Abstract
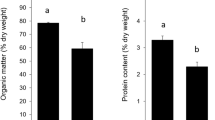
Lichen-gastropod interactions generally focus on the potential deterrent or toxic role of secondary metabolites. To better understand lichen-gastropod interactions, a controlled feeding experiment was designed to identify the parts of the lichen Argopsis friesiana consumed by the Subantarctic land snail Notodiscus hookeri. Besides profiling secondary metabolites in various lichen parts (apothecia, cephalodia, phyllocladia and fungal axis of the pseudopodetium), we investigated potentially beneficial resources that snails can utilize from the lichen (carbohydrates, amino acids, fatty acids, polysaccharides and total nitrogen). Notodiscus hookeri preferred cephalodia and algal layers, which had high contents of carbohydrates, nitrogen, or both. Apothecia were avoided, perhaps due to their low contents of sugars and polyols. Although pseudopodetia were characterized by high content of arabitol, they were also rich in medullary secondary compounds, which may explain why they were not consumed. Thus, the balance between nutrients (particularly nitrogen and polyols) and secondary metabolites appears to play a key role in the feeding preferences of this snail.




Similar content being viewed by others
References
Adams MA, Chen Z, Landman P, Colmer TD (1999) Simultaneous determination by capillary gas chromatography of organic acids, sugars, and sugar alcohols in plant tissue extracts as their trimethylsilyl derivatives. Anal Biochem 266:77–84. https://doi.org/10.1006/abio.1998.2906
Arakelova ES, Chebotareva MA, Zabelinskii SA, Ivanova VP (2009) Effect of habitat and motor activity of molluscs on fatty acid composition of triglycerides and phospholipids. J Evol Biochem Physiol 45:51–58. https://doi.org/10.1134/S0022093009010049
Asplund J (2011) Snails avoid the medulla of Lobaria pulmonaria and L. scrobiculata due to presence of secondary compounds. Fungal Ecol 4:356–358. https://doi.org/10.1016/j.funeco.2011.05.002
Asplund J, Gauslaa Y (2010) The gastropod Arion fuscus prefers cyanobacterial to green algal parts of the tripartite lichen Nephroma arcticum due to low chemical defence. Lichenologist 42:113–117. https://doi.org/10.1017/S0024282909990284
Asplund J, Johansson O, Nybakken L, Palmqvist K, Gauslaa Y (2010) Simulated nitrogen deposition influences gastropod grazing in lichens. Ecoscience 17:83–89. https://doi.org/10.2980/17-1-3331
Asplund J, Solhaug KA, Gauslaa Y (2010) Optimal defense: snails avoid reproductive parts of the lichen Lobaria scrobiculata due to internal defense allocation. Ecology 91:3100–3105
Asplund J, Wardle DA (2013) The impact of secondary compounds and functional characteristics on lichen palatability and decomposition. J Ecol 101:689–700. https://doi.org/10.1111/1365-2745.12075
Asplund J, Wardle DA (2016) How lichens impact on terrestrial community and ecosystem properties. Biol Rev 92:1720–1738. https://doi.org/10.1111/brv.12305
Baron M, Albert P, Gorin J, Iacomini M (1989) Structural studies on a Galactomannan isolated from the lichen Stereocaulon ramulosum. Agric Biol Chem 53:1751–1758
Baron M, Iacomini M, Fantat ES, Gorin PAJ (1991) Galactomannan, lichenan and isolichenan from the polysaccharide-rich lichen Newropogon aurantiaco-ater. Phytochemistry 30:3125–3126. https://doi.org/10.1016/S0031-9422(00)98266-9
Baur B, Fröberg L, Baur A, Guggenheim R, Haase M (2000) Ultrastructure of snail grazing damage to calcicolous lichens. Nord J Bot 20:119–128. https://doi.org/10.1111/j.1756-1051.2000.tb00741.x
