Abstract
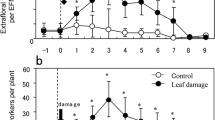
Plants secrete extrafloral nectar (EFN) that attracts predators. The efficiency of the resulting anti-herbivore defense depends on the quantity and spatial distribution of EFN. Thus, according to the optimal defense hypothesis (ODH), plants should secrete EFN on the most valuable organs and when herbivore pressure is high. Ricinus communis plants secreted most EFN on the youngest (i.e., most valuable) leaves and after the simulation of herbivory via the application of jasmonic acid (JA). Here, we investigated the physiological mechanisms that might produce these seemingly adaptive spatiotemporal patterns. Cell wall invertase (CWIN; EC 3.2.1.26) was most active in the hours before peak EFN secretion, its decrease preceded the decrease in EFN secretion, and CWIN activity was inducible by JA. Thus, CWIN appears to be a central player in EFN secretion: its activation by JA is likely to cause the induction of EFN secretion after herbivory. Shading individual leaves decreased EFN secretion locally on these leaves with no effect on CWIN activity in the nectaries, which is likely to be because it decreased the content of sucrose, the substrate of CWIN, in the phloem. Our results demonstrate how the interplay of two physiological processes can cause ecologically relevant spatiotemporal patterns in a plant defense trait.






Similar content being viewed by others
References
Appel HM, Arnold TM, Schultz JC (2012) Effects of jasmonic acid, branching and girdling on carbon and nitrogen transport in poplar. New Phytol 195:419–426
Arnold TM, Schultz JC (2002) Induced sink strength as a prerequisite for induced tannin biosynthesis in developing leaves of Populus. Oecologia 130:585–593
Arnold T, Appel HM, Patel V, Stocum E, Kavalier A, Schultz JC (2004) Carbohydrate translocation determines the phenolic content of Populus foliage: a test of the sink–source model of plant defense. New Phytol 164:157–164
Baker DA, Hall JL, Thorpe JR (1978) A study of the extrafloral nectaries of Ricinus communis. New Phytol 81:129–137
Bixenmann RJ, Coley PD, Kursar TA (2011) Is extrafloral nectar production induced by herbivores or ants in a tropical facultative ant-plant mutualism? Oecologia 165:417–425
Bogatek R, Côme D, Corbineau F, Ranjan R, Lewak S (2002) Jasmonic acid affects dormancy and sugar catabolism in germinating apple embryos. Plant Physiol Biochem 40:167–173
Bruinsma M, Ildema H, van Loon JJA, Dicke M (2008) Differential effects of jasmonic acid treatment of Brassica nigra on the attraction of pollinators, parasitoids, and butterflies. Entomol Exp Appl 128:109–116
Castrillon-Arbelaez P, Martinez-Gallardo N, Arnaut H, Tiessen A, Delano-Frier J (2012) Metabolic and enzymatic changes associated with carbon mobilization, utilization and replenishment triggered in grain amaranth (Amaranthus cruentus) in response to partial defoliation by mechanical injury or insect herbivory. BMC Plant Biol 12:163
Chamberlain SA, Holland JN (2009) Quantitative synthesis of context dependency in ant-plant protection mutualisms. Ecology 90:2384–2392
Claussen W, Loveys BR, Hawker JS (1985) Comparative investigations on the distribution of sucrose synthase activity and invertase activity within growing, mature and old leaves of some C3 and C4 plant species. Physiol Plant 65:275–280
Escalante-Pérez M, Jaborsky M, Lautner S, Fromm J, Müller T, Dittrich M, Kunert M, Boland W, Hedrich R, Ache P (2012) Poplar extrafloral nectaries: two types, two strategies of indirect defenses against herbivores. Plant Physiol 159:1176–1191
Gaffal KP (2012) How common is the ability of extrafloral nectaries to produce nectar droplets, to secrete nectar during the night and to store starch? Plant Biol 14:691–695
González-Teuber M, Heil M (2009) Nectar chemistry is tailored for both attraction of mutualists and protection from exploiters. Plant Signal Behav 4:809–813
Heil M (2004) Induction of two indirect defences benefits Lima bean (Phaseolus lunatus, Fabaceae) in nature. J Ecol 92:527–536
Heil M (2008) Indirect defence via tritrophic interactions. New Phytol 178:41–61
Heil M (2011) Nectar: generation, regulation and ecological functions. Trends Plant Sci 16:191–200
