Abstract
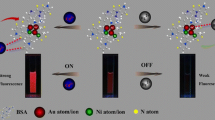
Interest in biosensing platforms using protein fluorescent gold nanoclusters (FGNCs) has grown significantly in the past due to the unique optical properties they offer. This study investigates the interaction of metal ions with FGNCs, and the structural modifications brought about by the interaction resulting in fluorescence changes of the cluster and its successful application in the detection of two heavy metals, cobalt and cadmium. The binding of cobalt and cadmium to FGNCs synthesized from BSA significantly altered the secondary structure of the protein, causing a change in its hydrophobicity. It also resulted in a change in fluorescence properties of FGNCs by intersystem crossing (ICT) and fluorescence resonance energy transfer (FRET). Cobalt and cadmium could successfully be detected in the range of 5–165 ng/mL (R2 = 0.95) and 20–1000 ng/ mL (R2 = 0.91), respectively, with appreciable sensitivity. The principle was also applied for the detection of Vitamin B12 in commercially available ampoules, validating the proposed method.

Proposed detection method of cadmium and cobalt using FGNCs.







Similar content being viewed by others
References
Chen LY, Wang CW, Yuan Z, H.T. (2015) Chang, fluorescent gold nanoclusters: recent advances in sensing and imaging. Anal Chem 87:216–229. https://doi.org/10.1021/ac503636j
Chaudhari K, Baksi A, Pradeep T (2012) Protein-protected luminescent noble metal quantum clusters: an emerging trend in atomic cluster nanoscience. Nanotechnol Rev 3:1–16. https://doi.org/10.3402/nano.v3i0.14767
Xu Y, Sherwood J, Qin Y, Crowley D, Bonizzoni M, Bao Y (2014) The role of protein characteristics in the formation and fluorescence of Au nanoclusters. Nanoscale. 6:1515–1524. https://doi.org/10.1039/c3nr06040c
Chen Y, Wang Y, Wang C, Li W, Zhou H, Jiao H, Lin Q, Yu C (2013) Papain-directed synthesis of luminescent gold nanoclusters and the sensitive detection of Cu2+. J Colloid Interface Sci 396:63–68. https://doi.org/10.1016/j.jcis.2013.01.031
Fernández TD, Pearson JR, Leal MP, Torres MJ, Blanca M, Mayorga C, Guével XL (2015) Intracellular accumulation and immunological properties of fluorescent gold nanoclusters in human dendritic cells. Biomaterials. 43:1–12. https://doi.org/10.1016/j.biomaterials.2014.11.045
Zhang XD, Wu D, Shen X, Liu PX, Fan FY, Fan SJ (2012) In vivo renal clearance, biodistribution, toxicity of gold nanoclusters. Biomaterials. 33:4628–4638. https://doi.org/10.1016/j.biomaterials.2012.03.020
Volden S, Lystvet SM, Halskau Ø, Glomm WR (2012) Generally applicable procedure for in situ formation of fluorescent protein-gold nanoconstructs. RSC Adv:11704–11711. https://doi.org/10.1039/c2ra21931j
Wen F, Dong Y, Feng L, Wang S, Zhang S, Zhang X (2011) Horseradish peroxidase functionalized fluorescent gold nanoclusters for hydrogen peroxide sensing. Anal Chem 83:1193–1196. https://doi.org/10.1021/ac1031447
Tay CY, Yu Y, Setyawati MI, Xie J, Leong DT (2014) Presentation matters: identity of gold nanocluster capping agent governs intracellular uptake and cell metabolism. Nano Res 7:805–815. https://doi.org/10.1007/s12274-014-0441-z
Gao S, Chen D, Li Q, Ye J, Jiang H, Amatore C, Wang X (2014) Near-infrared fluorescence imaging of cancer cells and tumors through specific biosynthesis of silver nanoclusters. Sci Rep 4:4384. https://doi.org/10.1038/srep04384
He X, Gao J, Gambhir SS, Cheng Z (2010) Near-infrared fluorescent nanoprobes for cancer molecular imaging: status and challenges. Trends Mol Med 16:574–583. https://doi.org/10.1016/j.molmed.2010.08.006
Kawasaki H, Hamaguchi K, Osaka I, Arakawa R (2011) Ph-dependent synthesis of pepsin-mediated gold nanoclusters with blue green and red fluorescent emission. Adv Funct Mater 21:3508–3515. https://doi.org/10.1002/adfm.201100886
