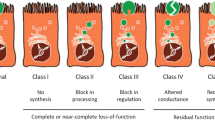
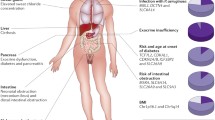
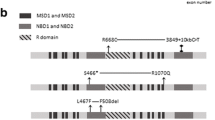
Purpose
To provide practice recommendations for genetic counselors whose clients are considering cystic fibrosis (CF) carrier testing or seeking information regarding CF molecular test results. The goals of these recommendations are to: 1) Provide updated information about the natural history, diagnosis, and treatment of CF and related conditions. 2) Supplement genetic counselors’ knowledge and understanding of the available carrier screening and diagnostic testing options. 3) Describe the current state of genotype/phenotype correlations for CFTR mutations and an approach to interpreting both novel and previously described variants. 4) Provide a framework for genetic counselors to assist clients’ decision-making regarding CF carrier testing, prenatal diagnosis, and pregnancy management.
Disclaimer The practice guidelines of the National Society of Genetic Counselors (NSGC) are developed by members of the NSGC to assist genetic counselors and other health care providers in making decisions about appropriate management of genetic concerns; including access to and/or delivery of services. Each practice guideline focuses on a clinical or practice-based issue, and is the result of a review and analysis of current professional literature believed to be reliable. As such, information and recommendations within the NSGC practice guidelines reflect the current scientific and clinical knowledge at the time of publication, are only current as of their publication date, and are subject to change without notice as advances emerge.
In addition, variations in practice, which take into account the needs of the individual patient and the resources and limitations unique to the institution or type of practice, may warrant approaches, treatments and/or procedures that differ from the recommendations outlined in this guideline. Therefore, these recommendations should not be construed as dictating an exclusive course of management, nor does the use of such recommendations guarantee a particular outcome. Genetic counseling practice guidelines are never intended to displace a health care provider’s best medical judgment based on the clinical circumstances of a particular patient or patient population.
Practice guidelines are published by NSGC for educational and informational purposes only, and NSGC does not “approve” or “endorse” any specific methods, practices, or sources of information.
Similar content being viewed by others
References
American College of Obstetricians and Gynecologists. (2005). Update on carrier screening for cystic fibrosis Committee Opinion No. 325. Obstetrics and Gynecology, 106(6), 1465–1468.
American College of Obstetricians and Gynecologists. (2011). Update on carrier screening for cystic fibrosis. Committee Opinion No. 486. Obstetrics and Gynecology, 117(4), 1028–1031.
American College of Obstetricians and Gynecologists, & American College of Medical Genetics. (2001). Preconception and prenatal carrier screening for cystic fibrosis. Clinical and Laboratory Guidelines. American College of Obstetricians and Gynecologists, Washington, DC.
Bobadilla, J. L., Macek, M., Jr., Fine, J. P., & Farrell, P. M. (2002). Cystic fibrosis: a worldwide analysis of CFTR mutations– correlation with incidence data and application to screening. Human Mutation, 19(6), 575–606.
Bombieri, C., Claustres, M., De Boeck, K., et al. (2011). Recommendations for the classification of diseases as CFTR-related disorders. Journal of Cystic Fibrosis, 10(Suppl2), S86–S102.
Borowitz, D., Parad, R., Sharp, J. K., et al. (2009). Cystic Fibrosis Foundation practice guidelines for the management of infants with cystic fibrosis transmembrane conductance regulator-related metabolic syndrome during the first two years of life and beyond. Journal of Pediatrics, 155, S106e16.
Buller, A., Olson, S., Redman, J. B., et al. (2004). Frequency of the cystic fibrosis 3199del6 mutation in individuals heterozygous for I148T. Genetics in Medicine, 6(2), 108–109.
Carlyle, B. E., Borowitz, D. S., & Glick, P. L. (2012). A review of pathophysiology and management of fetuses and neonates with meconium ileus for the pediatric surgeon. Journal of Pediatric Surgery, 47(4), 772–781.
Castellani, C., Cuppens, H., Macek, M., et al. (2008). Consensus on the use and interpretation of cystic fibrosis mutation analysis in clinical practice. Journal of Cystic Fibrosis, 7, 179–196.
Claustres, M., Altiére, J.-P., Guittard, C., et al. (2004). Are p.I148T, p.R74W and p.D1270N cystic fibrosis causing mutations? BMC Medical Genetics, 5, 19.
Cleveland, R. H., Zurakowski, D., Slattery, D., & Colin, A. A. (2009). Cystic fibrosis genotype and assessing rates of decline in pulmonary status. Radiology, 253(3), 813–821.