Heil M, Koch T, Hilpert A, Fiala B, Boland W, Linsenmair KE (2001) Extrafloral nectar production of the ant-associated plant, Macaranga tanarius, is an induced, indirect, defensive response elicited by jasmonic acid. Proc Natl Acad Sci U S A 98:1083–1088
Heil M, Greiner S, Meimberg H, Krüger R, Noyer J-L, Heubl G, Linsenmair KE, Boland W (2004) Evolutionary change from induced to constitutive expression of an indirect plant resistance. Nature 430:205–208
Heil M, Büchler R, Boland W (2005) Quantification of invertase activity in ants under field conditions. J Chem Ecol 30:431–437
Heil M, Barajas-Barron A, Orona-Tamayo D, Wielsch N, Svatos A (2014) Partner manipulation stabilises a horizontally transmitted mutualism. Ecol Lett 17:185–192
Herbers K, Meuwly P, Frommer WB, Metraux J-P, Sonnewald U (1996) Systemic acquired resistance mediated by the ectopic expression of invertase: possible hexose sensing in the secretory pathway. Plant Cell 8:793–803
Holland JN, Chamberlain SA, Horn KC (2009) Optimal defence theory predicts investment in extrafloral nectar resources in an ant-plant mutualism. J Ecol 97:89–96
Kaplan I, Halitschke R, Kessler A, Rehill BJ, Sardanelli S, Denno RF (2008) Physiological integration of roots and shoots in plant defense strategies links above-and belowground herbivory. Ecol Lett 11:841–851
Koricheva J, Romero GQ (2012) You get what you pay for: reward-specific trade-offs among direct and ant-mediated defences in plants. Biol Lett 8:628–630
Landgraf R, Schaarschmidt S, Hause B (2013) Repeated leaf wounding alters the colonization of Medicago truncatula roots by beneficial and pathogenic microorganisms. Plant Cell Environ 35:1344–1357
Lohaus G, Burba M, Heldt HW (1994) Comparison of the contents of sucrose and amino acids in the leaves, phloem sap and taproots of high and low sugar-producing hybrids of sugar beet (Beta vulgaris L.). J Exp Bot 45:1097–1101
Lunn JE, Hatch MD (1995) Primary partitioning and storage of photosynthate in sucrose and starch in leaves of C4 plants. Planta 197:385–391
Lüttge U (2013) Green nectaries: the role of photosynthesis in secretion. J Linn Soc Bot 173:1–11
Martin T, Frommer WB, Salanoubat M, Willmitzer L (1993) Expression of an Arabidopsis sucrose synthase gene indicates a role in metabolization of sucrose both during phloem loading and in sink organs. Plant J 4:367–377
McKey D (1974) Adaptive patterns in alkaloid physiology. Am Nat 108:305–320
McKey D (1979) The distribution of secondary compounds within plants. In: Rosenthal GA, Janzen DH (eds) Herbivores: their interactions with secondary plant metabolites. Academic Press, New York, pp 55–133
Nichol P, Hall JL (1988) Characteristics of nectar secretion by the extrafloral nectaries of Ricinus communis. J Exp Bot 39:573–586
Ohyama A, Nishimura S, Hirai M (1998) Cloning of cDNA for a cell wall-bound acid invertase from tomato (Lycopersicon esculentum) and expression of soluble and cell wall-bound invertases in plants and wounded leaves of L. esculentum and L. peruvianum. Genes Gen Syst 73:149–157
Orona-Tamayo D, Wielsch N, Escalante-Pérez M, Svatos A, Molina-Torres J, Muck A, Ramirez-Chávez E, Ádame-Alvarez R-M, Heil M (2013) Short-term proteomic dynamics reveal metabolic factory for active extrafloral nectar secretion by Acacia cornigera ant-plants. Plant J 73:546–554
Pharr DM, Sox HN (1984) Changes in carbohydrate and enzyme levels during the sink to source transition of leaves of Cucumis sativus L., a stachyose translocator. Plant Sci Lett 35:187–193
Radhika V, Kost C, Bartram S, Heil M, Boland W (2008) Testing the optimal defence hypothesis for two indirect defences: extrafloral nectar and volatile organic compounds. Planta 228:449–457
Radhika V, Kost C, Boland W, Heil M (2010a) The role of jasmonates in floral nectar secretion. PLoS One 5:e9265
Radhika V, Kost C, Mithöfer A, Boland W (2010b) Regulation of extrafloral nectar secretion by jasmonates in lima bean is light dependent. Proc Natl Acad Sci U S A 107:17228–17233
Ren G, Healy RA, Klyne AM, Horner HT, James MG, Thornburg RW (2007) Transient starch metabolism in ornamental tobacco floral nectaries regulates nectar composition and release. Plant Sci 173:277–290
