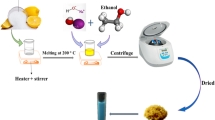
Abstract
In this study, a facile method is described for the Cu deposited Piperidine-4-carboxylic acid (Hpipe-4) coated magnetite (Fe3O4) nanoparticles. For which, Fe3O4 was used as magnetic core, Hpipe-4 as linker and active copper nanoparticles as the catalyst. For the reduction of Cu2+ to Cu0 was accomplished by NaBH4. The phase and morphology of the as-prepared product is characterized by means of X-ray powder diffractometry, energy dispersive spectrometry, Scanning electron microscopy. Magnetic and catalytic properties of the product were followed by vibrating sample magnetometer and UV–Vis spectrophometer respectively. Experiments showed that the as-prepared Fe3O4@Hpipe-4@Cu nanocatalyst owned strong catalytic activity for the reduction of some aromatic nitro-compounds and organic azo dyes and also emphasizes the effect of copper nanoparticles on the degradation of rate of hazardous compounds by NaBH4. Fe3O4@Hpipe-4@Cu nanocatalyst show great promise for the treatment of industrial dye pollutants hence environmental protection.
Graphical Abstract












Similar content being viewed by others
References
A. Manikandan, R. Sridhar, S. Arul Antony, S. Ramakrishna, J. Mol. Struct. 1076, 188–200 (2014)
C.O. Hendren, X. Mesnard, J. Droge, M.R. Wiesner, Environ. Sci. Technol. 45, 2562–2569 (2011)
R. Sharma, C. Sharma, Tetrahedron Lett. 51, 4415–4418 (2010)
K. Chinnaraj, A. Manikandan, P. Ramu, S. Arul Antony, P. Neeraja, J. Supercond. Nov. Magn. 28, 179–190 (2015)
N. Tripathy, R. Ahmad, J.E. Song, H.A. Ko, Y.B. Hahn, G. Khang, Mater. Lett. 136, 171–174 (2014)
I.A. Salem, Transition Met. Chem. 25, 599–604 (2000)
J. Fu, G.Z. Kyzas, Chin. J. Catal. 35, 1–7 (2014)
N. Baliarsingh, K.M. Parida, G.C. Pradhan, Ind. Eng. Chem. Res. 53, 3834–3841 (2014)
K.T. Chung, S.E. Stevens, Environ. Toxicol. Chem. 12, 2121–2132 (1993)
M.S. Lucas, J.A. Peres, Dyes Pigm. 71, 236–244 (2006)
B. Ho, T.M. Zabriskie, Bioorg. Med. Chem. Lett. 8, 739 (1998)
K. Lv, J. Wu, J. Wang, M. Liu, Z. Wei, J. Cao, Y. Sun, H. Guo, Bioorg. Med. Chem. Lett. 23, 1754–1759 (2013)
S.T. Harini, H.V. Kumar, J. Rangaswamy, N. Naik, Bioorg. Med. Chem. Lett. 22, 7588–7592 (2012)
G. Liu, C.H. Marrinan, S.A. Taylor, S. Black, A.D. Basso, P. Kirschmeier, W. Robert Bishop, M. Liu, B.J. Long, Anticancer Drugs 18, 923–931 (2007)
X. Chen, P. Zhan, C. Pannecouque, J. Balzarini, E. De Clercq, X. Liu, Eur. J. Med. Chem. 51, 60–66 (2012)
B.P. Bandgar, S.A. Patil, R.N. Gacche, B.L. Korbad, B.S. Hote, S.N. Kinkar, S.S. Jalde, Bioorg. Med. Chem. Lett. 20, 730–733 (2010)
E. Hollink, S.E. Tichy, E.E. Simanek, Ind. Eng. Chem. Res. 44, 1634–1639 (2005)
S.P. Frances, N. Van Dung, N.W. Beebe, M. Debboun, J. Med. Entomol. 39, 541–544 (2002)
V.D. Vitnik, Z.J. Vitnik, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 138, 1–12 (2015)
S.I. Mostafa, Transition Met. Chem. 32, 769–777 (2007)
