Abstract
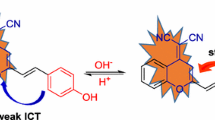
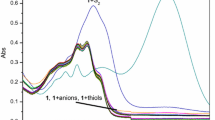
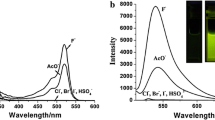
A new chemodosimeter based on dicyanomethylene-4H-chromene chromophore (probe 1) was developed as a ratiometric fluorescent probe in near-infrared range for F− with good selectivity in acetonitrile. Probe 1 could be used to directly visualize F− by the naked eye and showed more than 621-fold fluorescence enhancement at 715 nm upon reaction with F− upon excitation at 625 nm. The recognition of probe 1 to fluoride was featured by F−-induced red-shifts of both absorption (185 nm) and fluorescence peaks (132 nm) based on internal charge transfer (ICT) in acetonitrile. The desilylation reaction of 1 by F− was proposed for its dual absorption and emission ratiometric detection of fluoride.








Similar content being viewed by others
References
Gale PA (2001) Anion receptor chemistry: highlights from 1999. Coord Chem Rev 213:79–128
Sessler JL, Davis JM (2001) Sapphyrins: versatile anion-binding agents. Acc Chem Res 34:989–997
Beer PD, Gale PA (2001) Anion recognition and sensing: the state of the art and future perspectives. Angew Chem Int Ed 40:486–516
Santos-Figueroa LE, Moragues ME, Estela C, Alessandro A, Martinez-Manaez R, Sancenón F (2013) Chromogenic and fluorogenic chemosensors and reagents for anions. A comprehensive review of the years 2010-2011. Chem Soc Rev 42(8):3489–3613
Gunnlaugsson T, Glynn M, Tocci GM, Kruger PE, Pfeffer FM (2006) Anion recognition and sensing in organic and aqueous media using luminescent and colorimetric sensors. Coord Chem Rev 250:3094–3117
Kirk, KL. Biochemistry of the Halogens and Inorganic Halides; Plenum Press: New York, 1991; p 58
Kleerekoper M (1998) The role of fluoride in the prevention of osteoporosis. Endocrinol Metab Clin N Am 27:441–452
Weatherall JA (1969) Pharmacology of fluorides, in handbook of experimental pharmacology XX/1. Springer-Verlag, Berlin, Part 1:141–172
Gale PA (2006) Structural and molecular recognition studies with acyclic anion receptors. Acc Chem Res 39:465–475
Huang X, Guo Z, Zhu W, Xie Y, Tian H (2008) A colorimetric and fluorescent turn-on sensor for pyrophosphate anion based on a dicyanomethylene-4H-chromene framework. Chem Commun 44:5143–5145
Kim SK, Lee DH, Hong JI, Yoon J (2009) Chemosensors for pyrophosphate. Acc Chem Res 42:23–31
Lee CH, Miyaji H, Yoon DW, Sessler JL (2008) Strapped and other topographically nonplanar calixpyrrole analogues. Improved anion receptors. Chem Commun 44(1):24–34
Martínez-Máñez R, Sancenón F (2003) Fluorogenic and chromogenic chemosensors and reagents for anions. Chem Rev 103(11):4419–4476
Xu Z, Singh NJ, Lim J, Pan J, Kim HN, Park S, Kim KS, Yoon J (2009) Unique sandwich stacking of pyrene-adenine-pyrene for selective and ratiometric fluorescent sensing of ATP at physiological pH. J Am Chem Soc 131(42):15528–15533
Zhao JZ, Fyles TM, James TD (2004) Chiral binol-bisboronic acid as fluorescence sensor for sugar acids. Angew Chem Int Ed 116(26):3543–3546
Komatsu K, Urano Y, Kojima H, Nagano T (2007) Development of an iminocoumarin-based zinc sensor suitable for ratiometric fluorescence imaging of neuronal zinc. J Am Chem Soc 129(44):13447–13454
Lin WY, Long LL, Chen BB, Tan W (2009) A ratiometric fluorescent probe for hypochlorite based on a deoximation reaction. Chem Eur J 15(10):2305–2309
Qian F, Zhang CL, Zhang YM, He WJ, Gao X, Hu P, Guo ZJ (2009) Visible light excitable Zn2+ fluorescent sensor derived from an intramolecular charge transfer fluorophore and its in vitro and in vivo application. J Am Chem Soc 131(1):1460–1468
Srikun D, Miller EW, Dornaille DW, Chang CJ (2008) An ICT based approach to ratiometric fluorescence imaging of hydrogen peroxide produced in living cells. J Am Chem Soc 130(14):4596–4597
Coskun A, Akkaya EU (2006) Signal ratio amplification via modulation of resonance energy transfer: proof of principle in an emission ratiometric Hg(II) sensor. J Am Chem Soc 128:14474–14475
Lin W, Yuan L, Long L, Guo C, Feng J (2008) A fluorescent cobalt probe with a large ratiometric fluorescence response via modulation of energy acceptor molar absorptivity on metal ion binding. Adv Funct Mater 18:2366–2372
Lee MH, Kim JS, Sessler JL (2015) Small molecule-based ratiometric fluorescence probes for cations, anions, and biomolecules. Chem Soc Rev 44(13):4185–4191
Guo Z, Zhu W, Tian H (2012) Dicyanomethylene-4H-pyran chromophores for OLED emitters, logic gates and optical chemosensors. Chem Commun 48:6073–6084
Zhu W, Huang X, Guo Z, Wu X, Yu H, Tian H (2012) A novel NIR fluorescent turn-on sensor for the detection of pyrophosphate anion in complete water system. Chem Commun 48(12):1784–1786
Cao J, Zhao CC, Zhu WH (2012) A near-infrared fluorescence chemodosimeter for fluoride via specific Si-O cleavage. Tetrahedron Lett 53:2107–2110
Sun W, Fan J, Hu C, Cao J, Zhang H, Xiong X, Wang J, Cui S, Sun S, Peng X (2013) A two-photon fluorescent probe with near-infrared emission for hydrogen sulfide imaging in biosystems Chem. Commun 49:3890–3892
Li M, Wu X, Wang Y, Li Y, Zhu W, James TD (2014) A near-infrared colorimetric fluorescent chemodosimeter for the detection of glutathione in living cells. Chem Commun 50:1751–1753
Bao Y, Liu B, Wang H, Tian J, Bai R (2011) A “naked eye” and ratiometric fluorescent chemosensor for rapid detection of F− based on desilylation reaction and excited-state proton transfer. Chem Commun 47:3957–3959
Filik H, Hayvali M, Kilic E (2005) Sequential spectrophotometric determination of paracetamol and p-aminophenol with 2,2′-(1,4-phenylenedivinylene) bis-8-hydroxyquinoline as a novel coupling reagent after microwave assisted hydrolysis. Anal Chim Acta 535:177–182
Li ZF, Xiang Y, Tong AJ (2008) Ratiometric chemosensor for fluorescent determination of Zn2+ in aqueous ethanol. Anal Chim Acta 619:75–80
Acknowledgments
The authors thank the NSFC (project No. 21102037 and No. 21201057) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 1648 kb)
Rights and permissions
About this article
Cite this article
Xie, P., Guo, F., Gao, G. et al. A near-Infrared Fluorescent Chemodosimeter for Ratiometric Detecting Fluoride Based on Desilylation Reaction. J Fluoresc 26, 1737–1742 (2016). https://doi.org/10.1007/s10895-016-1865-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1865-5