Abstract
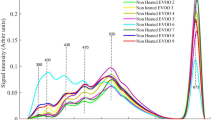
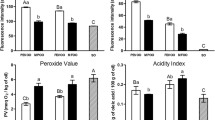
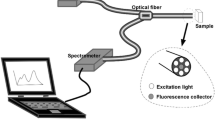
The olive oil production in Pakistan has recently been started with the cultivation of exotic cultivars that are successfully adapted at Barani Agriculture Research center (BARI), Chakwal, Pakistan in Potohar valley. Therefore, characterization of extra virgin olive oil (EVOO) from this agro-climatic region is mandatory in establishing its biochemical profile and thermal stability. Seventeen monovarietal EVOOs extracted from these cultivars were analysed using synchronous fluorescence spectroscopy (SFS) and subjected to heating at 115, 150 and 170 °C for 15 min to identify their thermal stability. SFS emission spectra differentiated EVOOs on the basis of phenolic compounds that are denatured at high temperature, further chlorophyll contents also decreased with increasing temperature. The strong emission at ca. 351 nm, suggested to be vanillic acid, 391–471 nm for blue green region (BGR) assigned to other phenolic compounds and two peaks at 672 and 723 nm for chlorophyll became the bases for grouping through Hierarchical clustering. Most of the EVOOs were stable at 150 °C but showed denatured spectra at 170 °C, the only EVOO extracted from Spanish cultivar Arbequina was found to have moderate fluorescence emission from both vanillic acid and BGR that are more likely to impart oxidative stability even after heating at 170 °C, also confirmed by lowest values of specific extinction co-efficient (K232 and K270). Moreover, variation in phenolic contents of Arbequina EVOO was observed with different harvesting stages and the early harvested olives produced more thermally stable oil as compared to late harvested olives. Arbequina oil grown in Pakistan can be better suited for cooking at high temperatures, moreover can be blended with other monovarietal EVOOs to enhance the nutritional benefits and thermal stability.







Similar content being viewed by others
References
Laincer F, Iaccarino N, Amato J, Pagano B, Pagano A, Tenore G, Tamendjari A, Rovellini P, Venturini S, Bellan G, Ritieni A, Mannina L, Novellino E, Randazzo A (2016) Characterization of monovarietal extra virgin olive oils from the province of Béjaïa ( Algeria ). FRIN 89:1123–1133. https://doi.org/10.1016/j.foodres.2016.04.024
Decker EA, Warner K, Richards MP, Shahidi F (2005) Measuring antioxidant effectiveness in food. J Agric Food Chem 53:4303–4310
Moreno JA, Lopez-Miranda J, Gomez P, Benkhalti F, El Boustani ES, Perez-Jimenez F (2004) Effect of phenolic compounds of virgin olive oil on LDL oxidation resistance med. Clin. 120:128–131
FAO (2010) The second report on the state of the world’s plant genetic resources for food and agriculture. FAO, Rome
Kalogeropoulos N, Tsimidou MZ (2014) Antioxidants in Greek virgin olive oils. Antioxidants. 3:387–413
Boskou D (2015) 1-Olive oil: properties and processing for use in food. In: Talbot G (ed) Specialty oils and fats in food and nutrition, 1st edn. Woodhead, Cambridge, UK, pp 3–38 ISBN 9781782423768
Navarra G, Cannas M, Amico MD et al (2011) Thermal oxidative process in extra-virgin olive oils studied by FTIR , rheology and time-resolved luminescence. Food Chem 126:1226–1231. https://doi.org/10.1016/j.foodchem.2010.12.010
Ahmed M, Pickova J, Ahmad T, Liaquat M, Farid A, Jahangir M (2016) Oxidation of lipids in foods. Polish Journal of Food and Nutrition Science 13:87–100. https://doi.org/10.17582/journal.sja/2016.32.3.230.23810