Collaco, J. M., & Cutting, G. R. (2008). Update on gene modifiers in cystic fibrosis. Current Opinion in Pulmonary Medicine, 14(6), 559–566.
Cuppens, H., Lin, W., Jaspers, M., Costes, B., Teng, H., Vankeerberghen, A., et al. (1998). Polyvariant mutant cystic fibrosis transmembrane conductance regulator genes. The polymorphic (Tg)m locus explains the partial penetrance of the T5 polymorphism as a disease mutation. Journal of Clinical Investigation, 101(2), 487–496.
Cystic Fibrosis Foundation. (1997). Clinical practice guidelines.
Cystic Fibrosis Foundation Patient Registry. (2011). Database, accessed 24 April 2013. Bethesda, Maryland.
Cystic Fibrosis Foundation Patient Registry. (2012). 2011 Annual data report.
De Boeck, K., Wilschanski, M., Castellani, C., et al. (2006). Cystic fibrosis: terminology and diagnostic algorithms. Thorax, 61, 627–635.
Dequeker, E., Stuhrmann, M., Morris, M. A., et al. (2009). Best practice guidelines for molecular genetic diagnosis of cystic fibrosis and CFTR-related disorders—updated European recommendations. European Journal of Human Genetics, 17(1), 51–65.
Di Sant’Agnese, P. A., Darling, R. C., Perera, G. A., & Shea, E. (1953). Sweat electrolyte disturbances associated with childhood pancreatic disease. American Journal of Medicine, 15(6), 777–784.
Drumm, M. L., Konstan, M. W., Schluchter, M. D., Handler, A., Pace, R., Zou, F., et al. (2005). Gene Modifier Study Group. New England Journal of Medicine, 353(14), 1443–1453.
Eshaque, B., & Dixon, B. (2006). Technology platforms for molecular diagnosis of cystic fibrosis. Biotechnology Advances, 24, 86–93.
Farrell, P. M., Kosorok, M. R., Laxova, A., et al. (1997). Wisconsin Cystic Fibrosis Neonatal Screening Study Group. Nutritional benefits of neonatal screening for cystic fibrosis. The New England Journal of Medicine, 337, 963–969.
Farrell, P. M., Rosenstein, B. J., White, T. B., et al. (2008). Guidelines for diagnosis of cystic fibrosis in newborns through older adults: Cystic Fibrosis Foundation consensus report. Journal of Pediatrics, 15(3), S4–S14.
Fiel, S. B. (1996). Pulmonary function during pregnancy in cystic fibrosis: implications for counseling. Current Opinion in Pulmonary Medicine, 2(6), 462–465.
Grody, W. W., Cutting, G. R., Klinger, K. W., et al. (2001). Laboratory standards and guidelines for population-based cystic fibrosis carrier screening. Genetics in Medicine, 3(2), 149–154.
Grody, W. W., Cutting, G. R., & Watson, M. S. (2007). The cystic fibrosis mutation “arms race”: when less is more. Genetics in Medicine, 9(11), 739–744.
Groman, J. D., Karczeski, B., Sheridan, M., et al. (2005). Phenotypic and genetic characterization of patients with features of “nonclassic” forms of cystic fibrosis. Journal of Pediatrics, 146(5), 675–680.
Groman, J. D., Hefferon, T. W., Casals, T., et al. (2004). Variation in a repeat sequence determines whether a common variant of the cystic fibrosis transmembrane conductance regulator gene is pathogenic or benign. American Journal of Human Genetics, 74(1), 176–179.
Howenstine, M., & Montegomery, G. S. (2009). Cystic fibrosis. Pediatrics in Review, 30, 302–310. doi:10.1542/pir.30-8-302.
Jones, A. M., & Helm, J. M. (2009). Emerging treatments in cystic fibrosis. Drugs, 69(14), 1903–1910.
Keiles, S., Koepke, R., Parad, R., et al. (2011). Impact of IVS8-(TG)m(T)n on IRT and sweat chloride levels in newborns identified by California CF newborn screening. Journal of Cystic Fibrosis (in press).
Kere, E., Reisman, J., Corey, M., et al. (1992). Prediction of mortality in patients with cystic fibrosis. New England Journal of Medicine, 326(8), 1187–1191.
Kiesewetter, S., Macek, M., Jr., Davis, C., et al. (1993). A mutation in CFTR produces different phenotypes depending on chromosomal background. Nature Genetics, 5(3), 274–278.
Koscik, R. L., Lai, H., Laxova, A., et al. (2005). Preventing early, prolonged vitamin E deficiency: an opportunity for better cognitive outcomes via early diagnosis through neonatal screening. Journal of Pediatrics, 147(3 Suppl), S51–S56.
