Abstract
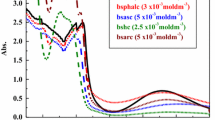
Novel benzothiazole Schiff bases L1 [1-((4,6-difluorobenzo[d]thiazol-2-ylimino)methyl) naphthalen-2-ol], L2 [3-((4,6-difluorobenzo[d]thiazol-2-ylimino) methyl)benzene-1,2-diol], L3 [2-((4,6-difluorobenzo[d]thiazol-2-ylimino)methyl)-5-methoxyphenol], L4 [2-((4,6-difluorobenzo[d]thiazol-2-ylimino)methyl)-4-chlorophenol] and their binary Cu(II) complexes were synthesized. The structures of all the compounds have been discussed on the basis of elemental analysis, FT-IR, NMR, UV-Visible, ESI-Mass, TGA, ESR, SEM, powder XRD and magnetic moments. Based on the analytical and spectral data a square planar geometry has been assigned to all complexes in which the Schiff bases act as monobasic bidentate ligands, coordinating through the azomethine nitrogen and phenolic oxygen atom. DNA binding ability of these complexes was studied on CT-DNA by using UV-Vis absorption, fluorescence and viscometry. DNA cleavage ability of the complexes was examined on pBR322 DNA by using gel electrophoresis method. All the DNA binding studies reveal that they are good intercalators. The bioefficacy of the ligands and their complexes was examined against the growth of bacteria and fungi in vitro to evaluate their antimicrobial potential. The screening data revealed that the complexes showed more antimicrobial activity than the corresponding free ligands.










Similar content being viewed by others
References
Song YM, Wu Q, Yang PJ, Luan NN, Wang LF, Liu YM (2006) DNA binding and cleavage activity of Ni(II) complex with all-trans retinoic acid. J Inorg Biochem 100:1685–1691
Tan C, Liu J, Chen L, Shi S, Ji L (2008) Synthesis, structural characteristics DNA binding properties and cytotoxicity studies of a series of Ru(III) complexes. J Inorg Biochem 102:1644–1653
Zuber G, Quada JC Jr, Hecht SM (1998) Sequence selective cleavage of a DNA Octanucleotide by chlorinated Bithiazoles and Bleomycins. J Am Chem Soc 120:9368–9369
Hall IH, Peaty NJ, Henry JR, Easmon J, Heinisch G, Purstinger G (1999) Investigations on the Mechanism of Action of the Novel Antitumor Agents 2-Benzothiazolyl, 2-Benzoxazolyl, and 2-Benzimidazolyl Hydrazones Derived from 2-Acetylpyridine. Arch Pharm 332:115–123
Hutchinson I, Bradshaw TD, Matthews CS, Stevens MFG, Westwell AD (2003) Antitumour benzothiazoles. Part 20:3′-Cyano and 3′-Alkynyl-substituted 2-(4′-Aminophenyl)benzothiazoles as new potent and selective analogues. Bioorg Med Chem Lett 13:471–474
D.M AE-A, Etaiw SEH, Ali EA (2013) Synthesis, spectroscopic, cytotoxic aspects and computational study of N-(pyridine-2-ylmethylene)benzo[d]thiazol-2-amine Schiff base and some of its transition metal complexes. J Mol Struct 1048:487–499
Benazzouz A, Boraud T, Dubedat P, Boireau A, Stutzmann JM, Gross C (1995) Riluzole prevents MPTP-induced parkinsonism in the rhesus monkey: a pilot study. Eur J Pharmacol 284:299–307
Arjunan V, Sakiladevi S, Rani T, Mythili CV, Mohan S (2012) FTIR, FT-Raman, FT-NMR, UV–visible and quantum chemical investigations of 2-amino-4 methylbenzothiazole. Spectrochim Acta A 88:220–231
Ma Y, Cao L, Kawabata T, Yoshino T, Yang B.B, Okada S (1998) Cupric nitrilotriacetate induces oxidative DNA damage and apoptosis in human leukemia HL-60 cells. Free Radic Biol Med 25:568–575.
Nalawade AM, Nalawade RA, Patange SM, Tase DR (2013) Thiazole Containing Schiffs Bases and Their Transition Metal Complexes. Int Eng Sci Inven 2:1–4
Li H, Li J, Chen H, Zhang Y, Huang D (2011) Synthesis and crystal structure of charge transfer complex (CTC) of 2-aminobenzothiazole with its Schiff Base. J Chem Crystallogr 41:1844–1849
Raman N, Selvan A, Sudharsan S (2011) Metallation of ethylenediamine based Schiff base with biologically active Cu(II), Ni(II) and Zn(II) ions: synthesis, spectroscopic characterization, electrochemical behaviour, DNA binding, photonuclease activity and in vitro antimicrobial efficacy. Spectrochim Acta A 79:873–883
Arjmand F, Mohani B, Parveen S (2006) New Dihydro OO'Bis(Salicylidene) 2,2' Aminobenzothiazolyl borate complexes: kinetic and Voltammetric studies of Dimethyltin copper complex with guanine, adenine, and calf thymus DNA. Bioinorg Chem Appl 1 http://dx.doi.org/10.1155/BCA/2006/32896.