Blakeney AB, Harris PJ, Henry RJ, Stone BA (1983) A simple and rapid preparation of alditol acetates for monosaccharide analysis. Carbohydr Res 113:291–299. https://doi.org/10.1016/0008-6215(83)88244-5
Bodo B, Molho D (1974) Structure of argopsin, new chlorodepsidone from lichen Argopsis megalospora. CR Acad Sci C 278:625–627
Charrier M, Marie A, Guillaume D, Bédouet L, le Lannic J, Roiland C, Berland S, Pierre JS, le Floch M, Frenot Y, Lebouvier M (2013) Soil calcium availability influences shell ecophenotype formation in the Sub-Antarctic land snail, Notodiscus hookeri. PLoS One 8:e84527. https://doi.org/10.1371/journal.pone.0084527
Charrier M, Rouland C (2001) Mannan-degrading enzymes purified from the crop of the brown garden snail Helix aspersa Müller (Gastropoda Pulmonata). J Exp Zool 290:125–135. https://doi.org/10.1002/jez.1042
Chevalier L, Le Coz-Bouhnik M, Charrier M (2003) Influence of inorganic compounds on food selection by the brown garden snail Cornu aspersum (Muller)(Gastropoda: Pulmonata). Malacologia 45:125–132
Coker PD (1967) Damage to lichens by gastropods. Lichenologist 3:428–428. https://doi.org/10.1017/S0024282967000465
Cook RT, Bailey SER, McCrohan CR et al (2000) The influence of nutritional status on the feeding behaviour of the field slug, Deroceras reticulatum (Müller). Anim Behav 59:167–176. https://doi.org/10.1006/anbe.1999.1275
Cook WE, Raisbeck MF, Cornish TE, Williams ES, Brown B, Hiatt G, Kreeger TJ (2007) Paresis and death in elk (Cervus elaphus) due to lichen intoxication in Wyoming. J Wildl Dis 43:498–503. https://doi.org/10.7589/0090-3558-43.3.498
Denny M (1980) The role of gastropod pedal mucus in locomotion. Nature 285:160–161. https://doi.org/10.1038/285160a0
Flari V, Matoub M, Rouland C (1995) Purification and characterization of a β-mannanase from the digestive tract of the edible snail Helix lucorum L. Carbohydr Res 275:207–213. https://doi.org/10.1016/0008-6215(95)00136-H
Gadea A, Le Pogam P, Biver G et al (2017) Which specialized metabolites does the native subantarctic gastropod Notodiscus hookeri extract from the consumption of the lichens Usnea taylorii and Pseudocyphellaria crocata? Molecules 22:425. https://doi.org/10.3390/molecules22030425
Galloway DJ (1980) The lichen genera Argopsis and Stereocaulon in New Zealand. Bot Notiser 133:261–279
Gargas A, DePriest PT, Grube M, Tehler A (1995) Multiple origins of lichen symbioses in fungi suggested by SSU rDNA phylogeny. Science 268:1492–1495. https://doi.org/10.1126/science.7770775
Gauslaa Y (2005) Lichen palatability depends on investments in herbivore defence. Oecologia 143:94–105. https://doi.org/10.1007/s00442-004-1768-z
Gerson U (1973) Lichen-arthropod associations. Lichenologist 5:434–443. https://doi.org/10.1017/S0024282973000484
Gravot A, Dittami SM, Rousvoal S, Lugan R, Eggert A, Collén J, Boyen C, Bouchereau A, Tonon T (2010) Diurnal oscillations of metabolite abundances and gene analysis provide new insights into central metabolic processes of the brown alga Ectocarpus siliculosus. New Phytol 188:98–110. https://doi.org/10.1111/j.1469-8137.2010.03400.x
Hervé M (2016) RVAideMemoire: diverse basic statistical and graphical functions
Hesbacher S, Baur B, Baur A, Proksch P (1995) Sequestration of lichen compounds by three species of terrestrial snails. J Chem Ecol 21:233–246. https://doi.org/10.1007/BF02036654
