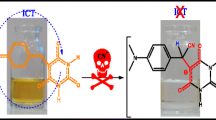
Abstract
A chemical sensor that can induce near-infrared red-shifted response represents a promising strategy for the design and development of anion probes. In this work, novel CH-controlled colorimetric probe 3 based on anthracene carboximide was developed for near-infrared detection of cyanide. Probe 3 consisted of CHCN binding site to anthracene carboximide fluorophore, and showed a significant visual change from yellow-green (535 nm) to deep violet (825 nm) with a larger redshift (≈ 290 nm) and fluorescence quenching at 480 nm and 520 nm upon interacting with cyanide. Job curves determined 1:1 binding stoichiometry of probe 3 with cyanide. Additonally, probe 3 detected cyanide ion conveniently in aqueous solution and could be reused after trifluoroacetic acid treatment. Colorimetric test paper was used to detect cyanide in aqueous solutions. The C–H deprotonation sensing mechanism was confirmed by 1H NMR titration. The near-infrared detection of cyanide by CH-controlled probes was founded for the first time.








Similar content being viewed by others
References
Chen D, Letcher RJ, Gauthier LT, Chu S, McCrindle R, Potter D (2011) Novel methoxylated polybrominated diphenoxybenzene congeners and possible sources in herring gull eggs from the Laurentian Great Lakes of North America. Environ Sci Technol 45:9523–9530
Goldwasser P, Manjappa NG, Luhrs CA, Barth RH (2011) Pseudohypobicarbonatemia caused by an endogenous assay interferent: a new entity. Am J Kidney Dis 58:617–620
Peng L, Wang M, Zhang G, Zhang D, Zhu D (2009) A Fluorescence turn-on detection of cyanide in aqueous solution based on the aggregation-induced emission. Org Lett 11:1943–1946
Lebeda FJ, Deshpande SS (1990) Potentiometric measurements of hydrogen and cyanide ions in buffered media. Anal Biochem 187:302–309
Dash RR, Gaur A, Balomajumder C (2009) Cyanide in industrial wastewaters and its removal: a review on biotreatment. J Hazard Mater 163:1–11
Zeng J, Cao Y, Chen J, Wang X, Yu J, Yu B, Yan Z, Chen X (2014) Au@Ag core/shell nanoparticles as colorimetric probes for cyanide sensing. Nanoscale 6:9939–9943
Peng MJ, Guo Y, Yang XF, Suzenet F, Li J, Li CW, Duan YW (2014) Coumarin-hemicyanine conjugates as novel reaction-based sensors for cyanide detection: convenient synthesis and ICT mechanism. RSC Adv 4:19077–19085
Wang L, Zhu L, Cao D (2015) A colorimetric probe based on diketopyrrolopyrrole and tert-butyl cyanoacetate for cyanide detection. New J Chem 39:7211–7218
Konidena RK, Justin Thomas KR (2014) Selective naked-eye cyanide detection in aqueous media using a carbazole-derived fluorescent dye. RSC Adv 4:22902–22910
Wang F, Wang L, Chen X, Yoon J (2014) Recent progress in the development of fluorometric and colorimetric chemosensors for detection of cyanide ions. Chem Soc Rev 43:4312–4324
Suzuki T, Hioki A, Kurahashi M (2003) Development of a method for estimating an accurate equivalence point in nickel titration of cyanide ions. Anal Chim Acta 476:159–165
Shan D, Mousty C, Cosnier S (2004) Subnanomolar cyanide detection at polyphenol oxidase/clay biosensors. Anal Chem 76:178–183
Isaad J, Achari AE (2011) Colorimetric sensing of cyanide anions in aqueous media based on functional surface modification of natural cellulose materials. Tetrahedron 67:4939–4947
Safavi A, Maleki N, Shahbaazi HR (2004) Indirect determination of cyanide ion and hydrogen cyanide by adsorptive stripping voltammetry at a mercury electrode. Anal Chim Acta 503:213–221
Zhang Y, Yu D, Feng G (2004) Colorimetric and near infrared fluorescent detection of cyanide by a new phenanthroimidazole- indolium conjugated probe. RSC Adv 4:14752–14757
Gao T, Yang S, Cao X, Dong J, Zhao N, Ge P, Zeng W, Cheng Z (2017) Smart self-assembled organic nanoprobe for protein—specific detection: design, synthesis, application, and mechanism studies. Anal Chem 89:10085–10093
Hua B, Shao L, Yu G, Huang F (2016) Fluorescence indicator displacement detection based on pillar [5] arene-assisted dye deprotonation. Chem Commun 52:10016–10019
Lee S, Hua Y, Park H, Flood AH (2010) Intramolecular hydrogen bonds preorganize an aryl-triazole receptor into a crescent for chloride binding. Org Lett 12:2100–2102
Bryantsev VS, Hay BP (2005) Influence of substituents on the strength of aryl C–H···anion hydrogen bonds. Org Lett 7:5031–5034
White NG, Beer PD (2013) A rotaxane host system containing integrated triazole C–H hydrogen bond donors for anion recognition. Org Biomol Chem 11:1326–1333
