Abstract
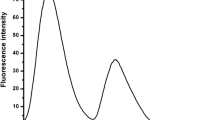
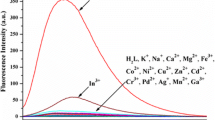
The condensation product of L-alanine and salicylaldehyde was synthesised and characterised which was found to be selective fluorescent “on” sensor for Al3+ ion with the detection limit 10−6 M. The sensor is free of interferences from metal ions - Na+, K+, Ca2+, Mn2+, Co2+, Ni2+, Cu2+, Pb2+, Cd2+, Hg2+ and Fe3+. The Fluorescence and the UV/visible spectral data reveals a 1:1 interaction between the sensor and Al3+ ion with binding constant 104.5. The DFT and TDDFT calculations confirm the structures of the sensor and the sensor-Al3+ complex.









Similar content being viewed by others
References
Goswami S, Paul S, Manna A (2013) Selective “naked eye” detection of Al(III) and PPi in aqueous media on a rhodamine–isatin hybrid moiety. RSC Adv 3:10639–10643
Verstraeten SV, Aimo L, Oteiza PI (2008) Aluminium and lead: molecular mechanisms of brain toxicity. Arch Toxicol 82:789–802
Wang B, Xing W, Zhao Y, Deng X (2010) Effects of chronic aluminum exposure on memory through multiple signal transduction pathways. Environ Toxicol Pharmacol 29:308–313
Delhaize E, Ryan PR (1995) Aluminum toxicity and tolerance in plants. Plant Physiol 107:315–321
Han T, Feng X, Tong B, Barcelo J, Poschenrieder C (2002) Environ Exp Bot 48:75–92
Shi L, Chen L, Zhi J, Dong T (2012) A novel “turn-on” fluorescent chemosensor for the selective detection ofAl3+ based on aggregation-induced emission. Chem Commun 48:416–418
Sang H, Liang P, Du D (2008) Determination of trace aluminum in biological and water samples by cloud point extraction preconcentration and graphite furnace atomic absorption spectrometry detection. J Hazard Mater 154:1127–11326
Djane SJ, Gra M, Korn C (2000) A separation method to overcome the interference of aluminium on zinc determination by inductively coupled plasma atomic emission spectroscopy. Spectrochim Acta B 55:389–394
Abbasi S, Farmany A (2009) Food Chem 116:1019–1023
Gupta VK, Jain AK, Maheshwari G (2007) Aluminum(III) selective potentiometric sensor based on morin in poly(vinyl chloride) matrix. Talanta 72:1469–1473
Fan L, X-hui J, Wang B-d, Yang Z-y (2014) 4-(8 -hydroxyquinolin-7 -yl)methyleneimino-1-phenyl-2,3- dimethyl-5-pyzole as a fluorescent chemosensor for aluminum ion in acid aqueous medium. Sensors Actuators B 205:249–254
J-can Q, T-rong L, Wang B-d, Yang Z-y, Fan L (2014) Fluorescent sensor for selective detection of Al3+ based on quinoline–coumarin conjugate. Spectrochim Acta A Mole Biomole Spectro 133:38–43
Malkondu S (2014) A highly selective and sensitive perylenebisimide-based fluorescent PET sensor for Al3+ determination in MeCN. Tetrahedron 70:5580–5584
Kima DH, Im YS, Kimb H, Kima C (2014) Solvent-dependent selective fluorescence sensing of Al3+ and Zn2+ using a single Schiff base. Inorg Chem Commun 45:15–19
Li T, Fang R, Wang B, Shao Y, Liu J, Zhang S, Yang Z (2014) A simple coumarin as a turn-on fluorescence sensor for Al(III) ions. Dalton Trans 43:2741–2743
Patil R, Moirangthem A, Butcher R, Singh N, Basu A, Tayade K, Fegade U, Hundiwale D, Kuwar A (2014) Al3+ selective colorimetric and fluorescent red shifting chemosensor: application in living cell imaging. Dalton Trans 43:2895–2899
Dutta K, Deka R C, Das D K (2014) A new fluorescent and electrochemical Zn2+ ion sensor based on Schiff base derived from benzil and L-tryptophan. Spectrochim Acta A Mol Biomol Spectrosc 124:124–129
Dutta K, Deka RC, Das DK (2013) A new on-fluorescent probe for manganese (II) ion. J Fluoresc 23:1173–1178
Kumar J, Bhattacharyya P, Das DK (2015) New duel fluorescent “on–off” and colorimetric sensor for copper(II): copper(II) binds through N coordination and pi cation interaction to sensor. Spectrochim Acta A Mol Biomol Spectrosc 138:99–104
Kumar J, Sarma MJ, Phukan P, Das DK (2015) A new simple Schiff base fluorescence “on” sensor for Al3+ and its living cell imaging. Dalton Trans 44:4576–45881
Parr RG (1995) Density-functional theory of the electronic structure of molecules. Annu Rev Phys Chem 46:701–728
Koch W, Holthausen MC (2001) A chemist’s guide to density functional theory, 2nd edn. Wiley-VCH, New York
Fiolhais C, Nogueira F, Marques M (2003) A primer in density functional theory. Springer-Verlag Berlin, Heidelberg
Mc Lean AD, Chandler GS (1980) Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z = 11–18. J Chem Phys 72:5639–5648
Raghavachari K, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650
Becke AD (1993) Density-functional thermochemistry. III The Role of Exact Exchange J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Development of the colle-salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789
Stratmann RE, Scuseria GE, Frisch MJ (1998) An efficient implementation of time-dependent density-functional theory for the calculation of excitation energies of large molecules. J Chem Phys 109:8218–8224
Bauernschmitt R, Ahlrichs R (1996) Treatment of electronic excitations within the adiabatic approximation of time dependent density functional theory. Chem Phys Lett 256:454
Casida ME, Jamorski C, Casida KC, Salahub DR (1998) Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: characterization and correction of the time-dependent local density approximation ionization threshold. J Chem Phys 108:4439–4449
Gaussian 09, Revision B.01, Frisch et al. Gaussian, Inc., Wallingford CT 2010
Shortreed M, Kopelman R, Kuhn M, Hoyland B (1996) Fluorescent fiber-optic calcium sensor for physiological measurements. Anal Chem 68:1414–1418
Acknowledgments
DST, New Delhi and UGC, New Delhi are thanked for financial support to the department through FIST-II and SAP respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sarma, S., Bhattacharyya, P.K. & Das, D.K. A New Fluorescent “Off-On” Sensor for Al3+ Derived from L-alanine and Salicylaldehyde. J Fluoresc 25, 1537–1542 (2015). https://doi.org/10.1007/s10895-015-1647-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-015-1647-5