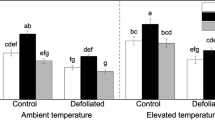
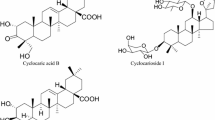
Abstract
Condensed tannins (CTs) are abundant, ecologically-relevant secondary metabolites in many plants, which respond to variables associated with anthropogenic environmental change. While many studies have reported how genetic and environmental factors affect CT concentrations, few have explored how they influence CT molecular structure. Here, using trembling aspen (Populus tremuloides) as a model organism, we report how foliar CT concentrations, polymer sizes, representation of procyanidins and prodelphinidins, and stereochemistry vary in response to changes in air temperature (warming and freeze damage), air composition (elevated CO2 and O3), soil quality (nutrients and microbiome), and herbivory (mammal and lepidopteran). Use of multiple aspen genotypes enabled assessment of genetic influences on aspen CTs. CT concentration and composition were analyzed by thiolysis-ultra high performance liquid chromatography/mass spectrometry in archived leaf samples from prior experiments. All environmental variables explored except for soil microbiome influenced both CT quantity and quality, with climate factors appearing to have larger effect magnitudes than herbivory. Climate, soil, and herbivory effects varied among genotypes, while air composition effects were consistent across genotypes. Considering that CT properties (concentrations and molecular structures) mediate functions at the organismal through ecosystem scales, intraspecific variation in responses of CT properties to environmental factors could provide a pathway through which environmental change exerts selective pressure on Populus populations. Future studies are needed to identify the molecular-level mechanisms by which environmental factors influence CT concentrations and structures, and to establish their ecological and evolutionary significance.






Similar content being viewed by others
Data Availability
References
Ayres MP, Clausen TP, Jr MacLean SF, Redman AM, Reichardt PB (1997) Diversity of structure and antiherbivore activity in condensed tannins. Ecology 78:1696–1712. https://doi.org/10.1890/0012-9658(1997)078[1696:DOSAAA]2.0.CO;2
Bailey A, Wooley SC, Lindroth RL, Whitham TG (2006) Importance of species interactions to community heritability: a genetic basis to trophic-level interactions. Ecol Lett 9:78–85
Barker HL, Riehl JF, Bernhardsson C, Rubert-Nason KF, Holeski LM, Ingvarsson PK, Lindroth RL (2019) Linking plant genes to insect communities: identifying the genetic bases of plant traits and community composition. Mol Ecol 28:4404–4421. https://doi.org/10.1111/mec.15158
Bate-Smith EC, Metcalfe CR (1957) Leucoanthocyanins. 3. The nature and systematic distribution of tannins in dicotyledonous plants. J Linn Soc 55:669–705. https://doi.org/10.1111/j.1095-8339.1957.tb00030.x
Bennett AE, Rubert-Nason KF, Lindroth RL (2020) Response of aspen genotypes to browsing damage is not influenced by soil community diversity. Plant Soil 452:153–170. https://doi.org/10.1007/s11104-020-04466-8
Bryant JP, Chapin FS III, Klein DR (1983) Carbon/nutrient balance of boreal plants in relation to vertebrate herbivory. Oikos 40:357–368. https://doi.org/10.2307/3544308
Cole CT (2005) Allelic and population variation of microsatellite loci in aspen (Populus tremuloides). New Phytol 167:155–164. https://doi.org/10.1111/j.1469-8137.2005.01423.x
Cole CT, Ingvarsson PK (2018) Pathway position constrains the evolution of an ecologically important pathway in aspens (Populus tremula L). Mol Ecol 27:3317–3330. https://doi.org/10.1111/mec.14785
Couture JJ, Lindroth RL (2014) Atmospheric change alters frass quality of forest canopy herbivores. Arthropod-Plant Interact 8:33–47. https://doi.org/10.1007/s11829-013-9286-8
Couture JJ, Meehan TD, Rubert-Nason KF, Lindroth RL (2017) Effects of elevated atmospheric carbon dioxide and tropospheric ozone on phytochemical composition of trembling aspen (Populus tremuloides) and paper birch (Betula papyrifera). J Chem Ecol 43:26–38. https://doi.org/10.1007/s10886-016-0798-4
Gaur RK, Abreu IN, Albrectsen BR (2022) Compensatory phenolic induction dynamics in aspen after aphid infestation. Sci Rep 12:9582. https://doi.org/10.1038/s41598-022-13225-x
Harding SA, Xue LJ, Du L, Nyamdari B, Lindroth RL, Sykes R, Davis MF, Tsai CJ (2014) Condensed tannin biosynthesis and polymerization synergistically condition carbon use, defense and growth in Populus. Tree Physiol 34:1240–1251. https://doi.org/10.1093/treephys/tpt097