Langfelder-Schwind, E., Kloza, E., Sugarman, E., et al. (2005). Cystic fibrosis prenatal screening in genetic counseling practice: recommendations of the National Society of Genetic Counselors. Journal of Genetic Counseling, 14(1), 1–15.
Lebo, R. V., & Grody, W. W. (2007). Testing and reporting ACMG cystic fibrosis mutation panel results. Genetic Testing, 11(1), 11–31.
Massie, R. J., Poplawski, N., Wilcken, B., et al. (2001). Intron-8 polythymidine sequence in Australasian individuals with CF mutations R117H and R117C. European Respiratory Journal, 17(6), 1195–1200.
Massie, R. J. H., Curnow, L., Glazner, J., et al. (2012). Lessons learned from 20 years of newborn screening for cystic fibrosis. The Medical Journal of Australia, 196(1), 67–70.
McKone, E. F., Emerson, S. S., Edwards, K. L., & Aitken, M. L. (2003). Effect of genotype on phenotype and mortality in cystic fibrosis: a retrospective cohort study. Lancet, 361(9370), 1671–1676.
McKone, E. F., Goss, C. H., & Aitken, M. L. (2006). CFTR genotype as a predictor of prognosis in cystic fibrosis. Chest, 130(5), 1441–1447.
Mehta, G., Macek, M., Jr., Mehta, A., & European Registry Working Group. (2010). Cystic fibrosis across Europe: EuroCareCF analysis of demographic data from 35 countries. Journal of Cystic Fibrosis, 9(Suppl 2), S5–S21.
Mérelle, M. E., Scheffer, H., DeJong, D., & Dankert-Roelse, J. E. (2006). Extended gene analysis can increase specificity of neonatal screening for cystic fibrosis. Acta Paediatrica, 95, 1424–1428.
Mickle, J. E., & Cutting, G. R. (1998). Clinical implications of cystic fibrosis transmembrane conductance regulator mutations. Clinics in Chest Medicine, 19(3), 443–458.
Monaghan, K. G., Highsmith, W. E., Amos, J., et al. (2004). Genotype-phenotype correlation and frequency of the 3199del6 cystic fibrosis mutation among I148T carriers: results from a collaborative study. Genetics in Medicine, 6(5), 421–425.
Moran, A., Brunzell, C., Cohen, R. C., et al. (2010). CFRD Guidelines Committee. Clinical care guidelines for cystic fibrosis-related diabetes. Diabetes Care, 33(12), 2697–2708.
Morton, J., & Glanville, A. R. (2009). Lung transplantation in patients with cystic fibrosis. Seminars in Respiratory and Critical Care Medicine, 30(5), 559–568.
Moskowitz, S. M., Chmiel, J. F., Sternen, D. L., et al. (2008). Clinical practice and genetic counseling for cystic fibrosis and CFTR-related disorders. Genetics in Medicine, 10(12), 851–868.
Mussaffi, H., Prais, D., Mei-Zahav, M., & Blau, H. (2006). Cystic fibrosis mutations with widely variable phenotype: the D1152H example. Pediatric Pulmonology, 41(3), 250–254.
Ogino, S., Wilson, R. B., & Grody, W. W. (2004). Bayesian risk assessment for autosomal recessive diseases: fetal echogenic bowel with one or not detectable CFTR mutation. Journal of Medical Genetics, 41(5), e70.
O’Neill, S. C., DeMarco, T., Peshkin, B. N., et al. (2006). Tolerance for uncertainty and perceived risk among women receiving uninformative BRCA1/2 test results. American Journal of Medical Genetics. Part C, Seminars in Medical Genetics, 142C(4), 251–259.
Palomaki, G. E., FitzSimmons, S. C., & Haddow, J. E. (2004). Clinical sensitivity of prenatal screening for cystic fibrosis via CFTR carrier testing in a United States panethnic population. Genetics in Medicine, 6(5), 405–414.
Quittner, A. L., Modi, A., & Cruz, I. (2008). Systematic review of health-related quality of life measures for children with respiratory conditions. Paediatric Respiratory Reviews, 9(3), 220–232.
Rohlfs, E. M., Sugarman, E. A., Heim, R. A., & Allitto, B. A. (2001). Frequency of carriers of two cystic fibrosis mutations in an apparently unaffected adult population. Genetics in Medicine, 3, 237.
Rohlfs, E. M., Zhou, Z., Sugarman, E. A., et al. (2002). The I148T CFTR allele occurs on multiple haplotypes: a complex allele is associated with cystic fibrosis. Genetics in Medicine, 4(5), 319–323.
Rohlfs, E. M., Zhou, Z., Heim, R. A., et al. (2011). Cystic fibrosis carrier testing in an ethnically diverse US population. Clinical Chemistry, 57(6), 841–848.
