Abstract
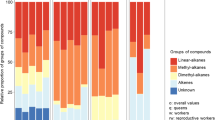
During evolution, the cuticle surface of insects acquired functions in communication, such as inter- and intra-specific recognition, identification of gender, physiological state, and fertility. In eusocial bees, the information in the cuticular surface is important not only to discriminate nestmates from non-nestmates but also to identify an individual’s class, life phase or task. A comparative study of the cuticular surface chemical profile of workers of Scaptotrigona postica in different phases of life, i.e., newly emerged workers (NE), brood comb area workers (CA), and forager workers (FO) was undertaken by gas chromatography linked to mass spectrometry. Multivariate statistical analysis was performed to verify how workers are grouped according to their chemical profile and to determine which compounds are responsible for separating them into groups. The cuticle surface of workers contains mainly hydrocarbons and a small amount of oxygenated compounds. Multivariate statistical analysis showed qualitative and quantitative variation in relation to the life phases/tasks performed, and all groups were distinct. The most abundant compound found in NE and CA was n-heptacosane, while in FO, it was (Z)-9-heptacosene. The compounds that differentiate NE from other groups are n-tricosane and n-hexacosane. A (Z)-X-octacosene and n-nonacosane are the chemicals that distinguish CA from NE and FO, while 11- and 13-methylpentacosane, (Z)-X-hexacosene, and (Z)-9-heptacosene characterize FO as distinct from NE and CA. The probable function of alkenes is nestmate recognition, mainly in FO. The results show that the cuticle surfaces of workers are characteristic of the phase of life/task performed by workers, allowing intra-colonial recognition.




Similar content being viewed by others
References
Abdalla FC, Jones GR, Morgan ED, Cruz-Landim C (2003) Ccomparative study of the cuticular hydrocarbons composition of Melipona bicolor Lepeletier, 1836 (Hymenoptera, Meliponini) workers and queens. Genet Mol Res 2:191–199
Baracchi D, Turillazzi S (2010) Differences in venom and cuticular peptides in individuals of Apis mellifera (Hymenoptera: Apidae) determined by MALDI-TOF MS. J Insect Physiol 56:366–375
Baracchi D, Dapporto L, Teseo S, Hashim R, Turillazzi S (2010) Medium molecular weight polar substances of the cuticle as tools in the study of the taxonomy, systematics and chemical ecology of tropical hover wasps (Hymenoptera: Stenogastrinae). J ZoolSyst Evol Res 48:109–114
Baracchi D, Fadda A, Turillazzi S (2012) Evidence for antiseptic behaviour towards sick adult bees in honey bee colonies. J Insect Physiol 58:1589–1596
Blomquist GJ, Dillwith JW (1985) Cuticular lipids. In: Kerkut GA, Gilbert LI (eds) Comprehensive insect physiology, biochemistry and pharmacology. Pergamon Press, Oxford, pp 117–154
Blomquist GJ, Jackson LL (1979) Chemistry and biochemistry of insect waxes. Prog Lipid Res 17:319–345
Blomquist GJ, Roubick DW, Buchmann SL (1985) Wax chemisty of two stingless bees of the Trigonisca group (Apidae, Meliponinae). Comp Biochem Physiol 82B:137–142
Blomquist GJ, Nelson DR, De Renobales M (1987) Chemistry, biochemistry and physiology of insect cuticular lipids. Arch Insect Biochem Physiol 6:227–265
Blomquist GJ, Tillman JA, Mpuru S, Seybold SJ (1998) The cuticle and cuticular hydrocarbons of insects: structure, function and biochemistry. In: Van der Meer RK, Breed MD, Winston ML, Espelie EK (eds) Pheromone communication in social insects. Westview, Boulder, pp 35–54
Breed MD, Williams KR, Fewell JH (1988) Comb wax mediates the acquisition of nest-mate recognition cues in honeybees. Proc Natl Acad Sci U S A 85:8766–8769
Breed MD, GarryMF PAN, Hibbard BE, Bjostad LB, Page RE (1995) The role of wax comb in honey bee nestmate recognition. Anim Behav 50:489–496