Hoebler C, Barry JL, David A, Delort-Laval J (1989) Rapid acid hydrolysis of plant cell wall polysaccharides and simplified quantitative determination of their neutral monosaccharides by gas-liquid chromatography. J Agric Food Chem 37:360–367. https://doi.org/10.1021/jf00086a020
Huneck S, Lamb IM (1975) 1′-Chloropannarin, a new depsidone from Argopsis friesiana: Notes on the structure of pannarin and on the chemistry of the lichen genus Argopsis. Phytochemistry 14:1625–1628. https://doi.org/10.1016/0031-9422(75)85363-5
Hyvärinen M, Koopmann R, Hormi O, Tuomi J (2000) Phenols in reproductive and somatic structures of lichens: a case of optimal defence? Oikos 91:371–375. https://doi.org/10.1034/j.1600-0706.2000.910217.x
Indahl UG, Liland KH, Næs T (2009) Canonical partial least squares—a unified PLS approach to classification and regression problems. J Chemometrics 23:495–504. https://doi.org/10.1002/cem.1243
Lamb IM (1974) The lichen genus Argopsis Th. Fr J Hattori Bot Lab 38:447–462
Lawrey JD (1983) Vulpinic and pinastric acids as lichen antiherbivore compounds: Contrary evidence. Bryologist 86:365. https://doi.org/10.2307/3243250
Lawrey JD (2009) Chemical defense in lichen symbioses. In: White JF, Torres MS (eds) Defensive Mutualism in Microbial Symbiosis. CRC Press, USA, pp 167–176
Le Pogam P, Le Lamer A-C, Legouin B et al (2016) In situ DART-MS as a versatile and rapid dereplication tool in lichenology: chemical fingerprinting of Ophioparma ventosa. Phytochem Anal 27:354–363. https://doi.org/10.1002/pca.2635
Le Pogam P, Legouin B, Le Lamer A-C et al (2015) Analysis of the cyanolichen Lichina pygmaea metabolites using in situ DART-MS: from detection to thermochemistry of mycosporine serinol. J Mass Spectrom 50:454–462. https://doi.org/10.1002/jms.3549
Lugan R, Niogret M-F, Kervazo L et al (2009) Metabolome and water status phenotyping of Arabidopsis under abiotic stress cues reveals new insight into ESK1 function. Plant Cell Environ 32:95–108. https://doi.org/10.1111/j.1365-3040.2008.01898.x
McKey D (1974) Adaptive patterns in alkaloid physiology. Am Nat 108:305–320. https://doi.org/10.1086/282909
Molnár K, Farkas E (2010) Current results on biological activities of lichen secondary metabolites: a review. Z Naturforsch C 65:157–173
Ng TPT, Saltin SH, Davies MS, Johannesson K, Stafford R, Williams GA (2013) Snails and their trails: the multiple functions of trail-following in gastropods. Biol Rev 88:683–700. https://doi.org/10.1111/brv.12023
Nicolai A, Filser J, Lenz R, Valérie B, Charrier M (2012) Composition of body storage compounds influences egg quality and reproductive investment in the land snail Cornu aspersum. Can J Zool 90:1161–1170. https://doi.org/10.1139/z2012-081
Nimis PL, Skert N (2006) Lichen chemistry and selective grazing by the coleopteran Lasioderma serricorne. Environ Exp Bot 55:175–182. https://doi.org/10.1016/j.envexpbot.2004.10.011
Nybakken L, Helmersen A-M, Gauslaa Y, Selås V (2010) Lichen compounds restrain lichen feeding by bank voles (Myodes glareolus). J Chem Ecol 36:298–304. https://doi.org/10.1007/s10886-010-9761-y
Pöykkö H, Hyvärinen M, Bačkor M (2005) Removal of lichen secondary metabolites affects food choice and survival of lichenivorous moth larvae. Ecology 86:2623–2632. https://doi.org/10.1890/04-1632
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rai A, Bergman B (2002) Cyanolichens. Biol Environ. Proc R Ir Acad 102:19–22. http://www.jstor.org/stable/20500135