Hunter MD (2016) The phytochemical landscape. Princeton University Press, Princeton, New Jersey
Irmisch S, McCormick AC, Günther J, Schmidt A, Boeckler GA, Gershenzon J, Unsicker SB, Köllner TG (2014) Herbivore-induced poplar cytochrome P450 enzymes of the CYP71 family convert aldoximes to nitriles which repel a generalist caterpillar. Plant J 80:1095–1107. https://doi.org/10.1111/tpj.12711
Jamieson MA, Schwartzberg EG, Raffa KF, Reich PB, Lindroth RL (2015) Experimental climate warming alters aspen and birch phytochemistry and performance traits for an outbreak insect herbivore. Glob Change Biol 21:2698–2710. https://doi.org/10.1111/gcb.12842
Jun JH, Lu N, Docampo-Palacios M, Wang X, Dixon RA (2021) Dual activity of anthocyanidin reductase supports the dominant plant proanthocyanidin extension unit pathway. Sci Adv 7:eabg4682. https://doi.org/10.1126/sciadv.abg4682
Keefover-Ring KK, Rubert-Nason KF, Bennett A, Lindroth RL (2016) Growth and chemical responses of trembling aspen to simulated browsing and ungulate saliva. J Plant Ecol 9:474–484. https://doi.org/10.1093/jpe/rtv072
Li Z, Rubert-Nason KF, Jamieson MA, Raffa KF, Lindroth RL (2021) Root secondary metabolites in Populus tremuloides: effects of simulated climate warming, defoliation, and genotype. J Chem Ecol 47:313–321. https://doi.org/10.1007/s10886-021-01259-w
Lin L, Sun S, Chen P, Monagas MJ, Harnly JM (2014) UHPLC-PDA-ESI/HRMSn profiling method to identify and quantify oligomeric proanthocyanidins in plant products. J Agric Food Chem 62:9387–9400. https://doi.org/10.1021/jf501011y
Madritch MD, Donaldson JR, Lindroth RL (2006) Genetic identity of Populus tremuloides litter influences decomposition and nutrient release in a mixed forest stand. Ecosystems 9:528–537. https://doi.org/10.1007/s10021-006-0008-2
Madritch MD, Jordan LM, Lindroth RL (2007) Interactive effects of condensed tannin and cellulose additions on soil respiration. Can J For Res 37:2063–2067. https://doi.org/10.1139/X07-047
Marles M, Ray H, Gruber M (2003) New perspectives on proanthocyanidin biochemistry and molecular regulation. Phytochemistry 64:367–383. https://doi.org/10.1016/s0031-9422(03)00377-7
McWilliam EL, Barry TN, Lopez-Villalobos N, Cameron PN, Kemp PD (2004) The effect of different levels of poplar (Populus) supplementation on the reproductive performance of ewes grazing low quality drought pasture during mating. Anim Feed Sci Technol 115:1–18. https://doi.org/10.1016/j.anifeedsci.2004.03.006
Mole S (1993) The systematic distribution of tannins in the leaves of angiosperms: a tool for ecological studies. Biochem Syst Ecol 21:833–846. https://doi.org/10.1016/0305-1978(93)90096-A
Monson RK, Trowbridge AM, Lindroth RL, Lerdau MT (2022) Coordinated resource allocation to plant growth–defense tradeoffs. New Phytol 233:1051–1066. https://doi.org/10.1111/nph
Müller MS, McWilliams SR, Podlesak D, Donaldson JR, Bothwell HM, Lindroth RL (2006) Tri-trophic effects of plant defenses: chickadees consume caterpillars based on host leaf chemistry. Oikos 114:507–517. https://doi.org/10.1111/j.2006.0030-1299.14668.x
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O’Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6–4 https://CRAN.R-project.org/package=vegan
Osier TL, Lindroth RL (2001) Effects of genotype, nutrient availability, and defoliation on aspen phytochemistry and insect performance. J Chem Ecol 27:1289–1313. https://doi.org/10.1023/A:1010352307301
Philippe RN, Bohlmann J (2007) Poplar defense against herbivores. Can J Bot 85:1111–1126. https://doi.org/10.1139/B07-109
Quideau S, Deffieux D, Douat-Casassus C, Pouysegu L (2011) Plant polyphenols: chemical properties, biological activities, and synthesis. Angewandte Chemie - International Edition 50:586–621. https://doi.org/10.1002/anie.201000044
R Core Team (2022). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Robinson KM, Ingvarsson PK, Jansson S, Albrectsen BR (2012) Genetic variation in functional traits influences arthropod community composition in aspen (Populus tremula L). PLoS ONE 7(5):e37679. https://doi.org/10.1371/journal.pone.0037679
Rubert-Nason KF, Lindroth RL (2019) Analysis of condensed tannins in Populus spp. using reversed phase UPLC-PDA-(-)esiMS following thiolytic depolymerization. Phytochem Anal 30:257–267. https://doi.org/10.1002/pca.2810
Rubert-Nason K, Lindroth RL (2021) Causes and consequences of condensed tannin variation in Populus: a molecules to ecosystems perspective. In: Reed J, de Freitas V, Quideau S (eds) Recent advances in polyphenol research, vol 7. Wiley-Blackwell, Hoboken, New Jersey, pp 69–112