Rosenstein, B. J., & Cutting, G. R. (1998). The diagnosis of cystic fibrosis: a consensus statement. Cystic Fibrosis Foundation consensus panel. Journal of Pediatrics, 132(4), 589–595.
Sawicki, G. S., Rasouliyan, L., McMullen, A. H., et al. (2010). Longitudinal assessment of health-related quality of life in an observational cohort of patients with cystic fibrosis. Pediatric Pulmonology, 46(1), 36–44.
Schibli, S., Durie, P. R., & Tullis, E. D. (2002). Proper usage of pancreatic enzymes. Current Opinion in Pulmonary Medicine, 8, 542–546.
Schlegel, P. N., Cohen, J., Goldstein, M., et al. (1995). Cystic fibrosis gene mutations do not affect sperm function during in vitro fertilization with micromanipulation for men with bilateral congenital absence of vas deferens. Fertility and Sterility, 64(2), 421–426.
Sebro, R., Levy, H., Schneck, K., et al. (2012). Cystic fibrosis mutations for p.F508del compound heterozygotes predict sweat chloride levels and pancreatic sufficiency. Clinical Genetics, 82, 546–551.
Smith, H. C. (2010). Fertility in men with cystic fibrosis assessment, investigations and management. Paediatric Respiratory Reviews, 11(2), 80–83.
Sosnay, P. R., Castellani, C., Corey, M., et al. (2011). Evaluation of the disease liability of CFTR variants. Methods in Molecular Biology, 742, 355–372.
Strom, C. M., Huang, D., Chen, C., et al. (2003). Extensive sequencing of the cystic fibrosis transmembrane regulator gene: assay validation and unexpected benefits of developing a comprehensive test. Genetics in Medicine, 5(1), 9–14.
Svensson, A. M., Chou, L. S., Miller, C. E., et al. (2010). Detection of large rearrangements in the cystic fibrosis transmembrane conductance regulator gene by multiplex ligation-dependent probe amplification assay when sequencing fails to detect two disease-causing mutations. Genetic Testing and Molecular Biomarkers, 14(2), 171–174.
Taylor, C. J., Hardcastle, J., & Southern, K. W. (2009). Physiological measurements confirming the diagnosis of cystic fibrosis: the sweat test and measurements of transepithelial potential difference. Paediatric Respiratory Reviews, 10(4), 220–226.
Thauvin-Robinet, C., Munck, A., Huet, F., et al. (2009). Collaborating Working Group on R117H. The very low penetrance of cystic fibrosis for the R117H mutation: a reappraisal for genetic counseling and newborn screening. Journal of Medical Genetics, 46(11), 752–758.
Tluczek, A., Zaleski, C., Stachiw-Hietpas, D., et al. (2011). A tailored approach to family-centered genetic counseling for cystic fibrosis newborn screening: the Wisconsin model. Journal of Genetic Counseling, 20(2), 115–128.
Uhlmann, W. (2009). Thinking it all through: Case preparation and management. In D. L. Baker, J. L. Schuette, & W. Uhlmann (Eds.), A guide to genetic counseling (2nd ed., pp. 108–110). New York: Wiley-Liss, Inc.
Vanscoy, L. L., Blackman, S. M., Collaco, J. M., Bowers, A., Lai, T., Naughton, K., et al. (2007). American Journal of Respiratory and Critical Care Medicine, 175(10), 1036–1043.
Waters, D. L., Wilcken, B., Irwig, L., et al. (1999). Clinical outcomes of newborn screening for cystic fibrosis. Archives of Disease in Childhood. Fetal and Neonatal Edition, 80(1), F1–F7.
Watson, M. S., Cutting, G. R., Desnick, R. J., et al. (2004). Cystic fibrosis population carrier screening: 2004 revision of American College of Medical Genetics mutation panel. Genetics in Medicine, 6(5), 387–391.
Weil, J. (Ed.). (2000). Psychosocial genetic counseling (pp. 133–134). New York: Oxford University Press.
Zielenski, J. (2000). Genotype and phenotype in cystic fibrosis. Respiration, 67(2), 117–133.
Whittey, J. E. (2010). Cystic fibrosis in pregnancy. Clinical Obstetrics and Gynecology, 53(2), 369–376.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Langfelder-Schwind, E., Karczeski, B., Strecker, M.N. et al. Molecular Testing for Cystic Fibrosis Carrier Status Practice Guidelines: Recommendations of the National Society of Genetic Counselors. J Genet Counsel 23, 5–15 (2014). https://doi.org/10.1007/s10897-013-9636-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10897-013-9636-9