Liu CL, Wu HT, Hsiao YH, Lai CW, Shih CW, Peng YK et al (2011) Insulin-directed synthesis of fluorescent gold nanoclusters: preservation of insulin bioactivity and versatility in cell imaging. Angew Chem Int Ed 50:7056–7060. https://doi.org/10.1002/anie.201100299
Lee H, Lee K, Kim IK, Park TG (2009) Fluorescent gold nanoprobe sensitive to intracellular reactive oxygen species. Adv Funct Mater 19:1884–1890. https://doi.org/10.1002/adfm.200801838
Xie J, Zheng Y, Ying JY (2009) Protein-directed synthesis of highly fluorescent gold nanoclusters. J Am Chem Soc 131:888–889. https://doi.org/10.1021/ja806804u
Liu Y, Ai K, Cheng X, Huo L, Lu L (2010) Gold-nanocluster-based fluorescent sensors for highly sensitive and selective detection of cyanide in water. Adv Funct Mater 20:951–956. https://doi.org/10.1002/adfm.200902062
Zhang L, Wang E (2014) Metal nanoclusters: new fluorescent probes for sensors and bioimaging. Nano Today 9:132–157. https://doi.org/10.1016/j.nantod.2014.02.010
Lin Y, Tseng W (2010) Ultrasensitive sensing of Hg 2+ and CH 3 Hg + based on the fluorescence quenching of lysozyme type VI-stabilized gold nanoclusters. Anal Chem 82:9194–9200
Durgadas CV, Sharma CP, Sreenivasan K (2011) Fluorescent gold clusters as nanosensors for copper ions in live cells. Analyst 136:933–940. https://doi.org/10.1039/c0an00424c
Xia X, Long Y, Wang J (2013) Glucose oxidase-functionalized fluorescent gold nanoclusters as probes for glucose. Anal Chim Acta 772:81–86. https://doi.org/10.1016/j.aca.2013.02.025
Venkatesh V, Shukla A, Sivakumar S, Verma S (2014) Purine-stabilized green fluorescent gold nanoclusters for cell nuclei imaging applications. ACS Appl Mater Interfaces 6:2185–2191. https://doi.org/10.1021/am405345h
Wang Y, Chen J, Irudayaraj J (2011) Nuclear targeting dynamics of gold nanoclusters for enhanced therapy of HER2 + breast cancer. ACS Nano 5:9718–9725. https://doi.org/10.1021/nn2032177
Chen L, Wang C, Yuan Z, Chang H (2015) Fluorescent gold nanoclusters: recent advances in sensing and imaging. Anal Chem 87(1):216–229. https://doi.org/10.1021/ac503636j
Wang HH, Lin CAJ, Lee CH, Lin YC, Tseng YM, Hsieh CL, Shen JL, Chan WH (2011) Fluorescent gold nanoclusters as a biocompatible marker for in vitro and in vivo tracking of endothelial cells. ACS Nano 5:4337–4344. https://doi.org/10.1021/nn102752a
Hu L, Deng L, Alsaiari S, Zhang D, Khashab NM (2014) “Light-on” sensing of antioxidants using gold nanoclusters. Anal Chem 86:4989–4994. https://doi.org/10.1021/ac500528m
Hu D, Sheng Z, Fang S, Wang Y, Gao D, Zhang P et al (2014) Folate receptor-targeting gold nanoclusters as fluorescence enzyme mimetic nanoprobes for tumor molecular colocalization diagnosis. Theranostics 4:142–153. https://doi.org/10.7150/thno.7266
Cho SJ, Maysinger D, Jain M, Röder B, Hackbarth S, Winnik FM (2007) Long-term exposure to CdTe quantum dots causes functional impairments in live cells. Langmuir 23:1974–1980. https://doi.org/10.1021/la060093j
Pan J, Plant JA, Voulvoulis N, Oates CJ, Ihlenfeld C (2010) Cadmium levels in Europe: implications for human health. Environ Geochem Health 32:1–12. https://doi.org/10.1007/s10653-009-9273-2
S.I. Report, Cadmium risks to freshwater life : derivation and validation of low-effect criteria values using laboratory and field studies scientific investigations report 2006–5245, Quality. (2010)
Godt J, Scheidig F, Grosse-Siestrup C, Esche V, Brandenburg P, Reich A, Groneberg DA (2006) The toxicity of cadmium and resulting hazards for human health. J Occup Med Toxicol 1:22. https://doi.org/10.1186/1745-6673-1-22
Xie J, Zheng Y, Ying JY (2010) Highly selective and ultrasensitive detection of Hg(2+) based on fluorescence quenching of Au nanoclusters by Hg(2+)-Au(+) interactions. Chem Commun (Camb) 46:961–963. https://doi.org/10.1039/b920748a
Wang ZX, Guo YX, Ding SN (2015) Fluorometric determination of cadmium (II) and mercury(II) using nanoclusters consisting of a gold-nickel alloy. Microchim Acta 182:2223–2231. https://doi.org/10.1007/s00604-015-1563-z