Rubert-Nason KF, Keefover-Ring KM, Lindroth RL (2012) Genotype and environment modulate the response of trembling aspen to simulated ungulate browsing. Ecological Society of America National Meeting, Portland, Oregon. Aug. 5–10, 2012. Session 7683. https://eco.confex.com/eco/2012/webprogram/Session7683.html. Accessed 2 Jan 2023
Rubert-Nason KF, Couture JJ, Major IT, Constabel CP, Lindroth RL (2015) Influence of genotype, environment, and gypsy moth herbivory on local and systemic chemical defenses in trembling aspen (Populus tremuloides). J Chem Ecol 41:651–661. https://doi.org/10.1007/s10886-015-0600-z
Rubert-Nason KF, Couture JJ, Gryzmala EA, Townsend PA, Lindroth RL (2017) Vernal freeze damage and genetic variation alter tree growth, chemistry, and insect interactions. Plant Cell Environ 40:743–2753. https://doi.org/10.1111/pce.13042
Schweitzer JA, Bailey A, Bangert RK, Hart SC, Whitham TG (2007) The role of plant genetic variation in determining above- and belowground microbial communities. In: Bailey MJ, Lilley AK, Timms-Wilson TM, Spencer Phillips PTN (eds) Microbial ecology of aerial plant surfaces. CABI Publishing, Wallingford, UK
Schweitzer JA, Madritch MD, Bailey JK, LeRoy C, Fischer DG, Rehill BJ, Lindroth RL, Hagerman AE, Wooley SC, Hart SC, Whitham TG (2008) From genes to ecosystems: the genetic basis of condensed tannins and their role in nutrient regulation in a Populus model system. Ecosystems 11:1005–1020. https://doi.org/10.1007/s10021-008-9173-9
Scioneaux AN, Schmidt MA, Moore MA, Lindroth RL, Wooley SC, Hagerman AE (2011) Qualitative variation in proanthocyanidin composition of Populus species and hybrids: Genetics is the key. J Chem Ecol 37:57–70. https://doi.org/10.1007/s10886-010-9887-y
Sibley DA (2009) The Sibley guide to trees, 1st edn. Alfred A. Knopf, New York
Slavov TT, Zhelev P (2010) Salient biological features, systematics, and genetic variation of Populus. In: Jansson S, Bhalerao RP, Groover AT (eds) Genetics and genomics of Populus. Springer, New York, pp 15–38
Taiz L, Zeiger E (2010) Plant physiology. Sinauer Associates, Oxford, United Kingdom
Tsai C, Harding SA, Tschaplinski TJ, Lindroth RL, Yuan Y (2006) Genome-wide analysis of the structural genes regulating defense phenylpropanoid metabolism in Populus. New Phytol 172:47–62. https://doi.org/10.1111/j.1469-8137.2006.01798.x
Tuomi J, Niemela P, Chapin FS III, Bryant JP, Siren S (1988) Defensive responses of trees in relation to their carbon/nutrient balance. In: Mattson WJ, Levieux J, Bernard-Dagan C (eds) Mechanisms of woody plant defenses against insects: search for pattern. Springer-Verlag, New York, pp 57–72
Villalba JJ, Burritt EA, St. Clair SB (2014) Aspen (Populus tremuloides Michx.) intake and preference by mammalian herbivores: the role of plant secondary compounds and nutritional context. J Chem Ecol 40:1135–1145. https://doi.org/10.1007/s10886-014-0507-0
Acknowledgements
Specimens analyzed herein were provided by investigators associated with the studies represented: John Couture oversaw the study of O3 and CO2 and assisted with statistical analysis of the corresponding data; Mary Jamieson oversaw the study of warming and lepidopteran defoliation; Ken Keefover-Ring oversaw the study of soil nutrients and mammal browsing; Elizabeth Gryzmala assisted with execution of the study of freeze damage; Zhengzhen Li provided advice on statistical analyses. We thank the two anonymous reviewers whose feedback improved the manuscript.
Funding
This research was funded by the National Science Foundation (DEB-0841609) and the USDA National Institute of Food and Agriculture (AFRI grant 2016–7013–25088).
Author information
Authors and Affiliations
Contributions
K. Rubert-Nason and R. Lindroth conceived this study and developed the manuscript. P. Yang collaborated with K. Rubert-Nason on the development and execution of the method used for analysis of condensed tannin quality. C. Morrow conducted and interpreted principal component ordination statistical analyses and R. Lindroth secured funding for the research.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Competing Interests
The authors have no competing interests to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rubert-Nason, K.F., Yang, P., Morrow, C.J. et al. Environment and Genotype Influence Quantitative and Qualitative Variation in Condensed Tannins in Aspen. J Chem Ecol 49, 325–339 (2023). https://doi.org/10.1007/s10886-023-01430-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-023-01430-5