Renner B (1982) Études microspectrophotométriques sur les céphalodies de Pseudocyphellaria faveolata. Can J Bot 60:630–633. https://doi.org/10.1139/b82-083
Rezanka T, Dembitsky VM (1999) Fatty acids of lichen species from Tian Shan mountains. Folia Microbiol 44:643–646. https://doi.org/10.1007/BF02825654
Rhee KC (2001) Determination of total nitrogen. Curr Protoc Food Analyt Chem B1(2):B1.2.1–B1.2.9. https://doi.org/10.1002/0471142913.fab0102s00
Richardson D, Hill D, Smith D (1968) Lichen physiology. New Phytol 67:469–486. https://doi.org/10.1111/j.1469-8137.1968.tb05476.x
Roullier C, Chollet-Krugler M, Pferschy-Wenzig E-M, Maillard A, Rechberger GN, Legouin-Gargadennec B, Bauer R, Boustie J (2011) Characterization and identification of mycosporines-like compounds in cyanolichens. Isolation of mycosporine hydroxyglutamicol from Nephroma laevigatum Ach. Phytochemistry 72:1348–1357. https://doi.org/10.1016/j.phytochem.2011.04.002
Rundel PW (1978) The ecological role of secondary lichen substances. Biochem Syst Ecol 6:157–170. https://doi.org/10.1016/0305-1978(78)90002-9
Sanders WB (2001) Lichens: the interface between mycology and plant morphology : whereas most other fungi live as an absorptive mycelium inside their food substrate, the lichen fungi construct a plant-like body within which photosynthetic algal symbionts are cultivated. Bioscience 51:1025–1035. https://doi.org/10.1641/0006-3568(2001)051[1025:LTIBMA]2.0.CO;2
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA, Chen W, Fungal Barcoding Consortium, Fungal Barcoding Consortium Author List, Bolchacova E, Voigt K, Crous PW, Miller AN, Wingfield MJ, Aime MC, An KD, Bai FY, Barreto RW, Begerow D, Bergeron MJ, Blackwell M, Boekhout T, Bogale M, Boonyuen N, Burgaz AR, Buyck B, Cai L, Cai Q, Cardinali G, Chaverri P, Coppins BJ, Crespo A, Cubas P, Cummings C, Damm U, de Beer ZW, de Hoog GS, del-Prado R, Dentinger B, Dieguez-Uribeondo J, Divakar PK, Douglas B, Duenas M, Duong TA, Eberhardt U, Edwards JE, Elshahed MS, Fliegerova K, Furtado M, Garcia MA, Ge ZW, Griffith GW, Griffiths K, Groenewald JZ, Groenewald M, Grube M, Gryzenhout M, Guo LD, Hagen F, Hambleton S, Hamelin RC, Hansen K, Harrold P, Heller G, Herrera C, Hirayama K, Hirooka Y, Ho HM, Hoffmann K, Hofstetter V, Hognabba F, Hollingsworth PM, Hong SB, Hosaka K, Houbraken J, Hughes K, Huhtinen S, Hyde KD, James T, Johnson EM, Johnson JE, Johnston PR, Jones EBG, Kelly LJ, Kirk PM, Knapp DG, Koljalg U, Kovacs GM, Kurtzman CP, Landvik S, Leavitt SD, Liggenstoffer AS, Liimatainen K, Lombard L, Luangsa-ard JJ, Lumbsch HT, Maganti H, Maharachchikumbura SSN, Martin MP, May TW, McTaggart AR, Methven AS, Meyer W, Moncalvo JM, Mongkolsamrit S, Nagy LG, Nilsson RH, Niskanen T, Nyilasi I, Okada G, Okane I, Olariaga I, Otte J, Papp T, Park D, Petkovits T, Pino-Bodas R, Quaedvlieg W, Raja HA, Redecker D, Rintoul TL, Ruibal C, Sarmiento-Ramirez JM, Schmitt I, Schussler A, Shearer C, Sotome K, Stefani FOP, Stenroos S, Stielow B, Stockinger H, Suetrong S, Suh SO, Sung GH, Suzuki M, Tanaka K, Tedersoo L, Telleria MT, Tretter E, Untereiner WA, Urbina H, Vagvolgyi C, Vialle A, Vu TD, Walther G, Wang QM, Wang Y, Weir BS, Weiss M, White MM, Xu J, Yahr R, Yang ZL, Yurkov A, Zamora JC, Zhang N, Zhuang WY, Schindel D (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci 109:6241–6246. https://doi.org/10.1073/pnas.1117018109
