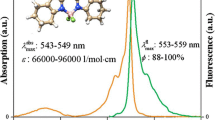
Abstract
In this study, the synthesis of boron dipyrromethene dyes containing mono, bis-2-naphthyloxyhexyloxy and 4-(benzyloxy)phenoxyhexyloxy groups has been reported. Boron dipyrromethene dyes were synthesized from the mono, bis-benzaldehyde derivatives with 2,4-dimethylpyrrole in dichloromethane in the presence of trifluoroacetic, 2,3-dichloro-5,6-dicyano-p-benzoquinon, triethyl amine and boron trifluoride diethyl etherate, respectively. Electrochemical characterization of boron dipyrromethene dyes were carried out with voltammetric measurements. Electrochemical studies show that boron dipyrromethene dyes containing mono, bis-2-naphthyloxyhexyloxy and 4-(benzyloxy)phenoxyhexyloxy groups have reversible one reduction potentials unlike irreversible one oxidation potentials.

ᅟ








Similar content being viewed by others
References
Zhao H, Wang B, Liao J, Wang H, Tan G (2013) Synthesis, spectral, electrochemical properties, and photovoltaic performance of structurally constrained BODIPY dyes with 4,4-dimethyltriphenylamine at the 2,6-positions. Tetrahedron Lett 54:6019–6022
Buyukcakir O, Bozdemir OA, Kolemen S, Erbas S, Akkaya EU (2009) Tetrastyryl-Bodipy dyes: convenient synthesis and characterization of elusive near IR fluorophores. Org Lett 11:4644–4647
Alamiry MAH, Harriman A, Mallon LJ, Ulrich G, Ziessel R (2008) Energy- and charge-transfer processes in a perylene–BODIPY–pyridine tripartite array. Eur J Org Chem 2008:2774–2782
Coşkun A, Akkaya EU (2006) Signal ratio amplification via modulation of resonance energy transfer: proof of principle in an emission ratiometric Hg(II) sensor. J Am Chem Soc 128:14474–14475
Zhang D, Martin V, Garcia-Moreno I, Costela A, Perez-Ojeda ME, Xiao Y (2011) Development of excellent long-wavelength BODIPY laser dyes with a strategy that combines extending π-conjugation and tuning ICT effect. Phys Chem Chem Phys 13:13026–13033
Duran-Sampedro G, Agarrabeitia AR, Garcia-Moreno I, Costela A, Bañuelos J, Arbeloa T, Arbeloa IL, Chiara JL, Ortiz MJ (2012) Chlorinated BODIPYs: Surprisingly efficient and highly photostable laser dyes. Eur J Org Chem 2012:6335–6350
McCusker C, Carroll JB, Rotello VM (2005) Cationic polyhedral oligomeric silsesquioxane (POSS) units as carriers for drug delivery processes. Chem Commun:996–998
Lai RY, Bard AJ (2003) Electrogenerated chemiluminescence 71. Photophysical, electrochemical, and electrogenerated chemiluminescent properties of selected dipyrromethene-BF2 dyes. J Phys Chem B 107:5036–5042
Lim SH, Thivierge C, Nowak-Sliwinska P, Han J, van den Bergh H, Wagnieres G (2010) In vitro and in vivo photocytotoxicity of boron dipyrromethene derivatives for photodynamic therapy. J Med Chem 53:2865–2874
Atilgan S, Ekmekci Z, Dogan AL, Guc D, Akkaya EU (2006) Water soluble distyryl boradiazaindacenes as efficient photosensitizers for photdynamic therapy. Chem Commun 2006:4398–4400
Kumaresan D, Thummel RP, Bura T, Ulrich G, Ziessel R (2009) Color tuning in new metal-free organic sensitizers (bodipys) for dye-sensitized solar cells. Chem Eur J 15:6335–6339
Harriman A, Mallon LJ, Elliot KJ, Haefele A, Ulrich G, Ziessel R (2009) Length dependence for ıntramolecular energy transfer in three- and four-color donor − spacer − acceptor arrays. J Am Chem Soc 131:13375–13386
Monsma FJ, Barton AC, Kang HC, Brasard DL, Haughland RP, Sibley DR (1989) Characterization of novel fluorescent ligands with high affinity for d1and d2 dopaminergic receptors. J Neurochem 52:1641–1644
