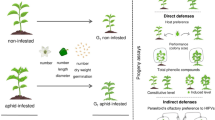
Abstract
Herbivorous insects are important problems in cranberry (Vaccinium macrocarpon Ait.) production. The use of chemical pesticides is common practice, but beneficial insects such as natural enemies of herbivores (e.g. predators and parasitoids) could be affected as well. Therefore, we studied the defensive mechanisms that cranberry plants use to combat pests, focusing on herbivore-induced plant volatiles (HIPVs), which can be used to recruit predators and parasitoids foraging for prey or hosts. Then, we used synthetic HIPVs to test the attraction of natural enemies. In a greenhouse, we assessed nine cranberry genotypes for expression of genes involved in HIPV biosynthesis and/or emission of HIPVs. In an experimental field, we assessed whether baiting traps with individual or combinations of HIPVs increased attractiveness to natural enemies. The results showed that different cranberry genotypes vary in their emission of monoterpenes and sesquiterpenes but not in their expression of two genes associated with terpene biosynthesis, α-humulene/β-caryophyllene synthase and (3S,6E)-nerolidol/R-linalool synthase. Induction with methyl jasmonate or herbivore (gypsy moth, Lymantria dispar L.) feeding increased the expression of these genes and emission of HIPVs. The HIPV methyl salicylate (MeSA), alone or in combination with other HIPVs, increased syrphid attraction by 6-fold in the field, while (Z)-3-hexenyl acetate and MeSA repelled ladybeetles and megaspilids, respectively. Linalool and β-caryophyllene elicited no behavioral responses of natural enemies. Elucidating the mechanisms of pest resistance, as well as experimentally augmenting plant defenses such as HIPVs, may contribute to the development of more sustainable pest management practices in crops, including cranberries.




Similar content being viewed by others
References
Alborn HT, Turlings TCJ, Jones TH, Stenhagen G, Loughrin JH, Tumlinson JH (1997) An elicitor of plant volatiles from beet armyworm oral secretion. Science 276:945–949. https://doi.org/10.1126/science.276.5314.945
Averill AL, Sylvia MM (1998) Cranberry insects of the Northeast - a guide to identification, biology and management. Cranberry Station Fact Sheets. Paper 30. http://www.umass.edu/cranberry/downloads/Cranberry%20Insects%20of%20the%20NorthEast.Averill.Sylvia.Franklin.2000.pdf. Accessed 09/17/2018
Beninger CW, Abou-Zaid MM (1997) Flavonol glycosides from four pine species that inhibit early instar gypsy moth (Lepidoptera: Lymantriidae) development. Biochem Syst Ecol 25:505–512. https://doi.org/10.1016/S0305-1978(97)00034-3
Braasch J, Wimp GM, Kaplan I (2012) Testing for phytochemical synergism: arthropod community responses to induced plant volatile blends across crops. J Chem Ecol 38:1264–1275. https://doi.org/10.1007/s10886-012-0202-y
Bricchi I, Leitner M, Foti M, Mithöfer A, Boland W, Maffei ME (2010) Robotic mechanical wounding (MecWorm) versus herbivore-induced responses: early signaling and volatile emission in lima bean (Phaseolus lunatus L.). Planta 232:719–729. https://doi.org/10.1007/s00425-010-1203-0
Bruce TJA, Smart LE, Birch ANE, Blok VC, MacKenzie K, Guerrieri E, Cascone P, Luna E, Ton J (2017) Prospects for plant defence activators and biocontrol in IPM – concepts and lessons learnt so far. Crop Prot 97:128–134. https://doi.org/10.1016/j.cropro.2016.10.003
Chandler FB, Demoranville I (1958) Cranberry varieties of North America. Mass Agric Exp Sta Bull 513:1–23