Slansky F (1979) Effect of the lichen chemicals atranorin and vulpinic acid upon feeding and growth of larvae of the yellow-striped armyworm, Spodoptera ornithogalli. Environ Entomol 8:865–868. https://doi.org/10.1093/ee/8.5.865
Solhaug KA, Gauslaa Y (2001) Acetone rinsing-a method for testing ecological and physiological roles of secondary compounds in living lichens. Symbiosis 30:301–315
Solhaug KA, Gauslaa Y (2012) Secondary lichen compounds as protection against excess solar radiation and herbivores. In: Lüttge U, Beyschlag W, Büdel B, Francis D (eds) Progress in Botany, 73rd edn. Springer Berlin Heidelberg, pp 283–304
Speiser B (2001) Food and feeding behaviour. In: Baker G (ed) The Biology of Terrestrial Molluscs, Baker GM. CAB International, UK, pp 259–288
Stocker-Wörgötter E (2008) Metabolic diversity of lichen-forming ascomycetous fungi: culturing, polyketide and shikimatemetabolite production, and PKS genes. Nat Prod Rep 25:188–200. https://doi.org/10.1039/B606983P
Van den Berg RA, Hoefsloot HC, Westerhuis JA et al (2006) Centering, scaling, and transformations: improving the biological information content of metabolomics data. BMC Genomics 7:142. https://doi.org/10.1186/1471-2164-7-142
Van der Horst DJ, Zandee DI (1973) Invariability of the composition of fatty acids and other lipids in the pulmonate land snail Cepaea nemoralis (L.) during an annual cycle. J Comp Physiol A 85:317–326. https://doi.org/10.1007/BF00696388
Vu TH, Catheline D, Delmail D et al (2016) Gas chromatographic analysis to compare the fatty acid composition of fifteen lichen species, with a focus on Stereocaulon. Species Lichenologist 48:323–337. https://doi.org/10.1017/S0024282916000141
Wieners PC, Bilger W, Gauslaa Y (2018) Carbon-based secondary compounds in the lichen Hypogymnia physodes deter detrivorous woodlice. Fungal Ecol 31:54–58. https://doi.org/10.1016/j.funeco.2017.11.002
Yosioka I, Hino K, Fujio M, Kitagawa I (1973) The structure of caloploicin, a new lichen trichloro-depsidone. Chem Pharm Bull 21:1547–1553. https://doi.org/10.1248/cpb.21.1547
Acknowledgements
Aurélie BERNARD, Corentin DAUGAN and Nathalie MARNET are acknowledged for their technical assistance. The authors are also indebted to David RONDEAU for giving access to DART-HRMS (DReAM platform, IETR) and Arnaud BONDON and Sandrine POTTIER (PRISM, BioGenOuest) for NMR material access. This work used analytical facilities of the P2M2 platform for primary metabolites (amino acids and carbohydrates) analyses thanks to Alain BOUCHEREAU. The field trip to the Subantarctic was funded by l’Institut Polaire Paul-Émile Victor, Plouzané, France (IPEV, programme 136). Aude BOUTET and Julien TOMMASINO are warmly thanked for their help during the fieldwork. Prof. Yngvar GAUSLAA is warmly acknowledged for his useful comments and discussions about this manuscript.
Funding
This research was funded by the French Polar Institute (Institut Polaire Paul-Émile Victor) within the context of the program 136 “SUBANTECO”. Collecting and transport of specimens of the species Notodiscus hookeri were authorized by the Prefect of Ille-et-Vilaine, France, licence N°35–120 delivered in October 2015.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 947 kb)
Rights and permissions
About this article
Cite this article
Gadea, A., Le Lamer, AC., Le Gall, S. et al. Intrathalline Metabolite Profiles in the Lichen Argopsis friesiana Shape Gastropod Grazing Patterns. J Chem Ecol 44, 471–482 (2018). https://doi.org/10.1007/s10886-018-0953-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-0953-1