Abstract
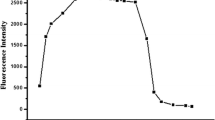
In this paper, probe 1 based on N, N-bis (pyridin-2-ylmethyl) aniline is prepared as an effective fluorescent probe for Cu2+. It exhibits good sensitivity and selectivity for Cu2+ over other metal ions in aqueous solution. The detection limit of probe 1 is 56 nM. Furthermore, probe 1 is pH-insensitive under near-neutral conditions and can work well in environmental samples.





Similar content being viewed by others
References
Gaggelli E, Kozlowski H, Valensin D, Valensin G (2006) Copper homeostasis and neurodegenerative disorders (Alzheimer's, prion, and Parkinson's diseases and amyotrophic lateral sclerosis. Chem Rev 106(6):1995–2044
Tarighat MA, Mohammadizadeh MR, Abdi GJ (2013) Simultaneous spectrophotometric determination of Cd2+, Cu2+, and Zn2+in Rice and vegetal samples with dimethyl-spiro[isobenzofurane-1,6′-pyrorolo[2,3-d]pyrimidine]-2′,3,4,5′(1′H,3′H,7′H)tetraone using wavelet transformation–feed forward neural networks. J Agric Food Chem 61(28):6832–6840
Hua C, Zhang WH, Almeida SRMD, Ciampi S, Gloria D, Liu GZ, Harper JB, Gooding JJ (2012) A novel route to copper(II) detection using ‘click’ chemistry-induced aggregation of gold nanoparticles. Analyst 137(1):82–86
Ciesienski KL, Hyman LM, Derisavifard S, Franz KJ (2010) Toward the detection of cellular copper(II) by a light-activated fluorescence increase. Inorg Chem 49(15):6808–6810
Millhauser GL (2004) Copper binding in the prion protein. Acc Chem Res 37(2):79–85
Que EL, Domaille DW, Chang CJ (2008) Metals in neurobiology: probing their chemistry and biology with molecular imaging. Chem Rev 108(5):1517–1549
Lee JC, Gray HB, Winkler JR (2008) Copper(II) binding to α-Synuclein, the Parkinson’s protein. J Am Chem Soc 130(22):6898–6899
Bull PC, Thomas GR, Rommens JM, Forbes JR, Cox DW (1993) The Wilson disease gene is a putative copper transporting P–type ATPase similar to the Menkes gene. Nat Genet 5(4):327–337
Taki M, Iyoshi S, Ojida A, Hamachi I, Yamamoto Y (2010) Development of highly sensitive fluorescent probes for detection of intracellular copper(I) in living systems. J Am Chem Soc 132(17):5938–5939
Liu J, Lu Y (2007) A DNAzyme catalytic beacon sensor for paramagnetic Cu2+ ions in aqueous solution with high sensitivity and selectivity. J Am Chem Soc 129(32):9838–9839
Chan MS, Huang SD (2000) Direct determination of cadmium and copper in seawater using a transversely heated graphite furnace atomic absorption spectrometer with Zeeman-effect background corrector. Talanta 51(2):373–380
Gattas-Asfura KM, Leblanc RM (2003) Peptide-coated CdS quantum dots for the optical detection of copper(II) and silver(I. Chem Commun 21:2684–2685
Yantasee W, Hongsirikarn K, Warner CL, Choi D, Sangvanich T, Toloczko MB, Warner MG, Fryxell GE, Addleman RS, Timchalk C (2008) Direct detection of Pb in urine and Cd, Pb, Cu, and Ag in natural waters using electrochemical sensors immobilized with DMSA functionalized magneticnanoparticles. Analyst 133(3):348–355
Huang CC, Chang HT (2007) Parameters for selective colorimetric sensing of mercury(II) in aqueous solutions using mercaptopropionic acid-modified gold nanoparticles. Chem Commun 12:1215–1217
Lu CH, Wang YW, Ye SL, Chen GN, Yang HH (2012) Ultrasensitive detection of Cu2+ with the naked eye and application in immunoassays. NPG Asia Materials 4:1
XY X, Daniel WL, Wei W, Mirkin CA (2010) Colorimetric Cu2+ detection using DNA-modified gold-nanoparticle aggregates as probes and click chemistry. Small 6(5):623–626
Meng QT, Zhang XL, He C, He GJ, Zhou P, Duan CY (2010) Multifunctional mesoporous silica material used for detection and adsorption of Cu2+ in aqueous solution and biological applications in vitro and in vivo. Adv Funct Mater 20(12):1903–1909
Cheng GH, He M, Peng HY, Hu B (2012) Dithizone modified magnetic nanoparticles for fast and selective solid phase extraction of trace elements in environmental and biological samples priorto their determination by ICP-OES. Talanta 88:507–515
Liu JM, Lin LP, Wang XX, Lin SQ, Cai WL, Zhang LH, Zheng ZY (2012) Highly selective and sensitive detection of Cu2+ with lysine enhancing bovine serum albumin modified-carbon dots fluorescent probe. Analyst 137:2637–2642
Lee JJ, Choi YW, You GR, Lee SY, Cheal K (2015) A phthalazine-based two-in-one chromogenic receptor for detecting Co2+ and Cu2+ in an aqueous environment. Dalton Trans 44:13305–13314
Xu W, Ren C, Teoh CL, Peng J, Gadre SH, Rhee H, Lee CK, Chang Y (2014) An artificial tongue fluorescent sensor array for identification and quantitation of various heavy metal ions. Anal Chem 86(17):8763–8769
Li YM, Zhang XL, Zhu BC, Xue J, Zhu Z, Tan WH (2011) A simple but highly sensitive and selective colorimetric and fluorescent probe for Cu2+ ion in aqueous media. Analyst 136:1124–1128
Yang SL, Xia BY, Zeng XD, Luo SL, Wei WZ, Liu XY (2010) Fabrication of DNA functionalized carbon nanotubes/Cu2+ complex by one-step electrodeposition and its sensitive determination of nitrite. Anal Chim Acta 667(1–2):57–62
Liu J, Lu Y (2007) Colorimetric Cu2+ detection with a ligation DNAzyme and nanoparticles. Chem Commun 48:4872–4874
You GR, Park GJ, Lee JJ, Kim C (2015) A colorimetric sensor for the sequential detection of Cu2+ and CN− in fully aqueous media: practical performance of Cu2+. Dalton Trans 44(19):9120–9129
Wei X, Zhou ZP, Hao TF, Li HJ, YQ X, Lu K, YL W, Dai JD, Pan JM, Yan YS (2015) Highly-controllable imprinted polymer nanoshell at the surface of silica nanoparticles based room-temperature phosphorescence probe for detection of 2,4-dichlorophenol. Anal Chim Acta 870:83–91
Acknowledgments
We gratefully acknowledge assistance from Dr. Hailiang Nie.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(1H-NMR of Compound 1, HRMS of Probe 1 and Job plot of the probe 1 and Cu2+ are shown in Supporting Information. DOC 239 kb)
Rights and permissions
About this article
Cite this article
Liu, X., Zhang, Y. A Readily-Synthesized Fluorescent Probe Based on N, N-Bis (Pyridin-2-Ylmethyl) Aniline for Copper(II) Detection in Aqueous Solution. J Fluoresc 26, 2267–2270 (2016). https://doi.org/10.1007/s10895-016-1922-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1922-0