Breed MD, Peery S, Bjostad LB (2004) Testing the blank slate hypothesis; why honey bee colonies accept young bees? Insect Soc 51:12–16
Buchwald R, Breed MD (2005) Nestmate recognition cues in a stingless bee, Trigona fulviventris. Anim Behav 70:1331–1337
Buckner JL (1993) Cuticular polar lipids in insects. In: Stanley-Samuelson DW, Nelson DR (eds) Insect lipids: chemistry, biochemistry and biology. University of Nebraska Press, Nebraska, pp 227–270
Carlin NF (1989) Discrimination between and within colonies of social insects: two null hypotheses. Neth J Zool 39:86–100
Carlson DA, Roan CS, Yost RA, Hector J (1989) Dimethyl disulfide derivatives of long-chain alkenes, alkadienes, and alkatrienes for gas-chromatography mass-spectrometry. Anal Chem 61:1564–1571
Chapman RF (1998) The insects: structure and function. Cambridge University Press, Cambridge, UK
Clément J-L, Bagnères AG (1998) Nestmate recognition in termites. In: Vander Meer RK, Breed MD, Winston ML, Espelie EK (eds) Pheromone communication in social insects. Westview, Boulder, pp 57–58
Cornette R, Farine JP, Quennedey B, Riviere S, Brossut R (2002) Molecular characterization of Lmap54, a new epicuticular surface protein in the cockroach Leucophaea maderae (Dictyoptera, Oxyhaloinae). Insect Biochem Mol Biol 32:1635–1642
Cruz-Landim C, Stort AC, Cruz MAC, Kitajima EW (1965) Órgão tibial dos machos de Euglossini. Estudo ao microscópio óptico e eletrônico. Rev Bras Biol 25:323–342
Cruz-Landim C, Ferreira-Caliman MJ, Gracioli-Vitti LF, Zucchi R (2012) Correlation between mandibular gland secretion and cuticular hydrocarbons in the stingless bee Melipona quadrifasciata. Gen Mol Res 11:966–977
Dani FR, Jones GR, Destri S, Spencer SH, Turillazi S (2001) Deciphering the recognition signature within the cuticular chemical profile of paper wasps. Anim Behav 62:165–171
Dani FR, Jones GR, Corsi S, Beard R, Pradella D, Turillazzi S (2005) Nestmate recognition cues in the honey bee: differential importance of cuticular alkanes and alkenes. Chem Senses 30:477–489
Ferreira-Caliman JM, Nascimento FS, Turatti IC, Mateus S, Lopes NP, Zucchi R (2010) The cuticular hydrocarbons profiles in the stingless bee Melipona marginata reflect task-related differences. J Insect Physiol 56:800–804
Free JB (1961) The social organization of the bumblebee colony. The Central Association of Bee-Keepers, Fleet
Gamboa JG, Grudzien TA, Espelie KE, Bura EA (1996) Kin recognition in social wasps: combining chemical and behavioural evidence. Anim Behav 51:625–629
Gibbs A (2002) Lipid melting and cuticular permeability: new insights into an old problem. J Insect Physiol 48:391–400
Hangartner W, Reichson JM, Wilson EO (1970) Orientation to nest material by the ant, Pogonoimyrmex badius (Latreille). Anim Behav 18:331–334
Howard RW (1993) Cuticular hydrocarbons and chemical communication. In: Stanley-Samuelson DW, Nelson DR (eds) Insect lipids: chemistry, biochemistry and biology. University of Nebraska Press, Lincoln, pp 179–226
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393
Hubbard MD (1974) Influence of the nest material and colony odour on digging in the ant Solenopsis invicta (Hymenoptera: Formicidae). J Georgia Entomol Soc 9:127–132
Kather R, Drijfhout FP, Martin SJ (2011) Task group differences in cuticular lipids in the honey bee Apis mellifera. J Chem Ecol 37:205–212
Lange R (1960) Über die futterweitergabe zwischen angehörigen verschiedener Waldameisenstaaten. Z Tierpsychol 17:389–401
Liang D, Silverman J (2000) “You are what you eat”: diet modifies cuticular hydrocarbons and nestmate recognition in the Argentine ant Linepithema humile. Naturwissenschaften 87:412–416
Lockey KH (1980) Insect cuticular hydrocarbons. Comp Biochem Physiol 65B:457–462
Lockey KH (1985) Insect cuticular lipids. Comp Biochem Physiol 81B:263–273