Rhoades DF (1979) Evolution of plant chemical defense against herbivores. In: Rosenthal GA, Janzen DH (eds) Herbivores: their interaction with secondary plant metabolites. Academic Press, New York, pp 4–53
Rogers WE, Siemann E, Lankau RA (2003) Damage induced production of extrafloral nectaries in native and invasive seedlings of Chinese tallow tree (Sapium sebiferum). Am Midl Nat 149:413–417
Roitsch T, González M-C (2004) Function and regulation of plant invertases: sweet sensations. Trends Plant Sci 9:606–613
Rostás M, Eggert K (2008) Ontogenetic and spatio-temporal patterns of induced volatiles in Glycine max in the light of the optimal defence hypothesis. Chemoecology 18:29–38
Rosumek FB, Silveira FAO, Neves FD, Barbosa NPD, Diniz L, Oki Y, Pezzini F, Fernandes GW, Cornelissen T (2009) Ants on plants: a meta-analysis of the role of ants as plant biotic defenses. Oecologia 160:537–549
Ruhlmann JM, Kram BW, Carter CJ (2010) CELL WALL INVERTASE 4 is required for nectar production in Arabidopsis. J Exp Bot 61:395–404
Samarakoon AB, Rauser WE (1979) Carbohydrate levels and photoassimilate export from leaves of Phaseolus vulgaris exposed to excess cobalt, nickel, and zinc. Plant Physiol 63:1165–1169
Schaarschmidt S, González M-C, Roitsch T, Strack D, Sonnewald U, Hause B (2007) Regulation of arbuscular mycorrhization by carbon. The symbiotic interaction cannot be improved by increased carbon availability accomplished by root-specifically enhanced invertase activity. Plant Physiol 143:1827–1840
Schaffer AA, Sagee O, Goldschmidt EE, Goren R (1987) Invertase and sucrose synthase activity, carbohydrate status and endogenous IAA levels during citrus leaf development. Physiol Plant 69:151–155
Stephenson AG (1982) The role of the extrafloral nectaries of Catalpa speciosa in limiting herbivory and increasing fruit production. Ecology 63:663–669
Sturm A, Chrispeels MJ (1990) cDNA cloning of carrot extracellular beta-fructosidase and its expression in response to wounding and bacterial infection. Plant Cell 2:1107–1119
Turgeon R (1989) The sink-source transition in leaves. Annu Rev Plant Biol 40:119–138
Vassey TL (1989) Light/dark profiles of sucrose phosphate synthase, sucrose synthase, and acid invertase in leaves of sugar beets. Plant Physiol 89:347–351
Wäckers FL, Bonifay C (2004) How to be sweet? Extrafloral nectar allocation by Gossypium hirsutum fits optimal defense theory predictions. Ecology 85:1512–1518
Wäckers FL, Zuber D, Wunderlin R, Keller F (2001) The effect of herbivory on temporal and spatial dynamics of foliar nectar production in cotton and castor. Ann Bot 87:365–370
Weber MG, Keeler KH (2013) The phylogenetic distribution of extrafloral nectaries in plants. Ann Bot 111:1251–1261
Wooley SC, Donaldson JR, Gusse AC, Lindroth RL, Stevens MT (2007) Extrafloral nectaries in aspen (Populus tremuloides): heritable genetic variation and herbivore-induced expression. Ann Bot 100:1337–1346
Zangerl AR, Rutledge CE (1996) The probability of attack and patterns of constitutive and induced defense: a test of optimal defense theory. Am Nat 147:599–608
Zhang L, Cohn NS, Mitchell JP (1996) Induction of a pea cell-wall invertase gene by wounding and its localized expression in phloem. Plant Physiol 112:1111–1117
Acknowledgments
We thank Antonio Cisneros for taking the photographs of R. communis, CINVESTAV-Irapuato for providing all field facilities and Caroline Woods as well as three anonymous referees for multiple valuable comments on an earlier version of this manuscript. CONACyT of México is gratefully acknowledged for financial support to DOT (grant: 191236) and MH (project grants: 129678 and 130656).
Author information
Authors and Affiliations
Corresponding author
Additional information
Cynthia Millán-Cañongo and Domancar Orona-Tamayo have contributed equally to this paper
Rights and permissions
About this article
Cite this article
Millán-Cañongo, C., Orona-Tamayo, D. & Heil, M. Phloem Sugar Flux and Jasmonic Acid-Responsive Cell Wall Invertase Control Extrafloral Nectar Secretion in Ricinus communis . J Chem Ecol 40, 760–769 (2014). https://doi.org/10.1007/s10886-014-0476-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-014-0476-3