Barbarisi C, Di Stasio M, La Cara F, Nazzaro M, Siano F, Coppola R et al (2014) Shelf-life of extra virgin olive oils from southern Italy. Curr Nutr Food Sci 10:1–7
Krichene D, Salvador MD, Fregapane G (2015) Stability of virgin olive oil phenolic compounds during long-term storage (18 Months) at temperatures of 5−50 °C-J. Agric Food Chem 63:6779–6786
Visioli F, Galli C (1998) Olive oil phenols and their potential effects on human health. J Agric Food Chem 46:4292–4296
Volf I, Ignat I, Neamtu M, Popa VI (2014) Thermal stability, antioxidant activity, and photo-oxidation of natural polyphenols. Chem Pap 68:121–129
Franco MN, Galeano-Díaz T, López O, Fernández-Bolaños JG, Sánchez J, De Miguel C, Gil MV, Martín-Vertedor D (2014) Phenolic compounds and antioxidant capacity of virgin olive oil. Food Chem 163:289–298
Aparicio R, Aparicio-ruíz R (2000) Authentication of vegetable oils by chromatographic technique. J Chromatogra A 881:93–104
Del Giovine L, Fabietti F (2005) Copper chlorophyll in olive oils: identification and determination by LIF capillary electrophoresis. Food Control 16:267–272
Kyriakidis NB, Skarkalis P (2000) Fluorescence spectra measurement of olive oil and other vegetable oil. J AOAC Int 83:1435–1439
Sikorska E, Khmelinski I, Sikorski M (2012) Analysis of olive oils by fluorescence spectroscopy: methods and applications. In: Boskou D (ed) Olive oil–constituents, quality, health properties and bioconversions. Intech, Rijeka
Navarra G, Cannas M, Amico MD, Giacomazza D, Militello V, Vaccaro L, Leone M (2001) Thermal oxidative process in extra-virgin olive oils studied by FTIR, rheology and time-resolved luminescence. Food Chem 126:1226–1231 21
Hernández López M, Algarra González M, Isabel López Molina M (1999) Synchronous-derivative phosphorimetric determination of 1-and 2-naphthol in irrigation water by employing β-cyclodextrin. Talanta 49:679–689. https://doi.org/10.1016/S0039-9140(99)00067-3
Dupuy N, Le Dreau Y, Ollivier D, Artaud J, Pinatel C, Kister J (2005) Origin of French virgin olive oil registered designation of origins predicted by chemometric analysis of synchronous excitation-emission fluorescence spectra. J Agri Food Chem 53:9361–9368
Zandomeneghi M, Zandomeneghi G (2005) Comment on cluster analysis applied to the exploratory analysis of commercial spanish olive oils by means of excitation emission fluorescence spectroscopy. J Agri Food Chem 53:5829–5830
Tena N, Garcia-Gonzalez DL, Aparicio R (2009) Evaluation of virgin olive oil thermal deterioration by fluorescence spectroscopy. J Agri Food Chem 57:10505–10511
http://barichakwal.org/?page_id=3510 accessed on Jan., 2018
Iqbal MA, Hafiz IA, Abbasi NA, Shah KN (2019) Morphological, phenological characterization and adaptability of exotic olive cultivars in district Chakwal, Pakistan. Pak J Agric Sci 56:587–594. https://doi.org/10.21162/PAKJAS/19.8159
Žiak Ľ, Sádecká J, Májek P, Hroboňová K (2014) Simultaneous determination of phenolic acids and scopoletin in brandies using synchronous fluorescence spectrometry coupled with partial least squares. Food Anal Method 7:563–570
Zandomeneghi M, Carbonaro L, Caffarata C (2005) Fluorescence of vegetable oils: olive oils. J Agric Food Chem 53:759–766
Cabrera-Bañegil M, Hurtado-Sánchez MC, Galeano-Díaz T, Durán-Merás I (2017a) Front-face fluorescence spectroscopy combined with second-order multivariate algorithms for the quantification of polyphenols in red wine samples. Food Chem 220:168–176