Bonardi L, Kanaan H, Camerel F, Jolinat P, Retailleau P, Ziessel R (2008) Fine-tuning of yellow or red photo- and electroluminescence of functional difluoro-boradiazaindacene films. Adv Funct Mater 18:401–413
Wasielewski MR (2006) Energy, charge, and spin transport in molecules and self-assembled nanostructures ınspired by photosynthesis. J Organomet Chem 71:5051–5066
Elemans JAAW, van Hameren R, Nolte RJM, Rowan AE (2006) Molecular materials by self-assembly of porphyrins, phthalocyanines, and perylenes. Adv Mater 18:1251–1266
Lakshmi V, Ravikanth M (2013) Photophysical and electrochemical properties of sterically crowded polyarylated boron-dipyrromethenes. Chem Phys Lett 564:93–97
Jiang XD, Zhang H, Zhang Y, Zhao W (2012) Development of non-symmetric thiophene-fused BODIPYs. Tetrahedron 68:9795–9801
Shi WJ, Lo PC, Singh A, Ledoux-Rak I, Ng DKP (2012) Synthesis and second-order nonlinear optical properties of push-pull BODIPY derivatives. Tetrahedron 68:8712–8718
Treibs A, Kreuzer FH (1968) Difluorboryl-Komplexe von di- und Tripyrrylmethenen. Liebigs Ann Chem 718:208–223
Nepomnyashcaii AB, Bard AJ (2012) Electrochemistry and electrogenerated chemiluminescence of BODIPY dyes. Acc Chem Res 45:1844–1853
Burghart A, Kim H, Welch MB, Thoresen LH, Reibenspies J, Burgess K (1999) 3,5-Diaryl-4,4-difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY) dyes: synthesis, spectroscopic, electrochemical, and structural properties. J Organomet Chem 64:7813–7819
Algi MP, Tirkes S, Ertan S, Ergun EGC, Cihaner A, Algi F (2013) Design and synthesis of new 4,4′-difluoro-4-bora-3a,4a-diaza-s-indacene based electrochromic polymers. Electrochim Acta 109:766–774
Dick JE, Poirel A, Ziessel R, Bard AJ (2015) Electrochemistry, electrogenerated chemiluminescence, and electropolymerization of oligothienyl-bodıpy derivatives. Electrochim Acta 178:234–239
Lakshmi V, Lee WZ, Ravikanth M (2014) Synthesis, structure and spectral and electrochemical properties of 3-pyrrolyl BODIPY-metal dipyrrin complexes. Dalton Trans 43:16006–16014
Xu L, Li Y, Jiang R, Qin Z, Li Y (2014) 2,6-Anthracenyl(anthraquinonyl)-substituted difluoroboron dipyrromethenes: synthesis, spectroscopy, electrochemistry and quantum chemical calculations. Dyes Pigments 107:90–96
Qi H, Teesdale JJ, Pupillo RC, Rosenthal J, Bard AJ (2013) Synthesis, electrochemistry, and electrogenerated chemiluminescence of two BODIPY-appended bipyridine homologues. J Am Chem Soc 135:13558–13566
Ab N, Pistner AJ, Bard AJ, Rosenthal J (2013) Synthesis, photophysics, electrochemistry and electrogenerated chemiluminescence of PEG-modified BODIPY dyes in organic and aqueous solutions. J Phys Chem C 117:5599–5609
Niu S, Ulrich G, Retailleau P, Ziessel R (2011) BODIPY-bridged push-pull chromophores: Optical and electrochemical properties. Tetrahedron Lett 52:4848–4853
Bura T, Ziessel R (2010) Design, synthesis and redox properties of a fluorene platform linking two different Bodipy dyes. Tetrahedron Lett 51:2875–2879
Acknowledgments
This study was supported by The Research Fund of Karadeniz Technical University, Trabzon, Turkey.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 38 kb)
Rights and permissions
About this article
Cite this article
Biyiklioglu, Z., Keleş, T. Design, Synthesis, Characterization and Electrochemical Properties of BODIPY Dyes Containing Mono, Bis-2-Naphthyloxyhexyloxy and 4-(Benzyloxy)Phenoxyhexyloxy Groups. J Fluoresc 26, 2257–2266 (2016). https://doi.org/10.1007/s10895-016-1921-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1921-1