Lockey KH (1988) Lipids of the insect cuticle: origin, composition and function. Comp Biochem Physiol 89B:595–645
Lorenzi MC, Bagnères AG, Clément J-L (1996) The role of cuticular hydrocarbons in social insects: is it the same in paper wasps? In: Turillazzi S, West-Eberhard MJ (eds) Natural history and evolution of paper wasps. Oxford University Press, UK, pp 178–189
Monnin T, Peters C (1999) Dominance hierarchy and reproductive conflits among subordinates in a monogynous queenless ant. Behav Ecol 10:23–32
Morgan ED (1990) Preparation of small scale samples from insects for chromatography. Anal Chim Acta 236:227–235
Morgan ED (2004) Biosymthesis in insects. The Royal Society of Chemistry, Cambridge
Nelson DR (1993) Methyl-branched lipids in insects. In: Stanley-Samuelson DW, Nelson DR (eds) Insect lipids: chemistry, biochemistry and biology. University of Nebraska Press, Nebraska, pp 271–315
Nunes TM, Nascimento FS, Turatti ICC, Lopes NP, Zucchi R (2008) Nestmate recognition in a stingless bee: does the similarity of chemical cues determine guard acceptance? Anim Behav 75:1165–1171
Nunes TM, Morgan ED, Drijfhout FP, Zucchi R (2010) Caste-specific cuticular lipids in the stingless bee. Apidologie 41:579–588
Obin MS, Vander Meer MK (1988) Sources of nestmate recognition cues in the imported fire ant Solenopsis invicta Buren (Hymenoptera: Formicidae). Anim Behav 36:1361–1370
Oldham NJ, Billen J, Morgan ED (1994) On the similarity of the Dufour gland secretion and the cuticular hydrocarbons of some bumblebees. Physiol Entomol 19:115–123
Richard F-J, Aubert A, Grozinger CM (2008) Modulation of social interactions by immune stimulation in honey bee, Apis mellifera, workers. BMC Biol 60:50
Singer TL, Espelie KE (1992) Social wasps use nest paper hydrocarbons for nestmate recognition. Anim Behav 44:63–68
Sledge MF, Dani FR, Cervo R, Dapporto L, Turillazzi S (2001) Recognition of social parasites as nestmates: adoption of colony-specific host cuticular odours by the paper wasp parasite Polistes sulficer. Proc Roy Soc London 268:2253–2260
Steinmetz I, Schmolz E, Ruther J (2003) Cuticular lipids as trail pheromone in a social wasp. Proc Roy Soc London 270:385–391
Turillazzi S, Mastrobuoni G, Dani FR, Moneti G, Pieraccini G, La Marca G, Bartolucci G, Perito B, Lambardi D, Cavallini V (2006) Dominulin A and B: two new antibacterial peptides identified on the cuticle and in the venom of the social paper wasp Polistes dominulus using MALDI-TOF, MALDITOF/TOF, and ESI-Ion Trap. J Am Soc Mass Spectrom 17:376–383
van Wilgenburg E, Felden A, Choe D-H, Sulc R, Luo J, Shea KJ, Elgar MA, Tsutsui ND (2012) Learning and discrimination of cuticular hydrocarbonsin a social insect. Biol Lett 8:17–20
Vander Meer RK, Morel L (1998) Nestmate recognition in ants. In: Vander Meer RK, Breed MD, Winston ML, Espelie EK (eds) Pheromone communication in social insects. Westview, Boulder, pp 79–103
Vogel S (1963) Duftdrüsen im Dienste der Bestaubung: Über Bau und Funktion der Osmophor en. Akad Wiss Lit Abh Math Naturwiss KI 1962:599–763
Vogel S (1974) Ólblumen and olsammelnde Bienen. Akad Wiss Lit Abh Math Naturwiss KI Trop Subtrop Pflanz 7:1–267
Zupko K, Sklan D, Lensky Y (1993) Proteins of the honeybee (Apis mellifera L.). Body surface and exocrine gland secretions. J Insect Physiol 39:41–46
Acknowledgments
The authors thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for the fellowship (Proc. 07/56682-1) and the reviewers for their valuable suggestions which helped to improve the article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poiani, S.B., Morgan, E.D., Drijfhout, F.P. et al. Separation of Scaptotrigona postica Workers into Defined Task Groups by the Chemical Profile on Their Epicuticle Wax Layer. J Chem Ecol 40, 331–340 (2014). https://doi.org/10.1007/s10886-014-0423-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-014-0423-3