Abstract
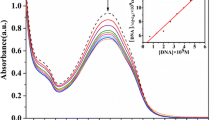
The focus of the present work is the preparation of new metal-based nanodrug to overcome limitations of chemotherapy such as poor water solubility of most common chemotherapeutic drugs. The copper(II) complex of 1,2,4-triazine derivatives, [Cu(dppt)2(H2O)2]2+ (dppt is 5,6-diphenyl- 3- (2-pyridyl)-1,2,4-triazine), has been synthesized at nano-size by sonochemical method and characterized by FTIR, zetasizer, and scanning electron microscopy (SEM). The interaction of the complex and nanocomplex with fish sperm DNA (FS–DNA) and BSA have been investigated under physiological conditions by a series of experimental methods. The results have indicated that the complex binds to FS–DNA by two biding modes, viz., electrostatic and intercalates into the base pairs of DNA. The competitive study with ethidium bromide (EB) shows that the complex and nanocomplex competes for the DNA–binding sites with EB. Protein binding studies show that the complex and nanocomplex could bind with BSA. The results of synchronous fluorescence of BSA show that additions of the complex affect the microenvironment of both tyrosine and tryptophan residues during the binding process. The in vitro cytotoxicity of the complex (solution in DMSO) and nanocomplex (colloid in H2O) against the human carcinoma cell lines (MCF-7 and A-549) was evaluated by MTT assay. The results of in vitro cytotoxicity indicate that the complex and nanocomplex have excellent cytotoxicity activity against MCF-7 and A-549. Results of the microscopic analyses of the cancer cells confirm the results of the cytotoxicity.

















Similar content being viewed by others
References
DeVita VT, Chu E (2008) A history of cancer chemotherapy. Cancer Res 68:8643–8653
Chabner BA, Roberts TG (2005) Chemotherapy and the war on cancer. Nat Rev Cancer 5:65–72
Rosenberg B, VanCamp L, Trosko JE, Mansour VH (1969) Platinum compounds: a new class of potent antitumor agents. Nature 222:385–386
Van Rijt SH, Sadler PJ (2009) Current applications and future potential for bioinorganic chemistry in the development of anticancer drugs. Drug Discov Today 14:1089–1097
Bruijnincx PC, Sadler PJ (2008) New trends for metal complexes with anticancer activity. Curr Opin Chem Biol 12:197–206
Peer D, Karp JM, Hong S, FaroKHzad OC, Margalit R, Langer R (2007) Nanocarriers as an emerging platform for cancer therapy. Nat Nanotechnol 2:751–760
Rosenblum D, Peer D (2014) Omics-based nanomedicine: the future of personalized oncology. Cancer Lett 352:126–136
Gregoriadis G, Apostolidis N, Romanos A, Paradellis T (1983) A comparative study of trace elements in normal and cancerous colorectal tissues. Cancer 52:508–519
Barve A, Kumbhar A, Bhat M, Joshi B, Butcher R, Sonawane U, Joshi R (2009) Mixed-ligand copper(II) maltolate complexes: synthesis, characterization, DNA binding and cleavage, and cytotoxicity. Inorg Chem 48:9120–9132
Bales BC, Kodama T, Weledji YN, Pitie M, Meunier B, Greenberg MM (2005) Mechanistic studies on DNA damage by minor groove binding copper–phenanthroline conjugates. Nucleic Acids Res 33:5371–5379
Mariano C, Pellei M, Alidori S (2006) New copper (I) phosphane complexes of dihydridobis(3-nitro-1,2,4-triazolyl)borate ligand showing cytotoxic activity. J Inorg Biochem 100:299–304
Sohail S, Aghmana R, Muhammad A, Rizwan H (2010) Synthesis, characterization and antibacterial activity of nickel (II) and copper (II) complexes of N-(alkyl (aryl)carbamothioyl)-4-nitrobenzamide. Eur J Chem 3:200–205
Ranford JD, Sadler PJ, Tocher DA (1993) Cytotoxicity and antiviral activity of transition-metal salycylato complexes and crystal-structure of bis(diisopropyl-salicylato)(1,10-phenanthroline)copper(II). J Chem Soc 22:3393–3399
Hac EE, Gagalo IT, Hac E (1983) Copper salicylate complex: thermoregulatory and biochemical effects. Drug Exp Clin Res 8:469–478
Fujimori T, Yamada S, Yasui H, Sakurai H, In Y, Ishida T (2005) Orally active antioxidative copper(II) aspirinate: synthesis, structure characterization, super-oxide scavenging activity, andin vitro andin vivo antioxidative evaluations. J Biol Inorg Chem 8:831–841