Chen YH, Gols R, Benrey B (2015) Crop domestication and its impact on naturally selected trophic interactions. Annu Rev Entomol 60:35–58. https://doi.org/10.1146/annurev-ento-010814-020601
Christensen SA, Nemchenko A, Borrego E, Murray I, Sobhy IS, Bosak L, DeBlasio S, Erb M, Robert CAM, Vaughn KA, Herrfurth C, Tumlinson J, Feussner I, Jackson D, Turlings TCJ, Engelberth J, Nansen C, Meeley R, Kolomiets MV (2013) The maize lipoxygenase, ZmLOX10, mediates green leaf volatile, jasmonate and herbivore-induced plant volatile production for defense against insect attack. Plant J 74:59–73. https://doi.org/10.1111/tpj.12101
Cranberry Marketing Committee (2018) Cranberry varieties. http://reports.uscranberries.com/TheCranberryStory/Varieties/. Accessed 17 Sep 2018
D'Alessandro M, Turlings TCJ (2006) Advances and challenges in the identification of volatiles that mediate interactions among plants and arthropods. Analyst 131:24–32. https://doi.org/10.1039/B507589K
Dana MN (1983) Cranberry cultivar list (Vaccinium macrocarpon). Fruit Var J 37:88–95
De Lange ES, Balmer D, Mauch-Mani B, Turlings TCJ (2014) Insect and pathogen attack and resistance in maize and its wild ancestors, the teosintes. New Phytol 204:329–341. https://doi.org/10.1111/nph.13005
De Lange ES, Farnier K, Gaudillat B, Turlings TCJ (2016) Comparing the attraction of two parasitoids to herbivore-induced volatiles of maize and its wild ancestors, the teosintes. Chemoecology 26:33–44. https://doi.org/10.1007/s00049-015-0205-6
Degen T, Dillmann C, Marion-Poll F, Turlings TCJ (2004) High genetic variability of herbivore-induced volatile emission within a broad range of maize inbred lines. Plant Physiol 135:1928–1938. https://doi.org/10.1104/pp.104.039891
Erb M, Flors V, Karlen D, de Lange E, Planchamp C, D’Alessandro M, Turlings TCJ, Ton J (2009) Signal signature of aboveground-induced resistance upon belowground herbivory in maize. Plant J 59:292–302. https://doi.org/10.1111/j.1365-313X.2009.03868.x
Erb M et al (2011) Synergies and trade-offs between insect and pathogen resistance in maize leaves and roots. Plant Cell Environ 34:1088–1103. https://doi.org/10.1111/j.1365-3040.2011.02307.x
Erb M, Meldau S, Howe GA (2012) Role of phytohormones in insect-specific plant reactions. Trends Plant Sci 17:250–259. https://doi.org/10.1016/j.tplants.2012.01.003
Flint HM, Salter SS, Walters S (1979) Caryophyllene: an attractant for the green lacewing. Environ Entomol 8:1123–1125. https://doi.org/10.1093/ee/8.6.1123
Hall DE, MacGregor KB, Nijsse J, Bown AW (2004) Footsteps from insect larvae damage leaf surfaces and initiate rapid responses. Eur J Plant Pathol 110:441–447. https://doi.org/10.1023/B:EJPP.0000021072.89968.de
Hare JD (2010) Ecological role of volatiles produced by plants in response to damage by herbivorous insects. Annu Rev Entomol 56:161–180. https://doi.org/10.1146/annurev-ento-120709-144753
Hernandez-Cumplido J, Giusti MM, Zhou Y, Kyryczenko-Roth V, Chen YH, Rodriguez-Saona C (2018) Testing the ‘plant domestication-reduced defense’ hypothesis in blueberries: the role of herbivore identity. Arthropod Plant Interact 12:483–493. https://doi.org/10.1007/s11829-018-9605-1
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
James DG (2003) Synthetic herbivore-induced plant volatiles as field attractants for beneficial insects. Environ Entomol 32:977–982. https://doi.org/10.1603/0046-225X-32.5.977
