Abstract
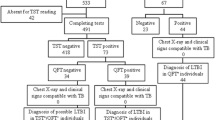
Immigrants and refugees age 2–14 years entering the United States from countries with estimated tuberculosis (TB) incidence rate ≥20 per 100,000 population are screened for TB. Children with TB disease are treated before US arrival. Children with positive tuberculin skin tests (TST), but negative TB evaluation during their pre-immigration examination, are classified with latent TB infection (LTBI) and are recommended for re-evaluation post-arrival. We examined post-immigration TB evaluation and therapy for children arriving with LTBI. We reviewed medical exam data from immigrant children with medical conditions and all refugee children arriving during 2010. Medical examination data were available for 67,334 children. Of these, 8231 (12 %) had LTBI pre-immigration; 5749 (70 %) were re-evaluated for TB post-immigration, and 64 % were retested by TST or IGRA. The pre-immigration LTBI diagnosis was changed for 38 % when retested by TST and for 71 % retested by IGRA. Estimated LTBI therapy initiation and completion rates were 68 and 12 %. In this population, testing with IGRA may limit the number of children targeted for therapy. Increased pre-immigration TB screening with post-immigration follow-up evaluation leading to completion of LTBI therapy should be encouraged to prevent TB reactivation.

Similar content being viewed by others
References
Miramontes R, Pratt R, Price SF, Jeffries C, Navin TR, Oramasionwu GE. Trends in tuberculosis—United States, 2011. MMWR Morb Mortal Wkly Rep. 2012;61(11):181–5.
Al-Orainey IO. Diagnosis of latent tuberculosis: can we do better? Ann Thorac Med. 2009;4(1):5.
Linas BP, Wong AY, Freedberg KA, Horsburgh CR. Priorities for screening and treatment of latent tuberculosis infection in the United States. Am J Respir Crit Care Med. 2011;184(5):590–601.
Mazurek GH, Jereb JA, Vernon A, LoBue P, Goldberg S, Castro KG. Updated guidelines for using interferon gamma release assays to detect Mycobacterium tuberculosis infection, United States, 2010. MMWR Recomm Rep. 2010;59(RR05);1–25.
Rose DN. Benefits of screening for latent Mycobacterium tuberculosis infection. Arch Intern Med. 2000;160(10):1513.
Hill A, Becerra J, Castro K. Modelling tuberculosis trends in the USA. Epidemiol Infect. 2012;140(10):1862.
Centers for Disease Control and Prevention. CDC immigration requirements: technical instructions for tuberculosis screening and treatment using cultures and directly observed therapy. Atlanta, GA: US Department of Health and Human Services; 2009.
Cohn DL, O’Brien RJ, Geiter LJ, Gordin F, Hershfield E, Horsburgh C. Targeted tuberculin testing and treatment of latent tuberculosis infection. MMWR Morb Mortal Wkly Rep. 2000;49(6):1–54.
US Department of Homeland Security, C.a.B.P. TECS, arrival file: fiscal years 2010 to 2011. Washington: C.a.B.P. US Department of Homeland Security; 2012.
Centers for Disease Control and Prevention: Technical instructions implementation. 2011 February 5, 2013; (cited 2013 February 14). https://doi.org/www.cdc.gov/immigrantrefugeehealth/exams/ti/panel/tuberculosis-implementation.html.
Centers for Disease Control and Prevention: Latent tuberculosis infection: a guide for primary health care providers 2010 November 24, 2010; (cited 2013 April 15). https://doi.org/www.cdc.gov/tb/publications/ltbi/diagnosis.htm.
Hirsch-Moverman Y, Bethel J, Colson P, Franks J, El-Sadr W. Predictors of latent tuberculosis infection treatment completion in the United States: an inner city experience. Int J Tuberc Lung Dis. 2010;14(9):1104–11.
Coly A, Morisky D. Predicting completion of treatment among foreign-born adolescents treated for latent tuberculosis infection in Los Angeles. Int J Tuberc Lung Dis. 2004;8(6):703–10.
LoBue PA, Moser KS. Use of isoniazid for latent tuberculosis infection in a public health clinic. Am J Respir Crit Care Med. 2003;168(4):443–7.
Young J, Edick T, Klee D, O’Connor ME. Successful treatment of pediatric latent tuberculosis infection in a community health center clinic. Pediatr Infect Dis J. 2012;31(9):e147–51.
Hovell MF, Sipan CL, Blumberg EJ, Hofstetter CR, Slymen D, Friedman L, Moser K, Kelley NJ, Vera AY. Increasing Latino adolescents’ adherence to treatment for latent tuberculosis infection: a controlled trial. Am J Public Health. 2003;93(11):1871–7.
Centers for Disease Control and Prevention: Guidelines for the U.S. domestic medical examination for newly arriving refugees. 2012 September 24, 1012; (cited 2013 April 15). https://doi.org/www.cdc.gov/immigrantrefugeehealth/guidelines/domestic/domestic-guidelines.html.
Advisory Council for the Elimination of Tuberculosis (ACET). The Role of BCG Vaccine in the Prevention and Control of Tuberculosis in the United States. A Joint Statement by the Advisory Council for the Elimination of Tuberculosis and the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 1996;45(RR-4):1–27.
Farhat M, Greenaway C, Pai M, Menzies D. False-positive tuberculin skin tests: what is the absolute effect of BCG and non-tuberculous mycobacteria?[Review Article]. Int J Tuberc Lung Dis. 2006;10(11):1192–204.
Zwerling A, Behr MA, Verma A, Brewer TF, Menzies D, Pai M. The BCG World Atlas: a database of global BCG vaccination policies and practices. PLoS Med. 2011;8(3):e1001012.
Acknowledgments
We thank the staff of the CDC’s Electronic Disease Notification (EDN) team for updating and managing the CDC’s notification system for tuberculosis in immigrants and refugees, the staff of CDC’s quarantine stations for collecting information about overseas medical examinations, the panel physicians for performing overseas tuberculosis screening, and the staff of state and local health departments for conducting follow-up evaluations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taylor, E.M., Painter, J., Posey, D.L. et al. Latent Tuberculosis Infection Among Immigrant and Refugee Children Arriving in the United States: 2010. J Immigrant Minority Health 18, 966–970 (2016). https://doi.org/10.1007/s10903-015-0273-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10903-015-0273-2