Bal W, Christodoulou J, Sadler PJ, Tucker A (1998) Multi-metal binding site of serum albumin. J Inorg Biochem 70:33–39. https://doi.org/10.1016/S0162-0134(98)00010-5
Wei H, Wang Z, Yang L, Tian S, Hou C, Lu Y (2010) Lysozyme-stabilized gold fluorescent cluster: synthesis and application as Hg(2+) sensor. Analyst. 135:1406–1410. https://doi.org/10.1039/c0an00046a
Liu H, Xu Z, Liu X, Xi P, Zeng Z (2009) Analysis of binding interaction between bovine serum albumin and the cobalt (II) complex with salicylaldehyde-2-phenylquinoline-4-carboylhydrazone. Chem Pharm Bull (Tokyo) 57:1237–1242. https://doi.org/10.1248/cpb.57.1237
Sokołowska M, Wszelaka-Rylik M, Poznański J, Bal W (2009) Spectroscopic and thermodynamic determination of three distinct binding sites for Co (II) ions in human serum albumin. J Inorg Biochem 103:1005–1013. https://doi.org/10.1016/j.jinorgbio.2009.04.011
Liang H, Huang J, Tu C, Zhang M, Zhou Y, Shen P (2001) The subsequent effect of interaction between Co 2 1 and human serum albumin or bovine serum albumin. J Inorg Biochem 85:167–171
Qu SS, Liu Y, Wang TZ, Gao WY (2002) Thermodynamics of binding of cadmium to bovine serum albumin. Chemosphere. 46:1211–1214. https://doi.org/10.1016/S0045-6535(01)00213-2
Andersen O (1983) Chelation of cadmium. Environ Health Perspect 54:249–266. https://doi.org/10.1289/ehp.8454249
Martins E, Drakenberg T (1982) Cadmium (II), Zinc (II), and Copper (II) ions binding to bovine serum albumin. A 113Cd NMR study. Inorg Chim Acta 67:71–74
Cheng T, Xu Y, Zhang S, Zhu W, Qian X, Duan L (2008) A highly sensitive and selective OFF-ON fluorescent sensor for cadmium in aqueous solution and living cell. J Am Chem Soc 130:16160–16161. https://doi.org/10.1021/ja806928n
Williams NJ, Gan W, Reibenspies JH, Hancock RD (2009) Possible steric control of the relative strength of chelation enhanced fluorescence for zinc (II) compared to cadmium (II): metal ion complexing properties of tris(2-quinolylmethyl) amine, a crystallographic, UV-visible, and fluorometric study. Inorg Chem 48:1407–1415. https://doi.org/10.1021/ic801403s
Peng X, Du J, Fan J, Wang J, Wu Y, Zhao J et al (2007) A selective fluorescent sensor for imaging Cd2+ in living cells. J Am Chem Soc 129:1500–1501. https://doi.org/10.1021/ja0643319
Yang Y, Cheng T, Zhu W, Xu Y, Qian X (2011) Highly selective and sensitive near-infrared fluorescent sensors for cadmium in aqueous solution. Org Lett 13:264–267. https://doi.org/10.1021/ol102692p
Huang C-C, Yang Z, Lee K-H, Chang H-T (2007) Synthesis of highly fluorescent gold nanoparticles for sensing mercury (II). Angew Chem 119:6948–6952. https://doi.org/10.1002/ange.200700803
Wei H, Wang Z, Zhang J, House S, Gao Y-G, Yang L, Robinson H, Tan LH, Xing H, Hou C, Robertson IM (2011) Time-dependent, protein-directed growth of gold nanoparticles within a single crystal of lysozyme. Nat Nanotechnol 6:93–97. https://doi.org/10.1038/nnano.2010.280
Russell BA, Kubiak-Ossowska K, Mulheran PA, Birch DJS, Chen Y (2015) Locating the nucleation sites for protein encapsulated gold nanoclusters: a molecular dynamics and fluorescence study. Phys Chem Chem Phys 17:21935–21941. https://doi.org/10.1039/c5cp02380g
Li J, Zhu JJ, Xu K (2014) Fluorescent metal nanoclusters: from synthesis to applications. TrAC - Trends Anal Chem 58:90–98. https://doi.org/10.1016/j.trac.2014.02.011
Sadler PJ, Tucker A, Viles JH (1994) Involvement of a lysine residue in the N-terminal Ni2+ and Cu2+ binding site of serum albumins. Comparison with Co2+, Cd2+ and Al3+. Eur J Biochem 220:193–200. https://doi.org/10.1111/j.1432-1033.1994.tb18614.x
Zhang YZ, Li HR, Dai J, Chen WJ, Zhang J, Liu Y (2010) Spectroscopic studies on the binding of cobalt (II) 1,10-Phenanthroline complex to bovine serum albumin. Biol Trace Elem Res 135:136–152. https://doi.org/10.1007/s12011-009-8502-y