James DG (2005) Further field evaluation of synthetic herbivore-induced plan volatiles as attractants for beneficial insects. J Chem Ecol 31:481–495. https://doi.org/10.1007/s10886-005-2020-y
Jones VP, Steffan SA, Wiman NG, Horton DR, Miliczky E, Zhang Q-H, Baker CC (2011) Evaluation of herbivore-induced plant volatiles for monitoring green lacewings in Washington apple orchards. Biol Control 56:98–105. https://doi.org/10.1016/j.biocontrol.2010.10.001
Kaplan I (2012) Attracting carnivorous arthropods with plant volatiles: the future of biocontrol or playing with fire? Biol Control 60:77–89. https://doi.org/10.1016/j.biocontrol.2011.10.017
Kappers IF, Hoogerbrugge H, Bouwmeester HJ, Dicke M (2011) Variation in herbivory-induced volatiles among cucumber (Cucumis sativus L.) varieties has consequences for the attraction of carnivorous natural enemies. J Chem Ecol 37:150–160. https://doi.org/10.1007/s10886-011-9906-7
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144. https://doi.org/10.1126/science.291.5511.2141
Köllner TG, Held M, Lenk C, Hiltpold I, Turlings TCJ, Gershenzon J, Degenhardt J (2008) A maize (E)-β-caryophyllene synthase implicated in indirect defense responses against herbivores is not expressed in most American maize varieties. Plant Cell 20:482–494. https://doi.org/10.1105/tpc.107.051672
Lattanzio V, Cardinali A, Linsalata V (2012) Plant phenolics: a biochemical and physiological perspective. In: Cheynier V, Sarni-Manchado P, Quideau S (eds) Recent advances in polyphenol research, vol 3. Wiley-Blackwell, Chichester, pp 1–39. https://doi.org/10.1002/9781118299753.ch1
Loughrin JH, Manukian A, Heath RR, Tumlinson JH (1995) Volatiles emitted by different cotton varieties damaged by feeding beet armyworm larvae. J Chem Ecol 21:1217–1227. https://doi.org/10.1007/bf02228321
Minott CW (1922) The gipsy moth on cranberry bogs. USDA Bulletin 1093:1–19
Müller CB, Godfray HCJ (1998) The response of aphid secondary parasitoids to different patch densities of their host. BioControl 43:129–139. https://doi.org/10.1023/a:1009948213250
Oerke EC (2006) Crop losses to pests. J Agric Sci 144:31–43. https://doi.org/10.1017/S0021859605005708
Peñaflor MFGV, Bento JMS (2013) Herbivore-induced plant volatiles to enhance biological control in agriculture. Neotrop Entomol 42:331–343. https://doi.org/10.1007/s13744-013-0147-z
Pickett JA, Khan ZR (2016) Plant volatile-mediated signalling and its application in agriculture: successes and challenges. New Phytol 212:856–870. https://doi.org/10.1111/nph.14274
Pieterse CMJ, Van der Does D, Zamioudis C, Leon-Reyes A, Van Wees SCM (2012) Hormonal modulation of plant immunity. Annu Rev Cell Dev Biol 28:489–521. https://doi.org/10.1146/annurev-cellbio-092910-154055
Polashock JJ, Vorsa N (2002) Development of SCAR markers for DNA fingerprinting and germplasm analysis of American cranberry. J Am Soc Hortic Sci 127:677–684
Polashock J, Zelzion E, Fajardo D, Zalapa J, Georgi L, Bhattacharya D, Vorsa N (2014) The American cranberry: first insights into the whole genome of a species adapted to bog habitat. BMC Plant Biol 14:165. https://doi.org/10.1186/1471-2229-14-165
Price PW, Bouton CE, Gross P, McPheron BA, Thompson JN, Weis AE (1980) Interactions among three trophic levels: influence of plants on interactions between insect herbivores and natural enemies. Annu Rev Ecol Syst 11:41–65
Rasmann S, Köllner TG, Degenhardt J, Hiltpold I, Toepfer S, Kuhlmann U, Gershenzon J, Turlings TCJ (2005) Recruitment of entomopathogenic nematodes by insect-damaged maize roots. Nature 434:732–737. https://doi.org/10.1038/nature03451
