Abstract
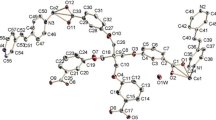
[2.2]Paracyclophane-layered polymers 4a and 4b end-capped by ferrocene and [2.2]paracyclophane, respectively, were synthesized by using a xanthene skeleton as a scaffold. Two model compounds, 4,5-bis(ferrocenylethynyl)-9,9-dimethylxanthene 7 and pseudo-p-bis(4-ferrocenylethynyl-9,9-dimethylxanthen-5-yl)[2.2]paracyclophane 8, comprising ferrocenes arranged in a face-to-face manner were also prepared. According to the X-ray molecular structure of compound 8, [2.2]paracyclophane between the ferrocenes had sufficient space for twisting and there was no interaction between two ferrocenes via [2.2]paracyclophane in the ground state. These polymers exhibited blue-light emission originating from the layered cyclophanes; however, the photoluminescence intensity of polymer 4a was lower than that of polymer 4b. The fluorescence emission of polymer 4a was effectively quenched by the end-capping ferrocene group.











Similar content being viewed by others
References
W.R. Salaneck, D.T. Clark, E.J. Samuelsen (eds.), Science and Applications of Conducting Polymers (Adam Hilger, Bristol, 1991)
H.S. Nalwa (ed.), Handbook of Organic Conductive Molecules (Wiley, Chichester, 1997)
T.A. Skothim, R.L. Elsenbaumer, J.R. Reynolds (eds.), Handbook of Conducting Polymers, 3rd edn. (Marcel Dekker, New York, 2006)
T. Nakano, K. Takewaki, T. Yade, Y. Okamoto, J. Am. Chem. Soc 123, 9182–9183 (2001)
T. Nakano, T. Yade, J. Am. Chem. Soc 125, 15474–15484 (2003)
T. Nakano, T. Yade, M. Yokoyama, N. Nagayama, Chem. Lett 33, 296–297 (2004)
T. Nakano, T. Yade, Y. Fukuda, T. Yamaguchi, S. Okumura, Macromolecules 38, 8140–8148 (2005)
T. Yade, T. Nakano, J. Polym. Part A: Polym. Chem. 44, 561–572 (2006)
T. Nakano, T. Yade, Chem. Lett. 37, 258–259 (2008)
A. García Martínez, J. Osío Barcina, A. de Fresno Cerezo, A.-D. Schlüter, J. Frahn, Adv. Mater. 11, 27–31 (1999)
S.A. Jenekhe, M.M. Alam, Y. Zhu, S. Jiang, A.V. Shevade, Adv. Mater. 19, 536–542 (2007)
Y. Morisaki, Y. Chujo, Tetrahedron. Lett. 46, 2533–2537 (2005)
Y. Morisaki, T. Murakami, Y. Chujo, Macromolecules 41, 5960–5963 (2008)
T. Otsubo, S. Mizogami, Y. Sakata, S. Misumi, Tetrahedron. Lett. 13, 2927–2930 (1972)
T. Otsubo, S. Mizogami, I. Otsubo, Z. Tozuka, A. Sakagami, Y. Sakata, S. Misumi, Bull. Chem. Soc. Jpn 46, 3519–3530 (1973)
M. Shibahara, M. Watanabe, T. Iwanaga, K. Ideta, T. Shinmyozu, J. Org. Chem. 72, 2865–2877 (2007)
K. Müllen (ed.), Electronic Materials: The Oligomeric Approach (Wiley-VHC, Weinheim, 1998)
J.M. Tour, Chem. Rev. 96, 537–554 (1996)
R.E. Martin, F. Diederich, Angew. Chem. Int. Ed 38, 1350–1377 (1999)
J.M. Tour, Acc. Chem. Rec 33, 791–804 (2000)
R.L. Carroll, C.B. Gorman, Angew. Chem. Int. Ed. 41, 4378–4400 (2002)
U.H.F. Bunz, Chem. Rev. 100, 1605–1644 (2000)
C. Weder (ed.), Poly(arylene ethynylene)s, Adv. Polym. Sci. 177 (2005)
D. Fichou (ed.), Handbook of Oligo- and Polythiophenes (Wiley-VHC, Weinheim, 1999)
T. Otsubo, Y. Aso, K. Takimiya, Bull. Chem. Soc. Jpn 74, 1789–1801 (2001)
T. Otsubo, Y. Aso, K. Takimiya, J. Mater. Chem. 12, 2565–2575 (2002)
Y. Morisaki, Y. Chujo, Angew. Chem. Int. Ed. 45, 6430–6437 (2006)
Y. Morisaki, Y. Chujo, Prog. Polym. Sci. 33, 346–364 (2008)
Y. Morisaki, Y. Chujo, Macromolecules 35, 587–589 (2002)
Y. Morisaki, Y. Chujo, Chem. Lett. 19, 194–195 (2002)