K. Takahama, T. Miyata, Y. Okano, M. Kataoka, T. Hitoshi, Y. Kase, Eur. J. Pharmacol. 81, 327–335 (1982)
M.F. Wilson, A.E. Bell, Phytochemistry 17, 403–411 (1978)
C.M. Adams, E.A. Bernays, Entomol. Exp. Appl. 23, 101–108 (1978)
E. Karaoglu, A. Baykal, M. Şenel, H. Sözeri, M.S. Toprak, Mater. Res. Bull. 47, 2480–2486 (2012)
H. Erdemi, A. Baykal, E. Karaoğlu, M.S. Toprak, Mater. Res. Bull. 47, 2193–2199 (2012)
E. Karaoglu, A. Baykal, H. Deligöz, M. Şenel, H. Sözeri, M.S. Toprak, J. Alloy. Compd. 509, 8460–8468 (2011)
A. Manikandan, L. John Kennedy, J. Arul Mary, A. Dinesh, J. Judith Vijaya, J. Ind. Eng. Chem. 20, 2077–2085 (2014)
M. Tang, S. Zhang, X. Li, X. Pang, H. Qiu, Mater. Chem. Phys. 148, 639–647 (2014)
H. Kavas, Z. Durmus, A. Baykal, A. Aslan, A. Bozkurt, M.S. Toprak, J. Noncrystalline Solids 356, 484–489 (2010)
B. Unal, M.S. Toprak, Z. Durmus, H. Sözeri, A. Baykal, J. Nanopart. Res. 12, 3039–3048 (2010)
Z. Durmus, H. Kavas, A. Baykal, H. Sozeri, L. Alpsoy, S.Ü. Çelik, M.S. Toprak, J. Alloy. Compd. 509, 2555–2561 (2011)
A.A.A. El-Deen, A.E.M.E.E. Askalany, R. Halaoui, B.J.J. Claude, I.S. Butler, S.I. Mostafa, J. Mol. Struc. 1036, 161–167 (2013)
S.I. Mostafa, Transition Met. Chem. 32, 769–777 (2007)
Y. Inomata, Y. Arai, T. Yamakoshi, F. Scott Howell, Inorg. Biochem. 98, 2149–2154 (2004)
P.I. Girginova, F.A. Almeida Paz, H.I.S. Nogueira, N.J.O. Silva, V.S. Amaral, J. Klinowski, T. Trindade, Polyhedron 24, 563–571 (2005)
E. Murugan, J.N. Jebaranjitham, J. Mol. Cat. A: Chem. 365, 128–135 (2012)
Y. Inomata, M. Ando, F.S. Howell, J. Mol. Struc. 616, 201–212 (2002)
U. Kurtan, A. Baykal, Mater. Res. Bull. 60, 79–87 (2014)
Y. Junejo, A. Baykal, T. J. Chem. 38, 765–774 (2014)
Y. Junejo, Sirajuddin, A. Baykal, M. Safdar, A. Balouch, Appl. Surf. Sci. 290, 499–503 (2014)
E. Karaoğlu, U. Özel, C. Caner, A. Baykal, M.M. Summak, H. Sözeri, Mater. Res. Bull. 47, 4316–4321 (2012)
N. Pradhan, A. Pal, T. Pal, Langmuir 17, 1800–1809 (2001)
J.M. Herrmann, Water treatment by heterogeneous photocatalysis, in Environmental Catalysis, Catalytic Science Series, vol. Vol. 1, ed. by F. Jansen, R.A. van Santen (Imperial College Press, London, 1999), pp. 171–194. Chapter 9
W. Lua, R. Ninga, X. Qin, Y. Zhang, G. Chang, S. Liu, Y. Luo, X. Sun, J. Hazard. Mater. 197, 320–326 (2011)
Q. Zhou, G. Qian, Y. Li, G. Zhao, Y. Chao, J. Zheng, Thin Solid Films 516, 953–956 (2008)
V.K. Vidhu, Daizy Philip, Micron 56, 54–62 (2014)
J. Feng, L. Su, Y. Ma, C. Ren, Q. Guo, X. Chena, Chem. Eng. J. 13, 16–24 (2013)
N. Pradhan, A. Pal, T. Pal, Colloids Surf. A 196, 247–257 (2002)
Acknowledgments
This work was supported by Fatih University under BAP Grant No: P50021301-Y (3146).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurtan, U., Onuş, E., Amir, M. et al. Fe3O4@Hpipe-4@Cu Nanocatalyst for Hydrogenation of Nitro-Aromatics and Azo Dyes. J Inorg Organomet Polym 25, 1120–1128 (2015). https://doi.org/10.1007/s10904-015-0218-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-015-0218-1