Reymond P, Weber H, Damond M, Farmer EE (2000) Differential gene expression in response to mechanical wounding and insect feeding in Arabidopsis. Plant Cell 12:707–719. https://doi.org/10.1105/tpc.12.5.707
Rodriguez-Saona CR, Rodriguez-Saona LE, Frost CJ (2009) Herbivore-induced volatiles in the perennial shrub, Vaccinium corymbosum, and their role in inter-branch signaling. J Chem Ecol 35:163–175. https://doi.org/10.1007/s10886-008-9579-z
Rodriguez-Saona C, Kaplan I, Braasch J, Chinnasamy D, Williams L (2011a) Field responses of predaceous arthropods to methyl salicylate: a meta-analysis and case study in cranberries. Biol Control 59:294–303. https://doi.org/10.1016/j.biocontrol.2011.06.017
Rodriguez-Saona C, Vorsa N, Singh AP, Johnson-Cicalese J, Szendrei Z, Mescher MC, Frost CJ (2011b) Tracing the history of plant traits under domestication in cranberries: potential consequences on anti-herbivore defences. J Exp Bot 62:2633–2644. https://doi.org/10.1093/jxb/erq466
Rodriguez-Saona C, Polashock J, Malo EA (2013) Jasmonate-mediated induced volatiles in the American cranberry, Vaccinium macrocarpon: from gene expression to organismal interactions. Front Plant Sci 4:115. https://doi.org/10.3389/fpls.2013.00115
Rosenthal JP, Dirzo R (1997) Effects of life history, domestication and agronomic selection on plant defence against insects: evidence from maizes and wild relatives. Evol Ecol 11:337–355. https://doi.org/10.1023/A:101842050
Rostás M, Turlings TCJ (2008) Induction of systemic acquired resistance in Zea mays also enhances the plant’s attractiveness to parasitoids. Biol Control 46:178–186. https://doi.org/10.1016/j.biocontrol.2008.04.012
Rowen E, Kaplan I (2016) Eco-evolutionary factors drive induced plant volatiles: a meta-analysis. New Phytol 210:284–294. https://doi.org/10.1111/nph.13804
Salamanca J, Pareja M, Rodriguez-Saona C, Resende ALS, Souza B (2015) Behavioral responses of adult lacewings, Chrysoperla externa, to a rose-aphid-coriander complex. Biol Control 80:103–112. https://doi.org/10.1016/j.biocontrol.2014.10.003
Salamanca J, Souza B, Lundgren JG, Rodriguez-Saona C (2017) From laboratory to field: electro-antennographic and behavioral responsiveness of two insect predators to methyl salicylate. Chemoecology 27:51–63. https://doi.org/10.1007/s00049-017-0230-8
Salamanca J, Souza B, Rodriguez-Saona C (2018) Cascading effects of combining synthetic herbivore-induced plant volatiles with companion plants to manipulate natural enemies in an agro-ecosystem. Pest Manag Sci 74:2133–2145. https://doi.org/10.1002/ps.4910
Schmidt MH, Lauer A, Purtauf T, Thies C, Schaefer M, Tscharntke T (2003) Relative importance of predators and parasitoids for cereal aphid control. Proc R Soc Lond B Biol Sci 270:1905–1909. https://doi.org/10.1098/rspb.2003.2469
Scott AJ, Knott M (1974) A cluster analysis method for grouping means in the analysis of variance. Biometrics 30:507–512. https://doi.org/10.2307/2529204
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52:591–611. https://doi.org/10.2307/2333709
Shlichta JG, Cuny MAC, Hernandez-Cumplido J, Traine J, Benrey B (2018) Contrasting consequences of plant domestication for the chemical defenses of leaves and seeds in lima bean plants. Basic Appl Ecol 31:10–20. https://doi.org/10.1016/j.baae.2018.05.012