Cabrera-Bañegil M, Martín-Vertedor D, Boselli E, Durán-Merás I (2018) Control of olive cultivar irrigation by front-face fluorescence excitation-emission matrices in combination with PARAFAC. J Food Compos Anal 69:189–196. https://doi.org/10.1016/j.jfca.2018.01.021
Alarcón-Flores MI, Romero-González R, Frenich AG, Vidal JLM (2012) Analysis of phenolic compounds in olive oil by solid-phase extraction and ultra-high performance liquid chromatography-tandem mass spectrometry. Food Chem 134:2465–2472
Tothova J, Sádecká J, Májek P (2009) Total luminescence spectroscopy for differentiating between brandies and wine distillates Czech. J Food Sci 27:425–43232
Cabrera-Bañegil M, del Hurtado-Sánchez MC, Galeano-Díaz T, Durán-Merás I (2017) Front-face fluorescence spectroscopy combined with second-order multivariate algorithms for the quantification of polyphenols in red wine samples. Food Chem 220:168–176 https://doi.org/10.1016/j.foodchem.2016.09.152
Rodriguez-Delgado MA, Malovana S, Perez JP, Borges T, Garcia Montelongo FJ (2001) Separation of phenolic compounds by high-performance liquid chromatography with absorbance and fluorimetric detection. J Chromatogr A 912:249–257
Aparicio R, Harwood J (2013) Handbook of olive oil: analysis and properties, 2nd edn. Springer New York LLC, New York. https://doi.org/10.1007/978-1-4614-7777-8
Morales F, Cerovic ZG, Moya I (1994) Characterization of blue-green fluorescence in the mesophyll of sugar beet (Beta vulgaris L) leaves affected by iron deficiency. Plant Physiol 106 127–133
Lichtenthaler HK, Schweiger J (1998) Cell wall bound ferulic acid, the major substance of the blue-green fluorescence emission of plants. J Plant Physiol 152:272–282
Cerovic ZG, Samson G, Morales F, Tremblay N, Moya I (1999) Ultraviolet-induced fluorescence for plant monitoring: present state and prospects. Agronomie 19:543–578
Meyer S, Cartelat A, Moya I, Cerovic ZG (2003) UV-induced blue-green and far-red fluorescence along wheat leaves: a potential signature of leaf ageing. J Exp Bot 54:757–769
Chappelle EW, McMurtrey JE, Wood FM, Newcomb WW (1984) Laser-induced fluorescence of green plants. 2: LIF caused by nutrient deficiencies in corn. Appl Opt 23:139–142
Snowden MC (2015) Effects of blue and green light on plant growth and development at low and high photosynthetic photon flux Utah State Univ. Digit. 57
Pedrós R, Moya I, Goulas Y, Jacquemoud S (2008) Chlorophyll fluorescence emission spectrum inside a leaf. Photochem Photobiol Sci 7:498–502
Atta BM, Saleem M, Ali H, Arshad HMI, Ahmed M (2018) Chlorophyll as a biomarker for early disease diagnosis. Laser Phys 28:065607
Lee E, Ahn H, Choe E (2014) Effects of light and lipids on chlorophyll degradation. Food Sci Biotech 23:1061–1065
Rasul HH, Inanc AL (2014) Thermal stability of chlorophyll pigments in virgin olive oil. KSU J Nat Sci 17:34–40
Réblová Z (2012) Effect of temperature on the antioxidant activity of phenolic acids. Czech J Food Sci 30:171–177. https://doi.org/10.17221/57/2011-cjfs
Gasson MJ, Kitamura Y, McLauchlan WR, Narbad A, Parr AJ, Parsons ELH, Payne J, Rhodes MJC, Walton NJ (1998) Metabolism of Ferulic acid to vanillin. J Biol Chem 273:4163–4170. https://doi.org/10.1074/jbc.273.7.4163
Kumar N, Pruthi V (2014) Potential applications of ferulic acid from natural sources. Biotechnol Reports 4:86–93. https://doi.org/10.1016/j.btre.2014.09.002
Sikorska E, Romaniuk A, Khmelinskii IV, Herance R, Bourdelande JL, Sikorski M, Koziol J (2004) Characterization of edible oils using total luminescence spectroscopy. J Fluoresc 14:25–35