Sato K, Arai S, Yamagishi T (1999) A new tripodal anion receptor with C–H···X- hydrogen bonding. Tetrahedron Lett 40:5129–5222
Sui B, Kim B, Zhang Y, Frazer A, Belfield KD (2013) Highly selective fluorescence turn-on sensor for fluoride detection. ACS Appl Mater Interfaces 5:2920–2923
Sessler JL, Cai J, Gong HY, Yang X, Arambula JF, Hay BP (2010) A pyrrolyl-based triazolophane: a macrocyclic receptor with CH and NH donor groups that exhibits a preference for pyrophosphate anions. J Am Chem Soc 132:14058–14060
Jo J, Olasz A, Chen CH, Lee D (2013) Interdigitated hydrogen bonds: electrophile activation for covalent capture and fluorescence turn-on detection of cyanide. J Am Chem Soc 135:3620–3632
Yang Z, Zhang Z, Meanwell NA, Kadow JF, Wang T (2002) A strategy for the synthesis of aryl α-ketoamides based upon the acylation of anions derived from cyanomethylamines followed by oxidative cleavage. Org Lett 4:1103–1105
Hunger M (1996) Multinuclear solid-state NMR studies of acidic and non-acidic hydroxyl protons in zeolites. Solid State Nucl Magn Reson 6:1–29
Zhu T, Li Z, Fu C, Chen L, Chen X, Gao C, Zhang S, Liu C (2020) Development of an anthraquinone-based cyanide colorimetric sensor with activated C–H group: large absorption red shift and application in food and water samples. Tetrahedron 76:131479
Li Z, Rao C, Chen L, Fu C, Zhu T, Chen X, Liu C (2019) Addition of α-cyanomethylpyridine to naphthalimide via trifluoromethyl- directed CH functionalization: cyanide sensing in aqueous media. J Org Chem 84:7518–7522
Chen Y, Hu X, Rao C, Li Z, Chen L, Fu C, Liu C (2018) A reusable cyanide sensor via activation of C–H group: trifluoromethylcarbinol-directed meta-C–H cyanomethylation of naphthalimide. Analyst 143:4655–4661
Zhang C, Ji K, Wang X, Wu H, Liu C (2015) A reversible and selective chemosensor based on intramolecular NH···NH2 hydrogen bonding for cyanide and pH detection. Chem Commun 51:8173–8176
Zhou M, Chen J, Liu C, Fu H, Zheng N, Zhang C, Chen Y, Cheng J (2014) Anion binding modes in cis-trans-isomers of a binding site-fluorophore-π-extended system. Chem Commun 50:14748–14751
Liu B, Yu TH (2005) A ratiometric fluorescent chemosensor for fluoride ions based on a proton transfer signaling mechanism. J Mater Chem 15:2681–2686
Ren J, Wu Z, Zhou Y, Li Y, Xu Z (2011) Colorimetric fluoride sensor based on 1, 8-naphthalimide derivatives. Dyes Pigm 91:442–445
Chen J, Liu C, Zhang J, Ding W, Zhou M, Wu F (2013) A novel chemodosimeter for fluoride ions based on deprotonation of the C–H group followed by an autoxidative decyanation process. Chem Commun 49:10814–10816
Yuan L, Lin W, Zheng K, He L, Huang W (2013) Far-red to near infrared analyte-responsive fluorescent probes based on organic fluorophore platforms for fluorescence imaging. Chem Soc Rev 42:622–661
Shi Z, Han X, Hu W, Bai H, Peng B, Ji L, Fan Q, Li L, Huang W (2020) Bioapplications of small molecule Aza-BODIPY: from rational structural design to in vivo investigations. Chem Soc Rev 49:7533–7567
Gao Z, Han B, Chen K, Sun J, Hou X (2017) A novel single- fluorophore-based ratiometric fluorescent probe for direct detection of isocyanates in air. Chem Commun 53:6231–6234
Xie Y, Ding Y, Li X, Wang C, Hill JP, Ariga K, Zhang W, Zhu W (2012) Selective, sensitive and reversible turn-on fluorescent cyanide probes based on 2,2-dipyridylaminoanthracene-Cu2+ ensembles. Chem Commun 48:11513–11515
Singh G, Kaur A, Sharma M, Bhalla V, Singh D, Arora S, Kumar M (2020) Reversible detection of hypochlorite using the deprotonation-protonation strategy: a search for new building blocks. Mater Adv 1:1347–1353
Acknowledgements
We thank the Natural Science Foundation of Shanghai (No. 17ZR1429900) and the Opening Fund of Shanghai Key Laboratory of Chemical Biology for financial support.
Funding
The Natural Science Foundation of Shanghai (No. 17ZR1429900) and the Opening Fund of Shanghai Key Laboratory of Chemical Biology.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. HS carried out the experiments, XC, TZ, ZC, YH and HZ conducted the data analyses, ZW and CL wrote and edited the paper.
Corresponding authors
Ethics declarations
Ethics Approval
Not Applicable.
Consent to Participate
Not Applicable.
Consent for Publication
Not Applicable.
Conflict of Interest
There are no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shao, H., Chen, X., Zhu, T. et al. A CH-Controlled Colorimetric Probe Based on Anthracene Carboximide for Near-Infrared Cyanide Detection. J Fluoresc 31, 1863–1869 (2021). https://doi.org/10.1007/s10895-021-02816-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-021-02816-y