Abstract
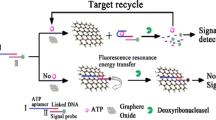
Graphene oxide (GO) was introduced as an efficient quencher for label-free and sensitive detection of DNA. Probe DNA (pDNA) was mixed with ethidium bromide (EB) and graphene oxide (GO). The interaction between EB and GO led to the fluorescent quenching. Upon the recognition of the target, EB was intercalated into duplex DNA and kept away from GO, which significantly hindered the long range resonance energy transfer (LrRET) from EB to GO and, thus, increased the fluorescence of EB. The changes in fluorescent intensity produced a novel method for sensitivity, and specificity detection of the target. Based on the structure-switching of aptamers, this strategy could be conveniently extended for detection of other biomolecules, which had been demonstrated by the detection of exonuclease activity.









Similar content being viewed by others
Abbreviations
- GO:
-
Graphene oxide
- pDNA:
-
Probe DNA
- EB:
-
Ethidium bromide
- LrRET:
-
Long range resonance energy transfer
- Exo III:
-
Exonuclease
- tDNA:
-
Target DNA
References
Dikin DA, Stankovich S, Zimney EJ, Piner RD, Dommett GHB, Evmenenko G, Nguyen ST, Ruoff RS (2007) Preparation and characterization of graphene oxide paper. Nature 448:457–460
Geim AK, Novoselov KS (2007) The rise of graphene. Nat Mater 6:183–191
Yang W, Ratinac KR, Ringer SP, Thordarson P, Gooding JJ, Braet F (2010) Carbon nanomaterials in biosensors: should you use nanotubes or graphene? Angew Chem Int Ed 49:2114–2138
Novoselov KS, Geim AK, Morozov SV, Jiang D, Zhang Y, Dubonos SV, Grigorieva IV, Firsov AA (2004) Electric field effect in atomically thin carbon films. Science 306:666–669
Liu LH, Lerner MM, Yan M (2010) Derivitization of pristine graphene with well-defined chemical functionalities. Nano Lett 10:3754–3756
Li J, Lu CH, Yao QH, Zhang XL, Liu JJ, Yang HH, Chen GN (2011) A graphene oxide platform for energy transfer-based detection of protease activity. Biosens Bioelectron 26:3894–3899
Varghese N, Mogera U, Govindaraj A, Das A, Maiti PK, Sood AK, Rao CNR (2009) Binding of DNA nucleobases and nucleosides with graphene. Chem Phys Chem 10:206–210
Dong H, Gao W, Yan F, Ji H, Ju H (2010) Fluorescence resonance energy transfer between quantum dots and graphene oxide for sensing biomolecules. Anal Chem 82:5511–5517
Jang H, Kim YK, Kwon HM, Yeo WS, Kim DE, Min DH (2010) A graphene-based platform for the assay of Duplex-DNA unwinding by helicase. Angew Chem Int Ed 49:5703–5707
Bi S, Zhao T, Luo B (2012) A graphene oxide platform for the assay of biomolecules based on chemiluminescence resonance energy transfer. Chem Commun 48:106–108
Song YJ, Qu KG, Zhao C, Ren JS, Qu XG (2010) Graphene oxide: intrinsic peroxidase catalytic activity and its application to glucose detection. Adv Mater 22:2206–2210
Zhang C, Yuan Y, Zhang S, Wang Y, Liu Z (2011) Biosensing platform based on fluorescence resonance energy transfer from upconverting nanocrystals to graphene oxide. Angew Chem Int Ed 50:6851–6854
Liu C, Wang Z, Jia H, Li Z (2011) Efficient fluorescence resonance energy transfer between upconversion nanophosphors and graphene oxide: a highly sensitive biosensing platform. Chem Commun 47:4661–4663
Swathi RS, Sebastian KL (2009) Long range resonance energy transfer from a dye molecule to graphene has (distance)−4 dependence. J Chem Phys 130:86–101
He F, Tang Y, Yu M, Feng F, An L, Sun H, Wang S, Li Y, Zhu D, Bazan GC (2006) Quadruplex-to-duplex transition of g-rich oligonucleotides probed by cationic water-soluble conjugated polyelectrolytes. J Am Chem Soc 128:6764–6765
Freeman R, Liu X, Willner I (2011) Amplified multiplexed analysis of DNA by the exonuclease iii-catalyzed regeneration of the target DNA in the presence of functionalized semiconductor quantum dots. Nano Lett 11:4456–4461
Chen H, Wang J, Liang G, Zhang P, Kong JL (2012) A novel exonuclease III aided amplification method for sensitive nucleic acid detection based on single walled carbon nanotube induced quenching. Chem Commun 48:269–271
Yang X, Zhang X, Liu Z, Ma Y, Huang Y, Chen Y (2008) High-efficiency loading and controlled release of doxorubicin hydrochloride on graphene oxide. J Phys Chem C 1121:7554–7558
Chen Q, Zhang W, Wang H, Zhou H (1996) Kinetics of inactivation of green crab (Scylla Serrata) alkaline phosphatase during removal of zinc ions by ethylenediaminetetraacetic acid disodium. Int J Biol Macromol 19:257–261
Whisnant A, Gilman S (2002) Studies of reversible inhibition, irreversible inhibition, and activation of alkaline phosphatase by capillary electrophoresis. Anal Biochem 307:226–234
Acknowledgments
This work has been supported by National Natural Science Foundation of China (No. 21165004, 21163002), the Guangxi Natural Science Foundation of China (2010GXNSFF013001, 2012GXNSFBA053022), Innovation Plan in Graduate Education of Guangxi Province (2010106020703 M70) and the project of Key Laboratory for the Chemistry and Molecular Engineering of Medicinal Resources(Guangxi Normal University), Ministry of Education of China(CMEMR2011-14).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, Y., Tian, J., Chen, S. et al. A Graphene Oxide–Based Sensing Platform for The Label-free Assay of DNA Sequence and Exonuclease Activity via Long Range Resonance Energy Transfer. J Fluoresc 23, 697–703 (2013). https://doi.org/10.1007/s10895-013-1189-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-013-1189-7