Yifei K, Chen J, Gao F, Brydson R, Johnson B, Heath G, Zhang Y, Wu L, Zhou D (2013) Near-infrared fluorescent ribonuclease-A-encapsulated gold nanoclusters: preparation, characterization, cancer targeting and imaging. Nanoscale 5(3):1009–1017. https://doi.org/10.1039/C2NR32760K
Moriyama Y, Ohta D, Hachiya K, Mitsui Y, Takeda K (1996) Fluorescence behavior of tryptophan residues of bovine and human serum albumins in ionic surfactant solutions: a comparative study of the two and one tryptophan(s) of bovine and human albumins. J Protein Chem 15:265–272. https://doi.org/10.1007/BF01887115
Ghisaidoobe ABT, Chung SJ (2014) Intrinsic tryptophan fluorescence in the detection and analysis of proteins: a focus on förster resonance energy transfer techniques. Int J Mol Sci 15:22518–22538. https://doi.org/10.3390/ijms151222518
Vivian JT, Callis PR (2001) Mechanisms of tryptophan fluorescence shifts in proteins. Biophys J 80:2093–2109. https://doi.org/10.1016/S0006-3495(01)76183-8
Hospes M, Hendriks J, Hellingwerf KJ (2013) Tryptophan fluorescence as a reporter for structural changes in photoactive yellow protein elicited by photo-activation. Photochem Photobiol Sci 12:479–488. https://doi.org/10.1039/C2PP25222H
Wang J, Yang X, Wang J, Xu C, Zhang W, Liu R, Zong W (2016) Probing the binding interaction between cadmium (II) chloride and lysozyme. New J Chem 40(4):3738–3746
Raut S, Chib R, Butler S, Borejdo J, Gryczynski Z, Gryczynski I (2014) Evidence of energy transfer from tryptophan to BSA/HSA protected gold nanoclusters. Methods Appl Fluores 2:35004. https://doi.org/10.1088/2050-6120/2/3/035004
Vinayaka AC, Thakur MS (2011) Photoabsorption and resonance energy transfer phenomenon in CdTe-protein bioconjugates: an insight into QD-biomolecular interactions. Bioconjug Chem 22:968–975. https://doi.org/10.1021/bc200034a
Medintz IL, Mattoussi H (2009) Quantum dot-based resonance energy transfer and its growing application in biology. Phys Chem Chem Phys 11:17–45. https://doi.org/10.1039/b813919a
Medintz IL, Clapp AR, Mattoussi H, Goldman ER, Fisher B, Mauro JM (2003) Self-assembled nanoscale biosensors based on quantum dot FRET donors. Nat Mater 2:630–638. https://doi.org/10.1038/nmat961
Chong EZ, Matthews DR, Summers HD, Njoh KL, Errington RJ, Smith PJ (2007) Development of FRET-based assays in the far-red using CdTe quantum dots. Biomed Res Int. https://doi.org/10.1155/2007/54169
Patel RC, Lange DC, Patel YC (2002) Photobleaching fluorescence resonance energy transfer reveals ligand-induced oligomer formation of human somatostatin receptor subtypes. Methods. 27:340–348. https://doi.org/10.1016/S1046-2023(02)00092-0
Wang S, Liu P, Qin Y, Chen Z, Shen J (2016) Rapid synthesis of protein conjugated gold nanoclusters and their application in tea polyphenol sensing. Sensors Actuators B Chem 223:178–185. https://doi.org/10.1016/j.snb.2015.09.058
Dieaconu M, Loanid A, Iftimie S, Antohe S (2012) UV-absorption mechanisms of Ni2+-binding bovine serum albumin. Dig J Nanomater Biostruct 7:1125–1137
Shaw AK, Pal SK (2008) Spectroscopic studies on the effect of temperature on pH-induced folded states of human serum albumin. J Photochem Photobiol B Biol 90:69–77. https://doi.org/10.1016/j.jphotobiol.2007.11.003
Selvakumar LS, Ragavan KV, Abhijith KS, Thakur MS (2013) Immunodipstick based gold nanosensor for vitamin B12 in fruit and energy drinks. Anal Methods 5:1806. https://doi.org/10.1039/c3ay26320g
Acknowledgements
The authors gratefully acknowledge Director CSIR-CFTRI for providing necessary facilities for the work. Mr. Akshath U. S. acknowledges the Indian Council of Medical Research (ICMR) for providing senior research fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akshath, U.S., Bhatt, P. & Singh, S.A. Differential Interaction of Metal Ions with Gold Nanoclusters and Application in Detection of Cobalt and Cadmium. J Fluoresc 30, 537–545 (2020). https://doi.org/10.1007/s10895-020-02509-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-020-02509-y