Snoeren TAL, Mumm R, Poelman EH, Yang Y, Pichersky E, Dicke M (2010) The herbivore-induced plant volatile methyl salicylate negatively affects attraction of the parasitoid Diadegma semiclausum. J Chem Ecol 36:479–489. https://doi.org/10.1007/s10886-010-9787-1
Sobhy IS, Erb M, Sarhan AA, El-Husseini MM, Mandour NS, Turlings TCJ (2012) Less is more: treatment with BTH and laminarin reduces herbivore-induced volatile emissions in maize but increases parasitoid attraction. J Chem Ecol 38:348–360. https://doi.org/10.1007/s10886-012-0098-6
Sobhy IS, Erb M, Lou Y, Turlings TCJ (2014) The prospect of applying chemical elicitors and plant strengtheners to enhance the biological control of crop pests. Philos Trans Royal Soc B Biol Sci 369:20120283. https://doi.org/10.1098/rstb.2012.0283
Sobhy IS, Erb M, Turlings TCJ (2015) Plant strengtheners enhance parasitoid attraction to herbivore-damaged cotton via qualitative and quantitative changes in induced volatiles. Pest Manag Sci 71:686–693. https://doi.org/10.1002/ps.3821
Sobhy IS, Bruce TJ, Turlings TC (2018) Priming of cowpea volatile emissions with defense inducers enhances the plant's attractiveness to parasitoids when attacked by caterpillars. Pest Manag Sci 74:966–977. https://doi.org/10.1002/ps.4796
Stenberg JA, Heil M, Åhman I, Björkman C (2015) Optimizing crops for biocontrol of pests and disease. Trends Plant Sci 20:698–712. https://doi.org/10.1016/j.tplants.2015.08.007
Stoyenoff JL, Witter JA, Montgomery ME, Chilcote CA (1994) Effects of host switching on gypsy moth (Lymantria dispar (L.)) under field conditions. Oecologia 97:143–157. https://doi.org/10.1007/bf00323144
Tahmasebi Z, Mohammadi H, Arimura G-I, Muroi A, Kant MR (2014) Herbivore-induced indirect defense across bean cultivars is independent of their degree of direct resistance. Exp Appl Acarol 63:217–239. https://doi.org/10.1007/s10493-014-9770-6
Tjiurutue MC, Sandler HA, Kersch-Becker MF, Theis N, Adler LS (2017) Gypsy moth herbivory induced volatiles and reduced parasite attachment to cranberry hosts. Oecologia 185:133–145. https://doi.org/10.1007/s00442-017-3915-3
Turlings TCJ, Erb M (2018) Tritrophic interactions mediated by herbivore-induced plant volatiles: mechanisms, ecological relevance, and application potential. Annu Rev Entomol 63:433–452. https://doi.org/10.1146/annurev-ento-020117-043507
Turlings TCJ, Tumlinson JH, Lewis WJ (1990) Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 250:1251–1253. https://doi.org/10.1126/science.250.4985.1251
Van den Boom CEM, Van Beek TA, Posthumus MA, De Groot A, Dicke M (2004) Qualitative and quantitative variation among volatile profiles induced by Tetranychus urticae feeding on plants from various families. J Chem Ecol 30:69–89. https://doi.org/10.1023/b:joec.0000013183.72915.99
Von Mérey G, Veyrat N, Mahuku G, Valdez RL, Turlings TCJ, D’Alessandro M (2011) Dispensing synthetic green leaf volatiles in maize fields increases the release of sesquiterpenes by the plants, but has little effect on the attraction of pest and beneficial insects. Phytochemistry 72:1838–1847. https://doi.org/10.1016/j.phytochem.2011.04.022
Von Mérey GE et al (2012) Minor effects of two elicitors of insect and pathogen resistance on volatile emissions and parasitism of Spodoptera frugiperda in Mexican maize fields. Biol Control 60:7–15. https://doi.org/10.1016/j.biocontrol.2011.09.010
Vorsa N (2010) Cranberry. In: Clark JR, Finn CE (eds) Register of new fruit and nut cultivars list 45. HortScience 45:716–756