Y. Morisaki, T. Ishida, Y. Chujo, Macromolecules 35, 7872–7877 (2002)
Y. Morisaki, Y. Chujo, Polym. Bull. 49, 209–215 (2002)
Y. Morisaki, F. Fujimura, Y. Chujo, Organometallics 22, 3553–3557 (2003)
Y. Morisaki, Y. Chujo, Macromolecules 36, 9319–9324 (2003)
Y. Morisaki, Y. Chujo, Macromolecules 37, 4099–4103 (2004)
Y. Morisaki, T. Ishida, H. Tanaka, Y. Chujo, J. Polym. Sci. Part A: Polym. Chem. 42, 5891–5899 (2004)
Y. Morisaki, Y. Chujo, Bull. Chem. Soc. Jpn 78, 288–293 (2005)
Y. Morisaki, N. Wada, Y. Chujo, Polym. Bull. 53, 73–80 (2005)
Y. Morisaki, N. Wada, Y. Chujo, Polymer 46, 5884–5889 (2005)
A.B. Pangborn, M.A. Giardello, R.H. Grubbs, R.K. Rosen, R.F.J. Timmers, Organometallics 15, 1518–1520 (1996)
L. Bondarenko, I. Dix, H. Hinrichs, H. Hopf, Synthesis 16, 2751–2759 (2004)
K. McWilliams, J.W. Kelly, J. Org. Chem. 61, 7408–7414 (1996)
A. Altomare, M. C. Burla, M. Camalli, G. Cascarano, C. Giacovazzo, A. Guagliardi, A. G. Moliteni, G. Polidori, R. Spagna, J. Appl. Cryst. 32, 115–119 (1999)
G.M. Sheldrick, SHELX97 Programs for Crystal Structure Analysis (University of Göttingen, Göttingen, Germany, 1997)
H. Hopf, I. Dix, Synlett 1416–1418 (2006)
M.T. Lee, B.M. Foxman, M. Rosenblum, Organometallics 4, 539–547 (1985)
R. Arnold, B.M. Foxman, M. Rosenblum, W.B. Euler, Organometallics 7, 1253–1259 (1998)
R. Arnold, S.A. Matchett, M. Rosenblum, Organometallics 7, 2261–2267 (1988)
R.H. Herber, I. Nowik, M. Rosenblum, Organometallics 21, 846–851 (2002)
M. Iyoda, T. Okabe, M. Katada, Y. Kuwatani, J. Organomet. Chem 569, 225–233 (1998)
H.M. Nugent, M. Rosenblum, P. Klemarczky, J. Am. Chem. Soc. 115, 3848–3849 (1993)
M. Rosenblum, H.M. Nugent, K.-S. Jang, M.M. Labes, W. Cahalane, P. Klemarczyk, W.M. Reiff, Macromolecules 28, 6330–6342 (1995)
R.D.A. Hudson, B.M. Foxman, M. Rosenblum, Organometallics 18, 4098–4106 (1999)
F. Cozzi, M. Cinquini, R. Annunziata, T. Dwyer, J.S. Siegel, J. Am. Chem. Soc. 114, 5729–5733 (1992)
F. Cozzi, M. Cinquini, R. Annunziata, J.S. Siegel, J. Am. Chem. Soc. 115, 5330–5331 (1993)
F. Cozzi, F. Ponzini, R. Annunziata, M. Cinquini, J.S. Siegel, Angew. Chem. Int. Ed. 34, 1019–1020 (1995)
R. Tsuji, K. Komatsu, K. Takeuchi, M. Shiro, S. Cohen, M. Rabinovitz, J. Phy. Org. Chem. 6, 435–444 (1993)
N. Kaneta, F. Mitamura, M. Uemura, Y. Murata, K. Komatsu, Tetrahedron. Lett. 37, 5385–5388 (1996)
Q. Zhou, T.M. Swager, J. Am. Chem. Soc. 117, 7017–7018 (1995)
M.J. Marsella, P.J. Carroll, T.M. Swager, J. Am. Chem. Soc. 117, 9832–9841 (1995)
Q. Zhou, T.M. Swager, J. Am. Chem. Soc. 117, 12593–12602 (1995)
R. Deans, J. Kim, M.R. Machacek, T.M. Swager, J. Am. Chem. Soc. 122, 8565–8566 (2000)
A. Rose, C.G. Lugmair, T.M. Swager, J. Am. Chem. Soc. 123, 11298–11299 (2001)
J. Zheng, T.M. Swager, Adv. Polym. Sci. 177, 151–179 (2005)
S.W. Thomas, G.D. Joly, T.M. Swager, Chem. Rev. 107, 1339–1386 (2007)
Acknowledgments
This work was supported by Grant-in-Aid for Creative Scientific Research of “Invention of Conjugated Electronic Structures and Novel Functions”, No. 16GS0209, from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Morisaki, Y., Murakami, T. & Chujo, Y. Synthesis, Structure, and Properties of Aromatic Ring-Layered Polymers Containing Ferrocene as a Terminal Unit. J Inorg Organomet Polym 19, 104–112 (2009). https://doi.org/10.1007/s10904-008-9236-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-008-9236-6