Py, R.R. Haering, Can. J. Phys. 61, 76–84 (1983)
M. Kertesz, R. Hoffmann, J. Am. Chem. Soc. 106, 3453–3460 (1984)
R.A. Gordon, D. Yang, E.D. Crozier, D.T. Jiang, R.F. Frindt. Phys. Rev. B. 65, 125407/1–125407/9 (2002).
H. Xu, R. Bissessur, D.C. Dahn, J. Inorg. Organomet. Polym. Mater. 24, 219–225 (2014)
R. Bissessur, H. Xu, Mater. Chem. Phys. 117, 335–337 (2009)
L. Wang, C. Wang, W. Liu, Q. Chen, M. He, Tetrahedron Lett. 57, 1771–1774 (2016)
Y.A. Kabachii, A.S. Golub, S.Y. Kochev, N.D. Lenenko, S.S. Abramchuk, M.Y. Antipin, P.M. Valetsky, B.D. Stein, W.E. Mahmoud, A.A. Al-Ghamdi, L.M. Bronstein, Chem. Mater. 25, 2434–2440 (2013)
S.-M. Paek, H. Jung, M. Park, J.-K. Lee, J.-H. Choy, Chem. Mater. 17, 3492–3498 (2005)
D. Astruc, F. Chardac, Chem. Rev. 101, 2991–3023 (2001)
E.U. Ikhuoria, A.U. Liyanage, M.M. Lerner, Mater. Chem. Phys. 139, 911–916 (2013)
A.U. Liyanage, E.U. Ikhuoria, A.A. Adenuga, V.T. Remcho, M.M. Lerner, Inorg. Chem. 52, 4603–4610 (2013)
Y. Piao, T. Wu, B. Chen, Ind. Eng. Chem. Res. 55, 6113–6121 (2016)
Y. Zhao, Y. Zhang, Z. Yang, Y. Yan, K. Sun, Sci. Technol. Adv. Mater. 14, 043501 (2013)
A.S. Abd-El-Aziz, C. Agatemor, N. Etkin, R. Bissessur, Macromol. Chem. Phys. 216, 369–379 (2014)
A. S. Abd-El-Aziz, C. Agatemor, N. Etkin, B.D. Wagner, Macromol. Rapid Commun. 37, 1235–1241 (2016)
R. Bissessur, D. Gallant, R. Brüning, Solid State Ion. 158, 205–209 (2003)
A. Hierlemann, J.K. Campbell, A. L, M. Baker, R, J. Crooks, A. Ricco, J. Am. Chem. Soc. 120, 5323–5324 (1998)
R. Bissessur, P.K.Y. Liu, Solid State Ion. 177, 191–196 (2006)
Acknowledgements
We thank Natural Science and Engineering Research Council of Canada for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dunlop, M.J., Agatemor, C., Abd-El-Aziz, A.S. et al. Nanocomposites Derived from Molybdenum Disulfide and an Organoiron Dendrimer. J Inorg Organomet Polym 27 (Suppl 1), 84–89 (2017). https://doi.org/10.1007/s10904-017-0582-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-017-0582-0