Anjomshoa M, Fatemi SJ, Torkzadeh-Mahani M, Hadadzadeh H (2014) DNA- and BSA- binding studies and anticancer activity against human breast cancer cells (MCF-7) of the zinc(II) complex coordinated by 5,6-diphenyl-3-(2-pyridyl)-1,2,4-triazine. Spectrochim Acta Part A 127:511–520
Anjomshoa M, Hadadzadeh H, Torkzadeh-Mahani M, Fatemi SJ, Adeli-Sardou M, Amiri Rudbari H, Nardo VM (2015) A mononuclear Cu(II) complex with 5,6-diphenyl-3-(2-pyridyl)-1,2,4-triazine: synthesis, crystal structure, DNA- and BSA-binding, molecular modeling, and anticancer activity against MCF-7, A-549, and HT-29 cell lines. Eur J Med Chem 96:66–82
Anjomshoa M, Hadadzadeh H, Fatemi SJ, Torkzadeh-Mahani M (2015) A mononuclear Ni(II) complex with 5,6-diphenyl-3-(2-pyridyl)-1,2,4-triazine: DNA- and BSA-binding and anticancer activity against human breast carcinoma cells. Spectrochim Acta Part A 136:205–215
Srishailam A, Kumar YP, Gabra NMD, Reddy PV, Deepika N, Veerababu N, Satyanarayana S (2013) Synthesis, DNA-binding, cytotoxicity, photocleavage, antimicrobial and docking studies of Ru(II) polypyridyl complexes. J Fluoresc 23:897–908
Uma R, Palaniandavar M, Butcher RJ (1996) Synthesis, structure, spectra and redox interconversions in copper (II) complexes of 5,6-diphenyl-3-(2-pyridyl)-1,2,4triazine. J Chem Soc Dalton Trans 2061–2066. doi:10.1039/DT9960002061
Anjomshoa M, Torkzadeh-Mahani M (2015) In vitro DNA and BSA–binding, cell imaging and anticancer activity against human carcinoma cell lines of mixed ligand copper (II) complexes. Spectrochim Acta Part A 150:390–402
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J lmmunol Methods 65:55–63
Suh D, Chaires JB (1995) Criteria for the mode of binding of DNA binding agents. Bioorg Med Chem 3:723–728
Yin H, Xu Y, Qian X (2007) Novel antitumor agent family of 1 H-benzo[c,d]indol-2-one with flexible basic side chains: synthesis and biological evaluation. Bioorg Med Chem 15:1356–1362
Guo Q, Li L, Dong J, Liu H, Xu T, Li J (2013) Synthesis, crystal structure and interaction ofL-valine schiff base divanadium(V) complex containing a V2O3 core with DNA and BSA. Spectrochim Acta Part A 106:155–162
Carter MT, Rodriguez M, Bard AJ (1989) Voltammetric studies of the interaction of metal chelates with DNA. 2. Tris-chelated complexes of cobalt(III) and iron(II) with 1,10-phenanthroline and 2,2’-bipyridine. J Am Chem Soc 111:8901–8911
Wang Y, Zhang H, Zhang G, Tao W, Tang S (2007) Interaction of the flavonoid hesperidin with bovine serum albumin: a fluorescence quenching study. J Lumin 126:211–218
Lakowicz JR (2006) Principles of fluorescence spectroscopy, third edn. Kluwer Academic Plenum Publishers, New York
Ray A, Seth BK, Pal U, Basu S (2012) Nickel(II)-schiff base complex recognizing domain II of bovine and human serum albumin: spectroscopic and docking studies. Spectrochim Acta Part A 92:164–174
Toneatto J, Arguello GA (2011) New advances in the study on the interaction of [Cr(phen)2(dppz)]3+complex with biological models; association to transporting proteins. J Inorg Biochem 105:645–651
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions: forces contributing to stability. Biochemistry 20:3096–3102
Da Silva JG, Recio Despaigne AA, Louro SRW, Bandeira CC, Souza-Fagundes EM, Beraldo H (2013) Cytotoxic activity, albumin and DNA binding of new copper(II) complexes with chalcone-derived thiosemicarbazones. Eur J Med Chem 65:415–426
Acknowledgments
We thank the head and director of the Institute of Science, High Technology & Environmental Science, Graduate University of Advanced Technology of Kerman and Keramn Cancer Research Center for their support of this work.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Anjomshoa, M., Torkzadeh-Mahani, M., Dashtrazmi, E. et al. Sonochemical Synthesis and Characterization of the Copper(II) Nanocomplex: DNA- and BSA-Binding, Cell Imaging, and Cytotoxicity Against the Human Carcinoma Cell Lines. J Fluoresc 26, 545–558 (2016). https://doi.org/10.1007/s10895-015-1739-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-015-1739-2