Rajarajeswari C, Ganeshpandian M, Palaniandavar M, Riyasdeen A, Akbarsha MA (2014) Mixed ligand copper(II) complexes of 1,10-phenanthroline with tridentate phenolate/pyridyl/(benz)imidazolyl Schiff base ligands: covalent vs non-covalent DNA binding, DNA cleavage and cytotoxicity. J Inorg Biochem 140:255–268
Wu H, Yuan J, Huang X, Kou F, Liu B, Jia F, Wang K, Bai Y (2012) Two zinc(II) and cadmium(II) complexes based on the V-shaped ligand 2,6-bis(2 benzimidazolyl)pyridine: synthesis, crystal structure, DNA-binding properties and antioxidant activities. Inorg Chim Acta 390:12–21
Easmon J, Pürstinger G, Heinisch G, Roth T, Fiebig HH, Holzer W, Jäger W, Jenny M, Hofmann J (2001) Synthesis, cytotoxicity, and antitumor activity of copper(II) and iron(II) complexes of 4 N-Azabicyclo[3.2.2]nonane thiosemicarbazones derived from acyl Diazines. J Med Chem 44:2164–2171
Tahghighi A (2014) Importance of metal complexes for development of potential leishmanicidal agents. J Organomet Chem 770:51–60
Chityala V K, kumar K S, Ramesh M, Parthasarathy T, Shivaraj (2014) DNA Cleavage, Cytotoxic Activities, and Antimicrobial Studies of Ternary Copper(II) Complexes of Isoxazole Schiff Base and Heterocyclic Compounds. Hindawi Pub bioinochem and app doi:http://dx.doi.org/10.1155/2014/691260.
Kumar MP, Tejaswi S, Rambabu A, Kalalbandi VKA, Shivaraj (2015) Synthesis, crystal structure, DNA binding and cleavage studies of copper(II) complexes with isoxazole Schiff bases. Polyhedron 102:111–120
Marmur J (1961) A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J Mol Biol 3:208–218. doi:10.1016/s0022-2836(61)80047-8
Kumar CV, Asuncion EH (1993) DNA binding studies and site selective fluorescence sensitization of an anthryl probe. J Am Chem Soc 115:8547–8553. doi:10.1021/ja00072a004
Tandon VK, Yadav DB, Singh RV, Vaish M, Chaturvedi AK, Shukla PK (2005) Synthesis and biological evaluation of novel 1,4-naphthoquinone derivatives as antibacterial and antiviral agents. Bioorg Med Chem Lett 15:3463–3466. doi:10.1016/j.bmcl.2005.04.075
Bhunora S, Mugo J, Bhaw-Luximon A, Mapolie S, Van Wyk J, Darkwa J, Nordlander E (2011) The use of Cu and Zn salicylaldimine complexes as catalyst precursors in ring opening polymerization of lactides: ligand effects on polymer characteristics. Appl Organomet Chem 25:133–145
Youssef NS, El-Zahany E, El-Seidy AMA, Caselli A, Fantauzzi S, Cenini S (2009) Synthesis and characterisation of new Schiff base metal complexes and their use as catalysts for olefin cyclopropanation. Inorg Chim Acta 362:2006–2014
Percy GC, Thornton DA (1973) Infrared spectra of N-aryl salicylaldimine complexes substituted in both aryl rings. J Inorg Nucl Chem 35:2319–2327
Nakamoto K (1997) Infrared and Raman spectra of inorganic and coordination compounds, fifth edn. Wiley-Interscience, New York
Bellamy LJ (1980) The Infrared Spectra of Complex Molecules, second edn. Chapmann and Hall, London
Cotton FA, Wilkinson G (1972) Advanced Inorganic Chemistry, third edn. Interscience Publisher, New York
Lever ABP (1984) Inorganic Electronic Spectroscopy, second edn. Elsevier, Amsterdam
Zhou Y, Ye X, Xin F, Xin X (1999) Solid state self-assembly synthesis of cobalt(II), nickel(II), copper(II) and zinc(II) complexes with a bis-Schiff base. Transit Met Chem 24:118–120
Ramesh R, Maheswaran S (2003) Synthesis, spectra, dioxygen affinity and antifungal activity of Ru(III) Schiff base complexes. J Inorg Biochem 96:457–462
Unver H, Hayvali Z (2010) Synthesis, spectroscopic studies and structures of square-planar nickel(II) and copper(II) complexes derived from 2-{(Z)-[furan-2 ylmethyl]imino]methyl}-6-methoxyphenol. Spectrochim Acta A 75:782–788
Patel RN, Singh N, Shukla KK, Chauhan UK, Nicols -Gutierrez J, Castineiras A (2004) Magnetic, spectroscopic, structural and biological properties of mixed-ligand complexes of copper(II) with N,N,N ′,N″,N″-pentamethyldiethylenetriamine and polypyridine ligands. Inorg Chim Acta 357:2469–2476
Kivelson D, Neiman R (1961) ESR studies on the bonding in copper complexes. J Chem Phys 35:149–155