Vorsa N, Johnson-Cicalese J (2012) American cranberry. In: Badenes ML, Byrne DH (eds) Fruit breeding. Springer US, Boston, pp 191–223. https://doi.org/10.1007/978-1-4419-0763-9_6
Whitehead SR, Turcotte MM, Poveda K (2017) Domestication impacts on plant-herbivore interactions: a meta-analysis. Philos Trans Royal Soc B Biol Sci 372:20160034. https://doi.org/10.1098/rstb.2016.0034
Whitman DW, Eller FJ (1990) Parasitic wasps orient to green leaf volatiles. Chemoecology 1:69–76. https://doi.org/10.1007/bf01325231
Whitman DW, Eller FJ (1992) Orientation of Microplitis croceipes (Hymenoptera: Braconidae) to green leaf volatiles: dose-response curves. J Chem Ecol 18:1743–1753. https://doi.org/10.1007/bf02751099
Wickremasinghe MGV, Van Emden HF (1992) Reactions of adult female parasitoids, particularly Aphidius rhopalosiphi, to volatile chemical cues from the host plants of their aphid prey. Physiol Entomol 17:297–304. https://doi.org/10.1111/j.1365-3032.1992.tb01025.x
Yu H, Zhang Y, Wu K, Gao XW, Guo YY (2008) Field-testing of synthetic herbivore-induced plant volatiles as attractants for beneficial insects. Environ Entomol 37:1410–1415. https://doi.org/10.1603/0046-225X-37.6.1410
Yu H, Khashaveh A, Li Y, Li X, Zhang Y (2018) Field trapping of predaceous insects with synthetic herbivore-induced plant volatiles in cotton fields. Environ Entomol 47:114–120. https://doi.org/10.1093/ee/nvx201
Zhu J, Park K-C (2005) Methyl salicylate, a soybean aphid-induced plant volatile attractive to the predator Coccinella septempunctata. J Chem Ecol 31:1733–1746. https://doi.org/10.1007/s10886-005-5923-8
Zhu J, Cossé AA, Obrycki JJ, Boo KS, Baker TC (1999) Olfactory reactions of the twelve-spotted lady beetle, Coleomegilla maculata and the green lacewing, Chrysoperla carnea to semiochemicals released from their prey and host plant: electroantennogram and behavioral responses. J Chem Ecol 25:1163–1177. https://doi.org/10.1023/a:1020846212465
Zhu J, Obrycki JJ, Ochieng SA, Baker TC, Pickett JA, Smiley D (2005) Attraction of two lacewing species to volatiles produced by host plants and aphid prey. Naturwissenschaften 92:277–281. https://doi.org/10.1007/s00114-005-0624-2
Acknowledgements
We thank Vera Kyryczenko-Roth, Rob Holdcraft, Kristy Adams, Manuel Chacón, Charles Corris, Kris Dahl, Diego Fraga, Jennifer Frake, Andrew Lux, Melissa Meagher, Caryn Michael, Sarah Ongaro, Langley Oudemans, Gabrielle Pintauro, and Mara Schiffhauer for technical assistance. We also thank Dr. Thomas Hartman (Rutgers University, Mass Spectrometry Support Facility) for assistance in volatile identification, Matthew Kramer (USDA-ARS, Biometric Consulting Service) for assistance in statistical analysis of gene expression data and Dr. Brittany Dodson (Copy Editor & Science Writer; Bellefonte, PA) for editing services. This work was supported by the hatch project No. NJ08192 and funds from the New Jersey Blueberry and Cranberry Research Council, Inc., Cranberry Institute, Cape Cod Cranberry Growers Association, Canadian Cranberry Growers Coalition, and Ocean Spray Cranberries, Inc. to CR-S.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 63 kb)
Rights and permissions
About this article
Cite this article
De Lange, E.S., Salamanca, J., Polashock, J. et al. Genotypic Variation and Phenotypic Plasticity in Gene Expression and Emissions of Herbivore-Induced Volatiles, and their Potential Tritrophic Implications, in Cranberries. J Chem Ecol 45, 298–312 (2019). https://doi.org/10.1007/s10886-018-1043-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-1043-0