Grigoriadou D, Tsimidou MZ (2006) Quality control and storage studies of virgin olive oil: exploiting UV spectrophotometry potential. Eur J Lipid Sci Tech 108:61–69
Gomez NA, Abonia R, Cadavid H, Vargas IH (2011) Chemical and spectroscopic characterization of a vegetable oil used as dielectric coolant in distribution transformers. J Brazilian Chem Society 22:2292–2303
Choe E, Min DB (2006) Mechanisms and factors for edible oil oxidation comprehensive reviews in food. Sci Food Safety 5:169–186
EEC Commission Regulation: No 2568/91 on the characteristics of olive oil and olive-residue oils and on the relevant methods of analysis L248 September 5 1991
Allouche Y, Jimenez A, Gaforio JJ, Uceda M, Beltran G (2007) How heating affects extra virgin olive oil quality ´ indexes and chemical composition. J Agric Food Chem 55:9646–9654
Malvis A, Šimon P, Dubaj T, Sládková A, Ház A, Jablonský M, Sekretár S, Schmidt Š, Kreps F, Burčová Z, Hodaifa G, Šurina I (2019) Determination of the thermal oxidation stability and the kinetic parameters of commercial extra virgin olive oils from different varieties. J Chem 2019:1–8. https://doi.org/10.1155/2019/4567973
Borges TH, Pereira JA, Cabrera-Vique C, Lara L, Oliveira AF, Seiquer I (2017) Characterization of Arbequina virgin olive oils produced in different regions of Brazil and Spain: physicochemical properties, oxidative stability and fatty acid profile. Food Chem 215:454–462. https://doi.org/10.1016/j.foodchem.2016.07.162
Uylaşer V (2015) Changes in phenolic compounds during ripening in Gemlik variety olive fruits obtained from different locations. CyTA – J Food 13:167–173
Gandul-Rojas B, Cepero MLR, Minguez-Mosquera MI (1999) Chlorophyll and carotenoid patterns in olive fruits, Olea europaea cv. Arbequina. J Agri Food Chem 47:2207–2212
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Figure 1
Comparative normalized synchronous fluorescence emission spectra of unheated and heated Picholine EVOO at three different temperatures for 15 min (PNG 5803 kb)
Supplementary Figure 2
Comparative normalized synchronous fluorescence emission spectra of unheated and heated Gemlik EVOO at three different temperatures for 15 min (PNG 5803 kb)
Supplementary Figure 3
Comparative normalized synchronous fluorescence emission spectra of unheated and heated Nocellara EVOO at three different temperatures for 15 min showing denatured spectrum at 170 °C (PNG 5803 kb)
Supplementary Figure 4
Comparative normalized synchronous fluorescence emission spectra of unheated and heated Chemlali EVOO at three different temperatures for 15 min showing denatured spectrum at 170 °C (PNG 5803 kb)
Supplementary Figure 5
Comparative normalized synchronous fluorescence emission spectra of unheated and heated Manzanilla EVOO at three different temperatures for 15 min showing denatured spectrum at 170 °C (PNG 5803 kb)
Supplementary Figure 6
Comparative normalized synchronous fluorescence emission spectra of unheated and heated VP-(Correggiolo) EVOO (11) untreated and thermally treated at three different temperatures for 15 min (PNG 5803 kb)
Rights and permissions
About this article
Cite this article
Ali, H., Iqbal, M.A., Atta, B.M. et al. Phenolic Profile and Thermal Stability of Monovarietal Extra Virgin Olive Oils Based on Synchronous Fluorescence Spectroscopy. J Fluoresc 30, 939–947 (2020). https://doi.org/10.1007/s10895-020-02538-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-020-02538-7