Hathaway BJ, Billing DE (1970) The electronic properties and stereochemistry of mono-nuclear complexes of the copper(II) ion. Coord Chem Rev 5:143–207
Barton JK, Danishefsky AT, Goldberg JM (1984) Tris(phenanthroline)ruthenium(II): stereoselectivity in binding to DNA. J Am Chem Soc 106:2172–2176
Tysoe SA, Morgan RJ, Baker AD, Strekas TC (1993) Spectroscopic investigation of differential binding modes of DELTA and LAMBDA.-Ru(bpy)2(ppz)2 with calf thymus DNA. J Phys Chem 97:1707–1711
Liu HK, Sadler PJ (2011) Metal complexes as DNA intercalators. Acc Chem Res 44:349–359
Pyle AM, Rehmann JP, Meshoyrer R, Kumar CV, Turro NJ, Barton JK (1989) Mixed-ligand complexes of ruthenium(II): factors governing binding to DNA. J Am Chem Soc 111:3051–3058
Li TR, Yang ZY, Wang BD, Qin DD (2008) Synthesis, characterization, antioxidant activity and DNA-binding studies of two rare earth(III) complexes with naringenin-2-hydroxy benzoyl hydrazone ligand. Eur J Med Chem 43:1688–1695
Sampath K, Sathiyaraj S, Jayabalakrishnan C (2013) DNA binding, DNA cleavage, antioxidant and cytotoxicity studies on ruthenium(II) complexes of benzaldehyde 4-methyl-3-thiosemicarbazones. Spectrochim Acta A 105:582–592
Waring MJ (1965) Complex formation between ethidium bromide and nucleic acids. J Mol Biol 13:269–282
Meyer-Almes FJ, Porschke D (1993) Mechanism of intercalation into the DNA double helix by ethidium. Biochemistry 32:4246–4253
Lepecq JB, Paoletti C (1967) A fluorescent complex between ethidium bromide and nucleic acids: physical-chemical characterization. J Mol Biol 27:87–106
Chen J, Wang X, Chao Y, Zhu JH, Zhu YG, Li YZ, Xu Q, Guo ZJ (2007) A Trinuclear copper(II) complex of 2,4,6-Tris(di-2-pyridylamine)-1,3,5-triazine shows prominent DNA cleavage activity. Inorg Chem 46:3306–3312. doi:10.1021/ic0614162
Tarui M, Doi M, Ishida T, Inoue M, Nakaike S, Kitamura K (1994) DNA-binding characterization of a novel anti-tumour benzo[a]phenazine derivative NC-182: spectroscopic and viscometric studies. Biochem J 304:271–279
Guo H, Lu J, Ruan Z, Zhang Y, Liu Y, Zang L, Jiang J, Huang J (2012) Synthesis, DNA-binding, cytotoxicity, and cleavage studies of unsymmetrical oxovanadium complexes. J Coord Chem 65:191–204
Satyanarayana S, Dabrowiak JC, Chaires JB (1993) Tris(phenanthroline)ruthenium(II) enantiomer interactions with DNA: mode and specificity of binding. Biochemistry 32:2573–2584
Zhen X S, Ye H B, Zhang L Q, Liu G J, Li H, Ji N L (1998) Synthesis characterization and effect of ligand planarity of [Ru(bipy)2 L)2+ on DNA binding affinity.
Sigman DS (1986) Nuclease activity of 1,10-phenanthroline-copper ion, nuclease activity of 1,10-phenanthroline-copper ion. Acc Chem Res 19:180–186
Belai S, Landreau A, Djebbar S, Benali-Baitich O, Bouet G, Bouchara JP (2008) Synthesis, characterization and antifungal activity of a series of manganese(II) and copper(II) complexes with ligands derived from reduced N,N′-O-phenylenebis(salicylideneimine). J Inorg Biochem 102:63–69
Tweedy BG (1964) Possible mechanism for reduction of elemental sulfur by Monilinia fructicola. Phytopathology 55:910–914
Sharma AK, Chandra S (2011) Complexation of nitrogen and Sulphur donor Schiff's base ligand to Cr(III) and Ni(II) metal ions: synthesis, spectroscopic and antipathogenic studies. Spectrochim Acta A 78:337–342
Chohan ZH (2004) Synthesis and biological properties of Cu(II) complexes with 1,1′-Disubstituted Ferrocenes. Synth React Inorg met Org Chem 34:833–846
Acknowledgments
We express our sincere thanks to the Head, Department of Chemistry for providing the necessary facilities. We are thankful to the Director, CFRD, Osmania University, Hyderabad, and the Director, IICT, Hyderabad, and the SAIF, IIT Bombay for providing spectral and analytical data. We are also thankful to DST-PURSE, DST-SERB and UGC-UPE (FAR) for providing financial assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tejaswi, S., Kumar, M.P., Rambabu, A. et al. Synthesis, Structural, DNA Binding and Cleavage Studies of Cu(II) Complexes Containing Benzothiazole Cored Schiff Bases. J Fluoresc 26, 2151–2163 (2016). https://doi.org/10.1007/s10895-016-1911-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1911-3